Allergy Asthma Immunol Res.
2010 Jul;2(3):188-194. 10.4168/aair.2010.2.3.188.
Eosinophilic Bronchitis, Eosinophilia Associated Genetic Variants, and Notch Signaling in Asthma
- Affiliations
-
- 1Genome Research Center for Allergy and Respiratory Disease, Division of Allergy and Respiratory Medicine, Soonchunhyang University Bucheon Hospital, Bucheon, Korea. mdcspark@unitel.co.kr
- KMID: 2133807
- DOI: http://doi.org/10.4168/aair.2010.2.3.188
Abstract
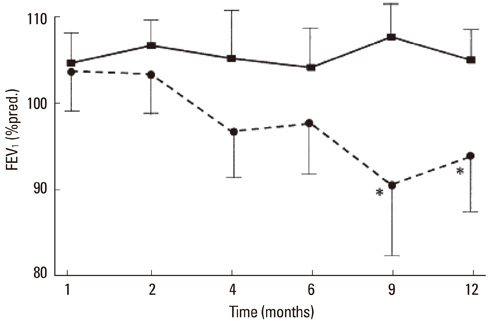
- While much has indeed been learned about the biology and role of eosinophils, the paradigm of eosinophils has the pros and cons in development of asthma. To answer the questions in the black box, this review firstly discusses the biological and morphological differences between asthma and eosinophilic bronchitis (EB). EB is an interesting clinical manifestation of eosinophilic airway disease that does not involve airway hyperresponsiveness (AHR), demonstrating that airway eosinophilia alone is insufficient to merit a diagnosis of asthma. Secondly, I will describe and discuss the effect(s) of single-nucleotide polymorphisms (SNPs) in the genes CCR3, IL-5 RECEPTOR ALPHA (IL5RA), and IL1RL1, and finally the in vitro and in vivo effects of Notch inhibition on both eosinophil differentiation and experimental asthma. Eosinophilic airway inflammation is not as important in the pathogenesis and maintenance of asthma as had previously been thought. However, the role of eosinophils in other asthma subphenotypes, including refractory or severely remodeled asthma, needs to be evaluated further. High-throughput methodologies such as genomics will facilitate the discovery of new markers of inflammation; these, in turn, will aid in the evaluation of the role of eosinophils in asthma and its various subphenotypes.
Keyword
MeSH Terms
Figure
Reference
-
1. Global Initiative for Asthma. Global strategy for asthma management and prevention. NHLBI/WHO workshop report. 1995. Bethesda (MD): National Institutes of Health, National Heart, Lung, and Blood Institute.2. Mould AW, Matthaei KI, Young IG, Foster PS. Relationship between interleukin-5 and eotaxin in regulating blood and tissue eosinophilia in mice. J Clin Invest. 1997. 99:1064–1071.3. Hogan SP, Rosenberg HF, Moqbel R, Phipps S, Foster PS, Lacy P, Kay AB, Rothenberg ME. Eosinophils: biological properties and role in health and disease. Clin Exp Allergy. 2008. 38:709–750.4. Bousquet J, Chanez P, Lacoste JY, Barneon G, Ghavanian N, Enander I, Venge P, Ahlstedt S, Simony-Lafontaine J, Godard P, Michel FB. Eosinophilic inflammation in asthma. N Engl J Med. 1990. 323:1033–1039.5. Leckie MJ, ten Brinke A, Khan J, Diamant Z, O'Connor BJ, Walls CM, Mathur AK, Cowley HC, Chung KF, Djukanovic R, Hansel TT, Holgate ST, Sterk PJ, Barnes PJ. Effects of an interleukin-5 blocking monoclonal antibody on eosinophils, airway hyper-responsiveness, and the late asthmatic response. Lancet. 2000. 356:2144–2148.6. Flood-Page PT, Menzies-Gow AN, Kay AB, Robinson DS. Eosinophil's role remains uncertain as anti-interleukin-5 only partially depletes numbers in asthmatic airway. Am J Respir Crit Care Med. 2003. 167:199–204.7. Gibson PG, Dolovich J, Denburg J, Ramsdale EH, Hargreave FE. Chronic cough: eosinophilic bronchitis without asthma. Lancet. 1989. 1:1346–1348.8. Brightling CE, Ward R, Goh KL, Wardlaw AJ, Pavord ID. Eosinophilic bronchitis is an important cause of chronic cough. Am J Respir Crit Care Med. 1999. 160:406–410.9. Joo JH, Park SJ, Park SW, Lee JH, Kim DJ, Uh ST, Kim YH, Park CS. Clinical features of eosinophilic bronchitis. Korean J Intern Med. 2002. 17:31–37.10. Brightling CE, Symon FA, Birring SS, Bradding P, Pavord ID, Wardlaw AJ. TH2 cytokine expression in bronchoalveolar lavage fluid T lymphocytes and bronchial submucosa is a feature of asthma and eosinophilic bronchitis. J Allergy Clin Immunol. 2002. 110:899–905.11. Brightling CE, Symon FA, Birring SS, Bradding P, Wardlaw AJ, Pavord ID. Comparison of airway immunopathology of eosinophilic bronchitis and asthma. Thorax. 2003. 58:528–532.12. Park SW, Park JS, Lee YM, Lee JH, Jang AS, Kim DJ, Hwangbo Y, Uh ST, Kim YH, Park CS. Differences in radiological/HRCT findings in eosinophilic bronchitis and asthma: implication for bronchial responsiveness. Thorax. 2006. 61:41–47.13. Brightling CE, Bradding P, Symon FA, Holgate ST, Wardlaw AJ, Pavord ID. Mast-cell infiltration of airway smooth muscle in asthma. N Engl J Med. 2002. 346:1699–1705.14. Berry MA, Parker D, Neale N, Woodman L, Morgan A, Monk P, Bradding P, Wardlaw AJ, Pavord ID, Brightling CE. Sputum and bronchial submucosal IL-13 expression in asthma and eosinophilic bronchitis. J Allergy Clin Immunol. 2004. 114:1106–1109.15. Park SW, Jangm HK, An MH, Min JW, Jang AS, Lee JH, Park CS. Interleukin-13 and interleukin-5 in induced sputum of eosinophilic bronchitis: comparison with asthma. Chest. 2005. 128:1921–1927.16. Park SW, Lee YM, Jang AS, Lee JH, Hwangbo Y, Kim DJ, Park CS. Development of chronic airway obstruction in patients with eosinophilic bronchitis: a prospective follow-up study. Chest. 2004. 125:1998–2004.17. Lee YA, Wahn U, Kehrt R, Tarani L, Businco L, Gustafsson D, Andersson F, Oranje AP, Wolkertstorfer A, v Berg A, Hoffmann U, Kuster W, Wienker T, Ruschendorf F, Reis A. A major susceptibility locus for atopic dermatitis maps to chromosome 3q21. Nat Genet. 2000. 26:470–473.18. Kotsimbos AT, Hamid Q. IL-5 and IL-5 receptor in asthma. Mem Inst Oswaldo Cruz. 1997. 92:Suppl 2. 75–91.19. Murata Y, Takaki S, Migita M, Kikuchi Y, Tominaga A, Takatsu K. Molecular cloning and expression of the human interleukin 5 receptor. J Exp Med. 1992. 175:341–351.20. Ober C, Tsalenko A, Parry R, Cox NJ. A second-generation genomewide screen for asthma-susceptibility alleles in a founder population. Am J Hum Genet. 2000. 67:1154–1162.21. Rioux JD, Stone VA, Daly MJ, Cargill M, Green T, Nguyen H, Nutman T, Zimmerman PA, Tucker MA, Hudson T, Goldstein AM, Lander E, Lin AY. Familial eosinophilia maps to the cytokine gene cluster on human chromosomal region 5q31-q33. Am J Hum Genet. 1998. 63:1086–1094.22. Lee JH, Chang HS, Kim JH, Park SM, Lee YM, Uh ST, Rhim T, Chung IY, Kim YH, Park BL, Park CS, Shin HD. Genetic effect of CCR3 and IL5RA gene polymorphisms on eosinophilia in asthmatic patients. J Allergy Clin Immunol. 2007. 120:1110–1117.23. Gudbjartsson DF, Bjornsdottir US, Halapi E, Helgadottir A, Sulem P, Jonsdottir GM, Thorleifsson G, Helgadottir H, Steinthorsdottir V, Stefansson H, Williams C, Hui J, Beilby J, Warrington NM, James A, Palmer LJ, Koppelman GH, Heinzmann A, Krueger M, Boezen HM, Wheatley A, Altmuller J, Shin HD, Uh ST, Cheong HS, Jonsdottir B, Gislason D, Park CS, Rasmussen LM, Porsbjerg C, Hansen JW, Backer V, Werge T, Janson C, Jonsson UB, Ng MC, Chan J, So WY, Ma R, Shah SH, Granger CB, Quyyumi AA, Levey AI, Vaccarino V, Reilly MP, Rader DJ, Williams MJ, van Rij AM, Jones GT, Trabetti E, Malerba G, Pignatti PF, Boner A, Pescollderungg L, Girelli D, Olivieri O, Martinelli N, Ludviksson BR, Ludviksdottir D, Eyjolfsson GI, Arnar D, Thorgeirsson G, Deichmann K, Thompson PJ, Wjst M, Hall IP, Postma DS, Gislason T, Gulcher J, Kong A, Jonsdottir I, Thorsteinsdottir U, Stefansson K. Sequence variants affecting eosinophil numbers associate with asthma and myocardial infarction. Nat Genet. 2009. 41:342–347.24. Carriere V, Roussel L, Ortega N, Lacorre DA, Americh L, Aguilar L, Bouche G, Girard JP. IL-33, the IL-1-like cytokine ligand for ST2 receptor, is a chromatin-associated nuclear factor in vivo. Proc Natl Acad Sci U S A. 2007. 104:282–287.25. Cherry WB, Yoon J, Bartemes KR, Iijima K, Kita H. A novel IL-1 family cytokine, IL-33, potently activates human eosinophils. J Allergy Clin Immunol. 2008. 121:1484–1490.26. Fiuza UM, Arias AM. Cell and molecular biology of Notch. J Endocrinol. 2007. 194:459–474.27. Kang JH, Lee DH, Lee JS, Kim HJ, Shin JW, Lee YH, Lee YS, Park CS, Chung IY. Eosinophilic differentiation is promoted by blockage of Notch signaling with a gamma-secretase inhibitor. Eur J Immunol. 2005. 35:2982–2990.28. Kang JH, Lee da H, Seo H, Park JS, Nam KH, Shin SY, Park CS, Chung IY. Regulation of functional phenotypes of cord blood derived eosinophils by gamma-secretase inhibitor. Am J Respir Cell Mol Biol. 2007. 37:571–577.29. Amsen D, Blander JM, Lee GR, Tanigaki K, Honjo T, Flavell RA. Instruction of distinct CD4 T helper cell fates by different notch ligands on antigen-presenting cells. Cell. 2004. 117:515–526.30. Kang JH, Kim BS, Uhm TG, Lee SH, Lee GR, Park CS, Chung IY. Gamma-secretase inhibitor reduces allergic pulmonary inflammation by modulating Th1 and Th2 responses. Am J Respir Crit Care Med. 2009. 179:875–882.31. Fang TC, Yashiro-Ohtani Y, Del Bianco C, Knoblock DM, Blacklow SC, Pear WS. Notch directly regulates Gata3 expression during T helper 2 cell differentiation. Immunity. 2007. 27:100–110.