Korean J Physiol Pharmacol.
2015 Nov;19(6):515-522. 10.4196/kjpp.2015.19.6.515.
Mind Bomb-2 Regulates Hippocampus-dependent Memory Formation and Synaptic Plasticity
- Affiliations
-
- 1Department of Biological Sciences, College of Natural Sciences, Seoul National University, Seoul 08826, Korea. kaang@snu.ac.kr
- KMID: 2070791
- DOI: http://doi.org/10.4196/kjpp.2015.19.6.515
Abstract
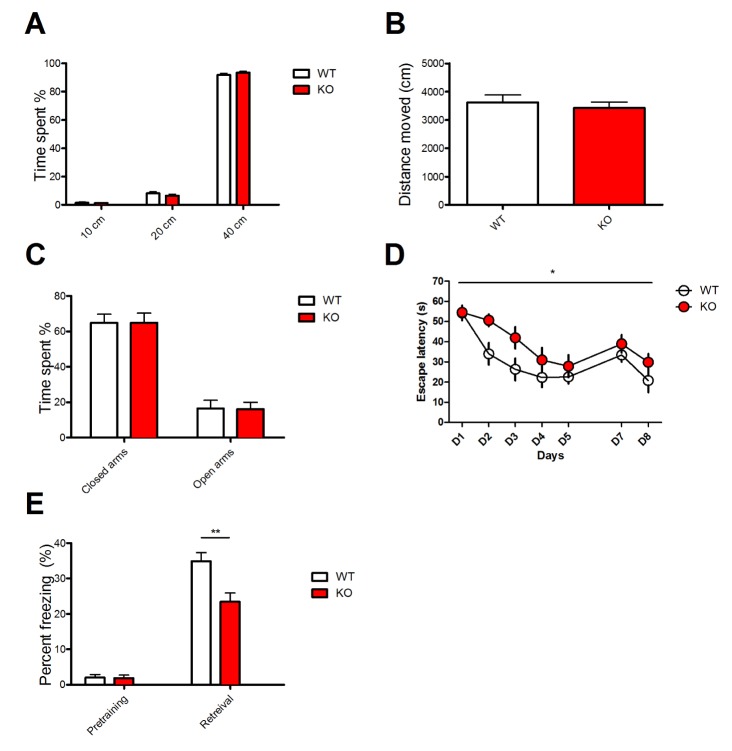
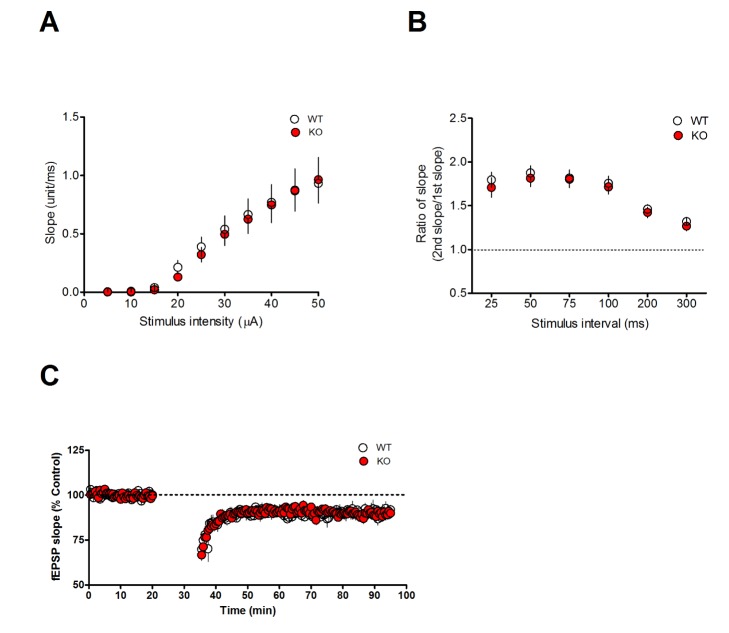
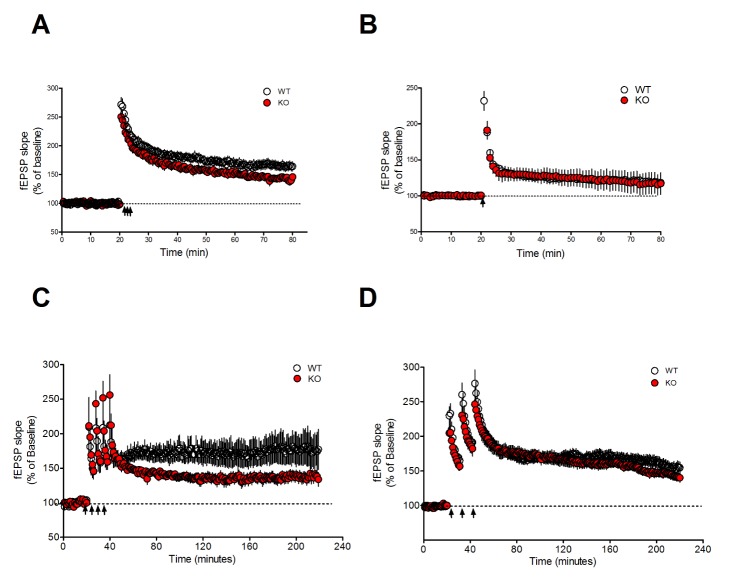
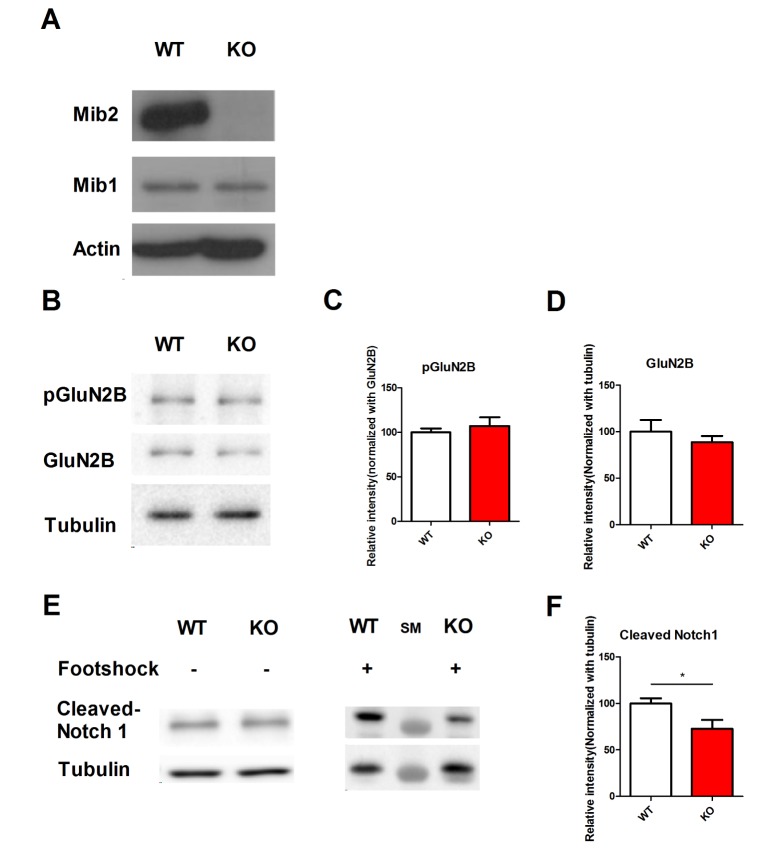
- Notch signaling is a key regulator of neuronal fate during embryonic development, but its function in the adult brain is still largely unknown. Mind bomb-2 (Mib2) is an essential positive regulator of the Notch pathway, which acts in the Notch signal-sending cells. Therefore, genetic deletion of Mib2 in the mouse brain might help understand Notch signaling-mediated cell-cell interactions between neurons and their physiological function. Here we show that deletion of Mib2 in the mouse brain results in impaired hippocampal spatial memory and contextual fear memory. Accordingly, we found impaired hippocampal synaptic plasticity in Mib2 knock-out (KO) mice; however, basal synaptic transmission did not change at the Schaffer collateral-CA1 synapses. Using western blot analysis, we found that the level of cleaved Notch1 was lower in Mib2 KO mice than in wild type (WT) littermates after mild foot shock. Taken together, these data suggest that Mib2 plays a critical role in synaptic plasticity and spatial memory through the Notch signaling pathway.
Keyword
MeSH Terms
Figure
Reference
-
1. Gazave E, Lapébie P, Richards GS, Brunet F, Ereskovsky AV, Degnan BM, Borchiellini C, Vervoort M, Renard E. Origin and evolution of the Notch signalling pathway: an overview from eukaryotic genomes. BMC Evol Biol. 2009; 9:249. PMID: 19825158.
Article2. Sorkin A, Von Zastrow M. Signal transduction and endocytosis: close encounters of many kinds. Nat Rev Mol Cell Biol. 2002; 3:600–614. PMID: 12154371.
Article3. Greenwald I. LIN-12/Notch signaling: lessons from worms and flies. Genes Dev. 1998; 12:1751–1762. PMID: 9637676.4. Artavanis-Tsakonas S, Rand MD, Lake RJ. Notch signaling: cell fate control and signal integration in development. Science. 1999; 284:770–776. PMID: 10221902.
Article5. Mumm JS, Kopan R. Notch signaling: from the outside in. Dev Biol. 2000; 228:151–165. PMID: 11112321.
Article6. Baron M. An overview of the Notch signalling pathway. Semin Cell Dev Biol. 2003; 14:113–119. PMID: 12651094.
Article7. Yoon K, Gaiano N. Notch signaling in the mammalian central nervous system: insights from mouse mutants. Nat Neurosci. 2005; 8:709–715. PMID: 15917835.
Article8. Ge X, Hannan F, Xie Z, Feng C, Tully T, Zhou H, Xie Z, Zhong Y. Notch signaling in Drosophila long-term memory formation. Proc Natl Acad Sci U S A. 2004; 101:10172–10176. PMID: 15220476.
Article9. Grant SG, O'Dell TJ, Karl KA, Stein PL, Soriano P, Kandel ER. Impaired long-term potentiation, spatial learning, and hippocampal development in fyn mutant mice. Science. 1992; 258:1903–1910. PMID: 1361685.10. Sargin D, Botly LC, Higgs G, Marsolais A, Frankland PW, Egan SE, Josselyn SA. Reprint of: disrupting Jagged1-Notch signaling impairs spatial memory formation in adult mice. Neurobiol Learn Mem. 2013; 105:20–30. PMID: 23850596.
Article11. Wang Y, Chan SL, Miele L, Yao PJ, Mackes J, Ingram DK, Mattson MP, Furukawa K. Involvement of Notch signaling in hippocampal synaptic plasticity. Proc Natl Acad Sci U S A. 2004; 101:9458–9462. PMID: 15190179.
Article12. Alberi L, Liu S, Wang Y, Badie R, Smith-Hicks C, Wu J, Pierfelice TJ, Abazyan B, Mattson MP, Kuhl D, Pletnikov M, Worley PF, Gaiano N. Activity-induced Notch signaling in neurons requires Arc/Arg3.1 and is essential for synaptic plasticity in hippocampal networks. Neuron. 2011; 69:437–444. PMID: 21315255.
Article13. Itoh M, Kim CH, Palardy G, Oda T, Jiang YJ, Maust D, Yeo SY, Lorick K, Wright GJ, Ariza-McNaughton L, Weissman AM, Lewis J, Chandrasekharappa SC, Chitnis AB. Mind bomb is a ubiquitin ligase that is essential for efficient activation of Notch signaling by Delta. Dev Cell. 2003; 4:67–82. PMID: 12530964.
Article14. Koo BK, Lim HS, Song R, Yoon MJ, Yoon KJ, Moon JS, Kim YW, Kwon MC, Yoo KW, Kong MP, Lee J, Chitnis AB, Kim CH, Kong YY. Mind bomb 1 is essential for generating functional Notch ligands to activate Notch. Development. 2005; 132:3459–3470. PMID: 16000382.
Article15. Koo BK, Yoon MJ, Yoon KJ, Im SK, Kim YY, Kim CH, Suh PG, Jan YN, Kong YY. An obligatory role of mind bomb-1 in notch signaling of mammalian development. PLoS One. 2007; 2:e1221. PMID: 18043734.16. Koo BK, Yoon KJ, Yoo KW, Lim HS, Song R, So JH, Kim CH, Kong YY. Mind bomb-2 is an E3 ligase for Notch ligand. J Biol Chem. 2005; 280:22335–22342. PMID: 15824097.
Article17. Zhang C, Li Q, Lim CH, Qiu X, Jiang YJ. The characterization of zebrafish antimorphic mib alleles reveals that Mib and Mind bomb-2 (Mib2) function redundantly. Dev Biol. 2007; 305:14–27. PMID: 17331493.
Article18. Yoon KJ, Lee HR, Jo YS, An K, Jung SY, Jeong MW, Kwon SK, Kim NS, Jeong HW, Ahn SH, Kim KT, Lee K, Kim E, Kim JH, Choi JS, Kaang BK, Kong YY. Mind bomb-1 is an essential modulator of long-term memory and synaptic plasticity via the Notch signaling pathway. Mol Brain. 2012; 5:40. PMID: 23111145.
Article19. Choi JH, Park P, Baek GC, Sim SE, Kang SJ, Lee Y, Ahn SH, Lim CS, Lee YS, Collingridge GL, Kaang BK. Effects of PI3Kγ overexpression in the hippocampus on synaptic plasticity and spatial learning. Mol Brain. 2014; 7:78. PMID: 25373491.
Article20. Vorhees CV, Williams MT. Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc. 2006; 1:848–858. PMID: 17406317.
Article21. Hayashi Y, Nabeshima Y, Kobayashi K, Miyakawa T, Tanda K, Takao K, Suzuki H, Esumi E, Noguchi S, Matsuda Y, Sasaoka T, Noda T, Miyazaki J, Mishina M, Funabiki K, Nabeshima Y. Enhanced stability of hippocampal place representation caused by reduced magnesium block of NMDA receptors in the dentate gyrus. Mol Brain. 2014; 7:44. PMID: 24893573.
Article22. Karpova A, Mikhaylova M, Thomas U, Knöpfel T, Behnisch T. Involvement of protein synthesis and degradation in long-term potentiation of Schaffer collateral CA1 synapses. J Neurosci. 2006; 26:4949–4955. PMID: 16672670.
Article23. Sanhueza M, Lisman J. The CaMKII/NMDAR complex as a molecular memory. Mol Brain. 2013; 6:10. PMID: 23410178.
Article24. Bliss TV, Collingridge GL. Expression of NMDA receptor-dependent LTP in the hippocampus: bridging the divide. Mol Brain. 2013; 6:5. PMID: 23339575.
Article25. Lynch MA. Long-term potentiation and memory. Physiol Rev. 2004; 84:87–136. PMID: 14715912.
Article26. Siarey RJ, Kline-Burgess A, Cho M, Balbo A, Best TK, Harashima C, Klann E, Galdzicki Z. Altered signaling pathways underlying abnormal hippocampal synaptic plasticity in the Ts65Dn mouse model of Down syndrome. J Neurochem. 2006; 98:1266–1277. PMID: 16895585.
Article27. Selcher JC, Weeber EJ, Christian J, Nekrasova T, Landreth GE, Sweatt JD. A role for ERK MAP kinase in physiologic temporal integration in hippocampal area CA1. Learn Mem. 2003; 10:26–39. PMID: 12551961.
Article28. Zhu G, Liu Y, Wang Y, Bi X, Baudry M. Different patterns of electrical activity lead to long-term potentiation by activating different intracellular pathways. J Neurosci. 2015; 35:621–633. PMID: 25589756.
Article29. Costa AC, Grybko MJ. Deficits in hippocampal CA1 LTP induced by TBS but not HFS in the Ts65Dn mouse: a model of Down syndrome. Neurosci Lett. 2005; 382:317–322. PMID: 15925111.
Article30. Jurd R, Thornton C, Wang J, Luong K, Phamluong K, Kharazia V, Gibb SL, Ron D. Mind bomb-2 is an E3 ligase that ubiquitinates the N-methyl-D-aspartate receptor NR2B subunit in a phosphorylation-dependent manner. J Biol Chem. 2008; 283:301–310. PMID: 17962190.
Article31. Costa RM, Honjo T, Silva AJ. Learning and memory deficits in Notch mutant mice. Curr Biol. 2003; 13:1348–1354. PMID: 12906797.
Article32. Ressler KJ, Paschall G, Zhou XL, Davis M. Regulation of synaptic plasticity genes during consolidation of fear conditioning. J Neurosci. 2002; 22:7892–7902. PMID: 12223542.
Article33. Kelleher RJ, Govindarajan A, Tonegawa S. Translational regulatory mechanisms in persistent forms of synaptic plasticity. Neuron. 2004; 44:59–73. PMID: 15450160.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Dopamine Receptor Supports the Potentiation of Intrinsic Excitability and Synaptic LTD in Temporoammonic-CA1 Synapse
- Long-term Synaptic Plasticity: Circuit Perturbation and Stabilization
- Downregulation of SIRT2 by Chronic Stress Reduces Expression of Synaptic Plasticity-related Genes through the Upregulation of Ehmt2
- Metabotropic glutamate receptor dependent long-term depression in the cortex
- Molecular Mechanisms Involved in Depotentiation and Their Relevance to Schizophrenia