Korean J Physiol Pharmacol.
2015 Jan;19(1):51-57. 10.4196/kjpp.2015.19.1.51.
Peptidoglycan Induces the Production of Interleukin-8 via Calcium Signaling in Human Gingival Epithelium
- Affiliations
-
- 1Department of Oral Biology, Yonsei University College of Dentistry, Seoul 120-752, Korea. minicleo@gachon.ac.kr
- 2BK21 PLUS Project, Yonsei University College of Dentistry, Seoul 120-752, Korea.
- 3Department of Physiology, Graduate School of Medicine, Gachon University, Incheon 406-799, Korea.
- KMID: 2071819
- DOI: http://doi.org/10.4196/kjpp.2015.19.1.51
Abstract
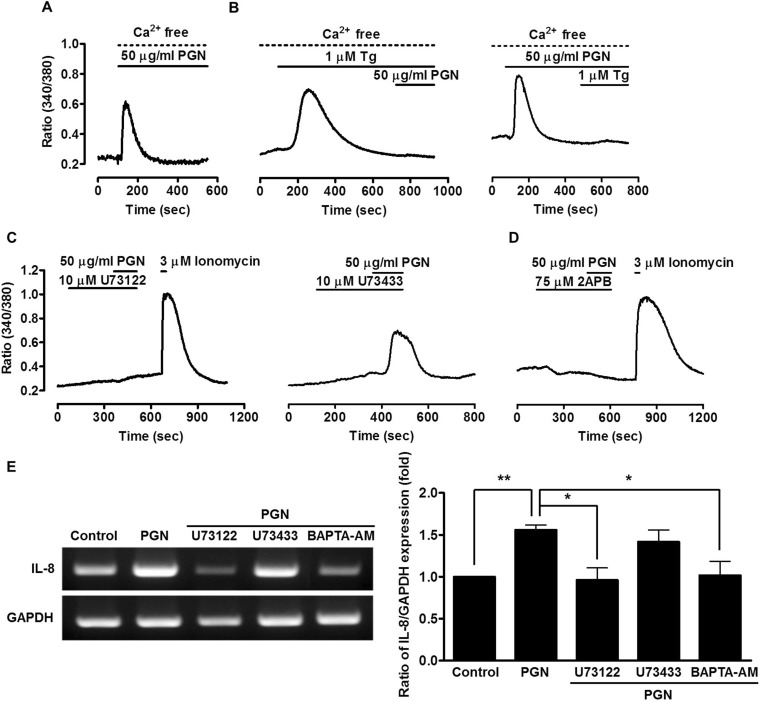
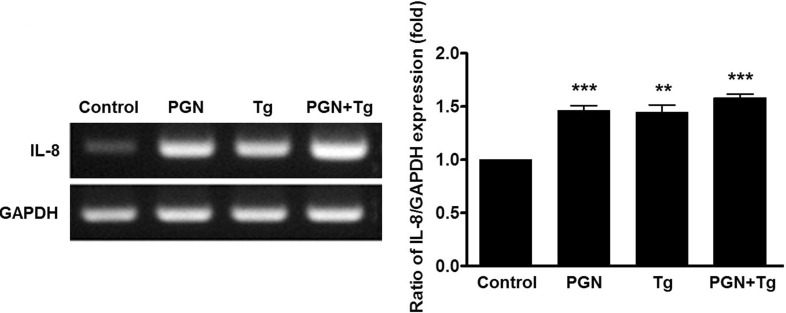
- The etiology of periodontal disease is multifactorial. Exogenous stimuli such as bacterial pathogens can interact with toll-like receptors to activate intracellular calcium signaling in gingival epithelium and other tissues. The triggering of calcium signaling induces the secretion of pro-inflammatory cytokines such as interleukin-8 as part of the inflammatory response; however, the exact mechanism of calcium signaling induced by bacterial toxins when gingival epithelial cells are exposed to pathogens is unclear. Here, we investigate calcium signaling induced by bacteria and expression of inflammatory cytokines in human gingival epithelial cells. We found that peptidoglycan, a constituent of gram-positive bacteria and an agonist of toll-like receptor 2, increases intracellular calcium in a concentration-dependent manner. Peptidoglycan-induced calcium signaling was abolished by treatment with blockers of phospholipase C (U73122), inositol 1,4,5-trisphosphate receptors, indicating the release of calcium from intracellular calcium stores. Peptidoglycan-mediated interleukin-8 expression was blocked by U73122 and 1,2-bis(2-aminophenoxy)ethane-N,N,N',N'-tetraacetic acid tetrakis (acetoxymethyl ester). Moreover, interleukin-8 expression was induced by thapsigargin, a selective inhibitor of the sarco/endoplasmic reticulum calcium ATPase, when thapsigargin was treated alone or co-treated with peptidoglycan. These results suggest that the gram-positive bacterial toxin peptidoglycan induces calcium signaling via the phospholipase C/inositol 1,4,5-trisphosphate pathway, and that increased interleukin-8 expression is mediated by intracellular calcium levels in human gingival epithelial cells.
MeSH Terms
-
Bacteria
Bacterial Toxins
Calcium
Calcium Signaling*
Calcium-Transporting ATPases
Cytokines
Epithelial Cells
Epithelium*
Gram-Positive Bacteria
Humans
Inflammation
Inositol 1,4,5-Trisphosphate Receptors
Interleukin-8*
Peptidoglycan*
Periodontal Diseases
Phospholipases
Reticulum
Thapsigargin
Toll-Like Receptor 2
Toll-Like Receptors
Type C Phospholipases
Bacterial Toxins
Calcium
Calcium-Transporting ATPases
Cytokines
Inositol 1,4,5-Trisphosphate Receptors
Interleukin-8
Peptidoglycan
Phospholipases
Thapsigargin
Toll-Like Receptor 2
Toll-Like Receptors
Type C Phospholipases
Figure
Cited by 1 articles
-
Bacterial PAMPs and Allergens Trigger Increase in [Ca2+]i-induced Cytokine Expression in Human PDL Fibroblasts
Ga-Yeon Son, Dong Min Shin, Jeong Hee Hong
Korean J Physiol Pharmacol. 2015;19(3):291-297. doi: 10.4196/kjpp.2015.19.3.291.
Reference
-
2. Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE. Defining the normal bacterial flora of the oral cavity. J Clin Microbiol. 2005; 43:5721–5732. PMID: 16272510.
Article3. Bik EM, Long CD, Armitage GC, Loomer P, Emerson J, Mongodin EF, Nelson KE, Gill SR, Fraser-Liggett CM, Relman DA. Bacterial diversity in the oral cavity of 10 healthy individuals. ISME J. 2010; 4:962–974. PMID: 20336157.
Article4. Nagata T. Relationhip between diabetes and periodontal disease. Clin Calcium. 2009; 19:1291–1298. PMID: 19721200.5. Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006; 124:783–801. PMID: 16497588.
Article6. Yoshimura A, Kaneko T, Kato Y, Golenbock DT, Hara Y. Lipopolysaccharides from periodontopathic bacteria Porphyromonas gingivalis and Capnocytophaga ochracea are antagonists for human toll-like receptor 4. Infect Immun. 2002; 70:218–225. PMID: 11748186.7. Ueki K, Tabeta K, Yoshie H, Yamazaki K. Self-heat shock protein 60 induces tumour necrosis factor-alpha in monocytederived macrophage: possible role in chronic inflammatory periodontal disease. Clin Exp Immunol. 2002; 127:72–77. PMID: 11882035.8. Nonnenmacher C, Dalpke A, Zimmermann S, Flores-De-Jacoby L, Mutters R, Heeg K. DNA from periodontopathogenic bacteria is immunostimulatory for mouse and human immune cells. Infect Immun. 2003; 71:850–856. PMID: 12540566.
Article9. Darby I, Curtis M. Microbiology of periodontal disease in children and young adults. Periodontol 2000. 2001; 26:33–53. PMID: 11452905.
Article10. Ara T, Kurata K, Hirai K, Uchihashi T, Uematsu T, Imamura Y, Furusawa K, Kurihara S, Wang PL. Human gingival fibroblasts are critical in sustaining inflammation in periodontal disease. J Periodontal Res. 2009; 44:21–27. PMID: 19515019.
Article11. Baik JE, Ryu YH, Han JY, Im J, Kum KY, Yun CH, Lee K, Han SH. Lipoteichoic acid partially contributes to the inflammatory responses to Enterococcus faecalis. J Endod. 2008; 34:975–982. PMID: 18634930.
Article12. Booth V, Downes J, Van den Berg J, Wade WG. Gram-positive anaerobic bacilli in human periodontal disease. J Periodontal Res. 2004; 39:213–220. PMID: 15206913.
Article13. Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004; 4:499–511. PMID: 15229469.
Article14. Sugawara Y, Uehara A, Fujimoto Y, Kusumoto S, Fukase K, Shibata K, Sugawara S, Sasano T, Takada H. Toll-like receptors, NOD1, and NOD2 in oral epithelial cells. J Dent Res. 2006; 85:524–529. PMID: 16723649.
Article15. Medzhitov R. Toll-like receptors and innate immunity. Nat Rev Immunol. 2001; 1:135–145. PMID: 11905821.
Article16. Wang Q, Dziarski R, Kirschning CJ, Muzio M, Gupta D. Micrococci and peptidoglycan activate TLR2-->MyD88-->IRAK-->TRAF-->NIK-->IKK-->NF-kappaB signal transduction pathway that induces transcription of interleukin-8. Infect Immun. 2001; 69:2270–2276. PMID: 11254583.17. Royet J, Dziarski R. Peptidoglycan recognition proteins: pleiotropic sensors and effectors of antimicrobial defences. Nat Rev Microbiol. 2007; 5:264–277. PMID: 17363965.
Article18. Lin HY, Tang CH, Chen JH, Chuang JY, Huang SM, Tan TW, Lai CH, Lu DY. Peptidoglycan induces interleukin-6 expression through the TLR2 receptor, JNK, c-Jun, and AP-1 pathways in microglia. J Cell Physiol. 2011; 226:1573–1582. PMID: 20945380.
Article19. Berridge MJ, Bootman MD, Roderick HL. Calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol. 2003; 4:517–529. PMID: 12838335.
Article20. Newton K, Dixit VM. Signaling in innate immunity and inflammation. Cold Spring Harb Perspect Biol. 2012; 4:pii: a006049.
Article21. Lilienbaum A, Israël A. From calcium to NF-kappa B signaling pathways in neurons. Mol Cell Biol. 2003; 23:2680–2698. PMID: 12665571.22. Firth AL, Won JY, Park WS. Regulation of Ca2+ signaling in pulmonary hypertension. Korean J Physiol Pharmacol. 2013; 17:1–8. PMID: 23439762.23. Aki D, Minoda Y, Yoshida H, Watanabe S, Yoshida R, Takaesu G, Chinen T, Inaba T, Hikida M, Kurosaki T, Saeki K, Yoshimura A. Peptidoglycan and lipopolysaccharide activate PLCgamma2, leading to enhanced cytokine production in macrophages and dendritic cells. Genes Cells. 2008; 13:199–208. PMID: 18233961.24. Xuan NT, Shumilina E, Matzner N, Zemtsova IM, Biedermann T, Goetz F, Lang F. Ca2+-dependent functions in peptidoglycanstimulated mouse dendritic cells. Cell Physiol Biochem. 2009; 24:167–176. PMID: 19710531.25. Chun J, Prince A. Activation of Ca2+-dependent signaling by TLR2. J Immunol. 2006; 177:1330–1337. PMID: 16818794.26. Yang YM, Jung HH, Lee SJ, Choi HJ, Kim MS, Shin DM. TRPM7 Is Essential for RANKL-Induced Osteoclastogenesis. Korean J Physiol Pharmacol. 2013; 17:65–71. PMID: 23440520.
Article27. Chang WC. Store-operated calcium channels and pro-inflammatory signals. Acta Pharmacol Sin. 2006; 27:813–820. PMID: 16787563.
Article28. Sun L, Ye RD. Role of G protein-coupled receptors in inflammation. Acta Pharmacol Sin. 2012; 33:342–350. PMID: 22367283.
Article29. He J, Li Y, Cao Y, Xue J, Zhou X. The oral microbiome diversity and its relation to human diseases. Folia Microbiol (Praha). 2014; [Epub ahead of print].
Article30. Volz T, Nega M, Buschmann J, Kaesler S, Guenova E, Peschel A, Röcken M, Götz F, Biedermann T. Natural Staphylococcus aureus-derived peptidoglycan fragments activate NOD2 and act as potent costimulators of the innate immune system exclusively in the presence of TLR signals. FASEB J. 2010; 24:4089–4102. PMID: 20522786.31. Schüffler H, Demircioglu DD, Kühner D, Menz S, Bender A, Autenrieth IB, Bodammer P, Lamprecht G, Götz F, Frick JS. NOD2 stimulation by Staphylococcus aureus-derived peptidoglycan is boosted by Toll-like receptor 2 costimulation with lipoproteins in dendritic cells. Infect Immun. 2014; 82:4681–4688. PMID: 25156723.32. Müller-Anstett MA, Müller P, Albrecht T, Nega M, Wagener J, Gao Q, Kaesler S, Schaller M, Biedermann T, Götz F. Staphylococcal peptidoglycan co-localizes with Nod2 and TLR2 and activates innate immune response via both receptors in primary murine keratinocytes. PLoS One. 2010; 5:e13153. PMID: 20949035.
Article33. Fleih MT, Al-Mathkhury HJF, Mahmod ZS. The pathological effect of peptidoglycan on rats' lungs part one: pathogenic bacteria streptococcus pneumoniae. J Al-Nahrain Univ. 2007; 10:87–93.
Article34. Eckmann L, Kagnoff MF, Fierer J. Epithelial cells secrete the chemokine interleukin-8 in response to bacterial entry. Infect Immun. 1993; 61:4569–4574. PMID: 8406853.
Article35. Kang SH, Lee JH, Choi KH, Rhim BY, Kim K. Roles of ERK and NF-kappaB in interleukin-8 expression in response to heat shock protein 22 in vascular smooth muscle cells. Korean J Physiol Pharmacol. 2008; 12:171–176. PMID: 19967052.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- House Dust Mite Extract Induces PLC/IP3-dependent Ca2+ Signaling and IL-8 Expression in Human Gingival Epithelial Cells
- The Expression Pattern of Toll-like Receptor (TLR) and Cytokine Production to TLR Agonists in Human Retinal Pigment Epithelial Cells
- IL-17 and Toll-like Receptor 2 or Toll-like Receptor 4 Combined Engagement Upregulates RANKL and IL-6 in Human Rheumatoid Synovial Fibroblasts
- A case of gingival hyperplasia caused by amlodipine
- Peptidoglycan Up-Regulates CXCL8 Expression via Multiple Pathways in Monocytes/Macrophages