Yonsei Med J.
2014 Nov;55(6):1656-1663. 10.3349/ymj.2014.55.6.1656.
DNA Hypomethylation-Mediated Overexpression of Carbonic Anhydrase 9 Induces an Aggressive Phenotype in Ovarian Cancer Cells
- Affiliations
-
- 1Department of Biochemistry, School of Medicine, Ewha Womans University, Seoul, Korea. ahnj@ewha.ac.kr
- 2Department of Obstetrics and Gynecology, School of Medicine, Ewha Womans University, Seoul, Korea. goodmorning@ewha.ac.kr
- KMID: 2070216
- DOI: http://doi.org/10.3349/ymj.2014.55.6.1656
Abstract
- PURPOSE
Both genetic and epigenetic alterations can lead to abnormal expression of metastasis-regulating genes in tumor cells. Recent studies suggest that aberrant epigenetic alterations, followed by differential gene expression, leads to an aggressive cancer cell phenotype. We examined epigenetically regulated genes that are involved in ovarian cancer metastasis.
MATERIALS AND METHODS
We developed SK-OV-3 human ovarian carcinoma cell xenografts in mice. We compared the mRNA expression and DNA methylation profiles of metastatic tissues to those of the original SK-OV-3 cell line.
RESULTS
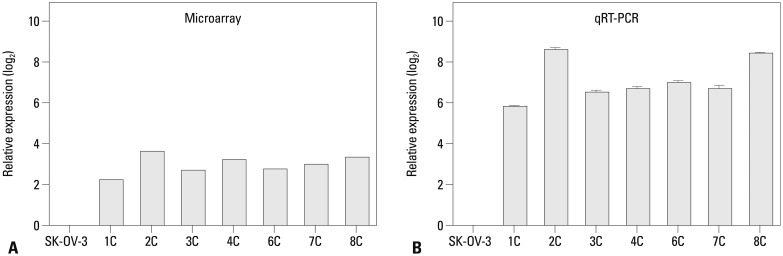
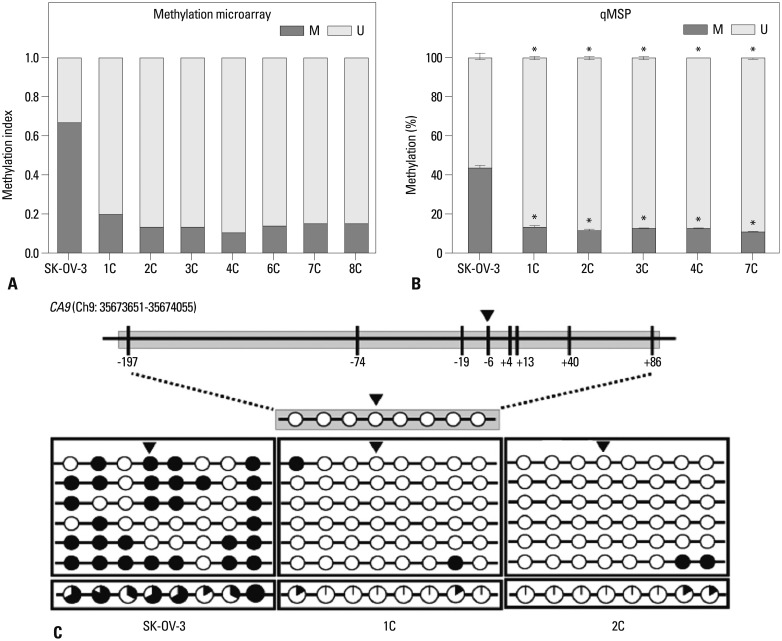
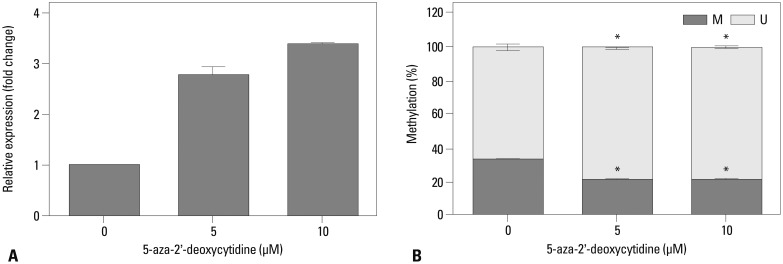
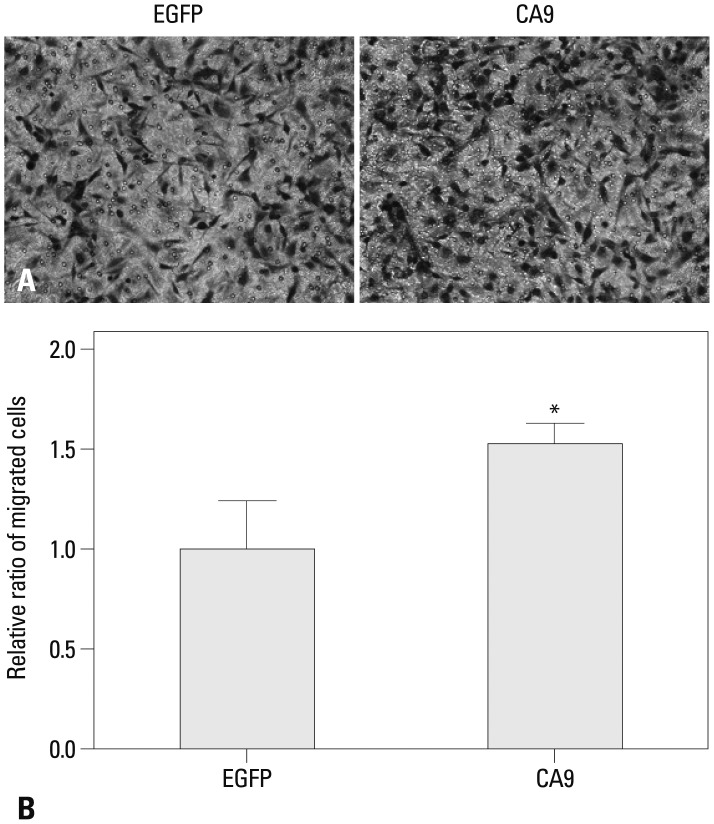
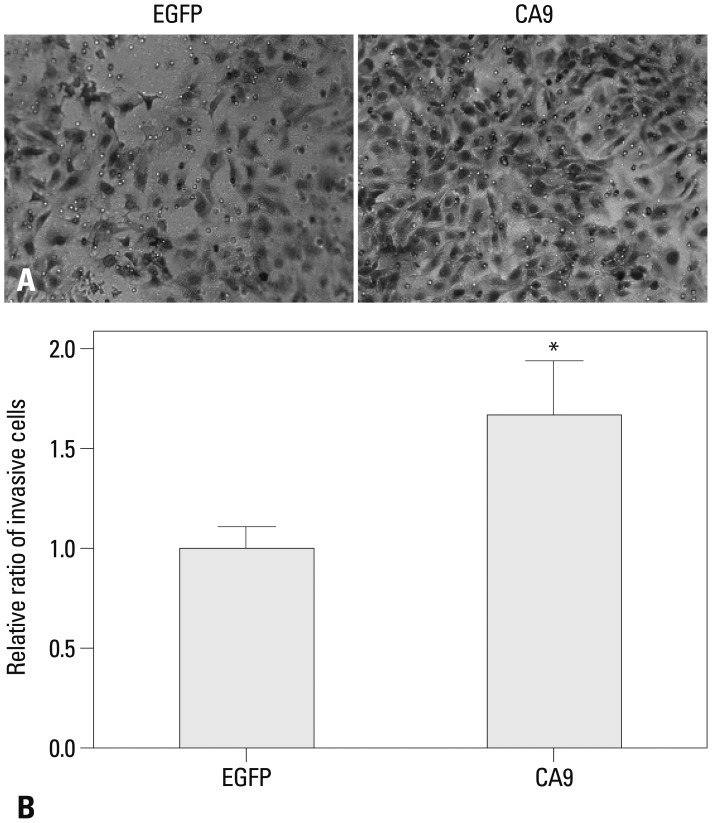
Metastatic implants showed increased mRNA expression of the carbonic anhydrase 9 (CA9) gene and hypomethylation at CpG sites in the CA9 promoter. Treatment of wild-type SK-OV-3 cells with the DNA methyltransferase inhibitor 5-aza-2'-deoxycytidine reduced methylation of the CA9 promoter and increased CA9 mRNA expression. Eight CpGs, which were located at positions -197, -74, -19, -6, +4, +13, +40, and +86, relative to the transcription start site, were hypomethylated in metastatic tumor implants, compared to that of wild-type SK-OV-3. Overexpression of CA9 induced an aggressive phenotype, including increased invasiveness and migration, in SK-OV-3 cells.
CONCLUSION
Alterations in the DNA methylation profile of the CA9 promoter were correlated with a more aggressive phenotype in ovarian cancer cells.
Keyword
MeSH Terms
-
Animals
Azacitidine/*analogs & derivatives/pharmacology
Carbonic Anhydrases/metabolism
*DNA Methylation
Female
Gene Expression Regulation, Neoplastic/*drug effects
Humans
Mice
Neoplasm Invasiveness/genetics
Neoplasm Metastasis/genetics/*pathology
Neoplasms, Experimental
Neoplasms, Glandular and Epithelial/genetics/*metabolism/pathology
Ovarian Neoplasms/genetics/*metabolism/pathology
Phenotype
Promoter Regions, Genetic
RNA, Messenger/metabolism
Azacitidine
Carbonic Anhydrases
RNA, Messenger
Figure
Cited by 2 articles
-
Aberrant Hypomethylation of Solute Carrier Family 6 Member 12 Promoter Induces Metastasis of Ovarian Cancer
Hye Youn Sung, San-Duk Yang, Ae Kyung Park, Woong Ju, Jung-Hyuck Ahn
Yonsei Med J. 2017;58(1):27-34. doi: 10.3349/ymj.2017.58.1.27.Ovarian Clear Cell Carcinoma Sub-Typing by ARID1A Expression
Jae Yoon Choi, Hyun Ho Han, Young Tae Kim, Joo Hyun Lee, Baek Gil Kim, Suki Kang, Nam Hoon Cho
Yonsei Med J. 2017;58(1):59-66. doi: 10.3349/ymj.2017.58.1.59.
Reference
-
1. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014; 64:9–29. PMID: 24399786.
Article2. Lengyel E. Ovarian cancer development and metastasis. Am J Pathol. 2010; 177:1053–1064. PMID: 20651229.
Article3. Kipps E, Tan DS, Kaye SB. Meeting the challenge of ascites in ovarian cancer: new avenues for therapy and research. Nat Rev Cancer. 2013; 13:273–282. PMID: 23426401.
Article4. Feki A, Berardi P, Bellingan G, Major A, Krause KH, Petignat P, et al. Dissemination of intraperitoneal ovarian cancer: Discussion of mechanisms and demonstration of lymphatic spreading in ovarian cancer model. Crit Rev Oncol Hematol. 2009; 72:1–9. PMID: 19179094.
Article5. Tan DS, Agarwal R, Kaye SB. Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol. 2006; 7:925–934. PMID: 17081918.
Article6. Jeon BH, Jang C, Han J, Kataru RP, Piao L, Jung K, et al. Profound but dysfunctional lymphangiogenesis via vascular endothelial growth factor ligands from CD11b+ macrophages in advanced ovarian cancer. Cancer Res. 2008; 68:1100–1109. PMID: 18281485.
Article7. Bussink J, Kaanders JH, van der Kogel AJ. Tumor hypoxia at the micro-regional level: clinical relevance and predictive value of exogenous and endogenous hypoxic cell markers. Radiother Oncol. 2003; 67:3–15. PMID: 12758235.
Article8. Ivanov S, Liao SY, Ivanova A, Danilkovitch-Miagkova A, Tarasova N, Weirich G, et al. Expression of hypoxia-inducible cell-surface transmembrane carbonic anhydrases in human cancer. Am J Pathol. 2001; 158:905–919. PMID: 11238039.
Article9. de Martino M, Klatte T, Seligson DB, LaRochelle J, Shuch B, Caliliw R, et al. CA9 gene: single nucleotide polymorphism predicts metastatic renal cell carcinoma prognosis. J Urol. 2009; 182:728–734. PMID: 19539328.
Article10. Bui MH, Seligson D, Han KR, Pantuck AJ, Dorey FJ, Huang Y, et al. Carbonic anhydrase IX is an independent predictor of survival in advanced renal clear cell carcinoma: implications for prognosis and therapy. Clin Cancer Res. 2003; 9:802–811. PMID: 12576453.11. Giatromanolaki A, Koukourakis MI, Sivridis E, Pastorek J, Wykoff CC, Gatter KC, et al. Expression of hypoxia-inducible carbonic anhydrase-9 relates to angiogenic pathways and independently to poor outcome in non-small cell lung cancer. Cancer Res. 2001; 61:7992–7998. PMID: 11691824.12. Liao SY, Ivanov S, Ivanova A, Ghosh S, Cote MA, Keefe K, et al. Expression of cell surface transmembrane carbonic anhydrase genes CA9 and CA12 in the human eye: overexpression of CA12 (CAXII) in glaucoma. J Med Genet. 2003; 40:257–261. PMID: 12676895.
Article13. Smyth GK. Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol. 2004; 3:Article3. PMID: 16646809.
Article14. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol. 1995; 57:289–300.
Article15. Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009; 4:44–57. PMID: 19131956.
Article16. Wilson WR, Hay MP. Targeting hypoxia in cancer therapy. Nat Rev Cancer. 2011; 11:393–410. PMID: 21606941.
Article17. Brahimi-Horn MC, Bellot G, Pouysségur J. Hypoxia and energetic tumour metabolism. Curr Opin Genet Dev. 2011; 21:67–72. PMID: 21074987.
Article18. Supuran CT. Carbonic anhydrases: novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov. 2008; 7:168–181. PMID: 18167490.
Article19. De Simone G, Supuran CT. Carbonic anhydrase IX: Biochemical and crystallographic characterization of a novel antitumor target. Biochim Biophys Acta. 2010; 1804:404–409. PMID: 19679200.
Article20. Hilvo M, Baranauskiene L, Salzano AM, Scaloni A, Matulis D, Innocenti A, et al. Biochemical characterization of CA IX, one of the most active carbonic anhydrase isozymes. J Biol Chem. 2008; 283:27799–27809. PMID: 18703501.
Article21. McDonald PC, Winum JY, Supuran CT, Dedhar S. Recent developments in targeting carbonic anhydrase IX for cancer therapeutics. Oncotarget. 2012; 3:84–97. PMID: 22289741.
Article22. Robertson N, Potter C, Harris AL. Role of carbonic anhydrase IX in human tumor cell growth, survival, and invasion. Cancer Res. 2004; 64:6160–6165. PMID: 15342400.
Article23. Williams E, Martin S, Moss R, Durrant L, Deen S. Co-expression of VEGF and CA9 in ovarian high-grade serous carcinoma and relationship to survival. Virchows Arch. 2012; 461:33–39. PMID: 22699808.
Article24. Svastová E, Zilka N, Zat'ovicová M, Gibadulinová A, Ciampor F, Pastorek J, et al. Carbonic anhydrase IX reduces E-cadherin-mediated adhesion of MDCK cells via interaction with beta-catenin. Exp Cell Res. 2003; 290:332–345. PMID: 14567991.25. Wykoff CC, Beasley NJ, Watson PH, Turner KJ, Pastorek J, Sibtain A, et al. Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Cancer Res. 2000; 60:7075–7083. PMID: 11156414.26. Cho M, Uemura H, Kim SC, Kawada Y, Yoshida K, Hirao Y, et al. Hypomethylation of the MN/CA9 promoter and upregulated MN/CA9 expression in human renal cell carcinoma. Br J Cancer. 2001; 85:563–567. PMID: 11506497.
Article27. Cho M, Grabmaier K, Kitahori Y, Hiasa Y, Nakagawa Y, Uemura H, et al. Activation of the MN/CA9 gene is associated with hypomethylation in human renal cell carcinoma cell lines. Mol Carcinog. 2000; 27:184–189. PMID: 10708480.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Enzyme Histochemical Study of Retinoblastoma
- Histochemical study on trematodes - Distribution of carbonic anhydrase activity
- The Effects of Topical Carbonic Anhydrase Inhibitors on Nitric Oxide Production in Trabecular Meshwork Cells
- Carbonic Anhyd rase Activity in Muller Cell
- Histochemical studies on Clonorchis sinensis [The distribution of carbonic anhydrase]