Restor Dent Endod.
2013 May;38(2):65-72. 10.5395/rde.2013.38.2.65.
Inhibition of Streptococcus mutans biofilm formation on composite resins containing ursolic acid
- Affiliations
-
- 1Department of Conservative Dentistry, Gangnam Severance Hospital, Yonsei University College of Dentistry, Seoul, Korea. pjw@yuhs.ac
- 2Department of Conservative Dentistry, Yonsei University College of Dentistry, Seoul, Korea.
- KMID: 1995461
- DOI: http://doi.org/10.5395/rde.2013.38.2.65
Abstract
OBJECTIVES
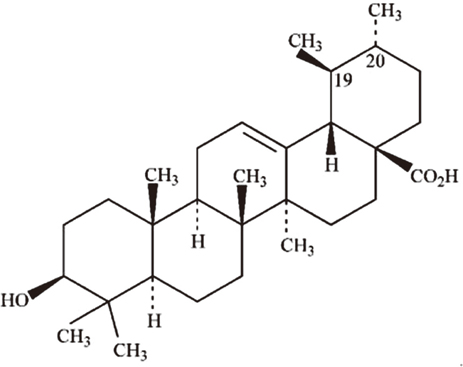
To evaluate the inhibitory effect of ursolic acid (UA)-containing composites on Streptococcus mutans (S. mutans) biofilm.
MATERIALS AND METHODS
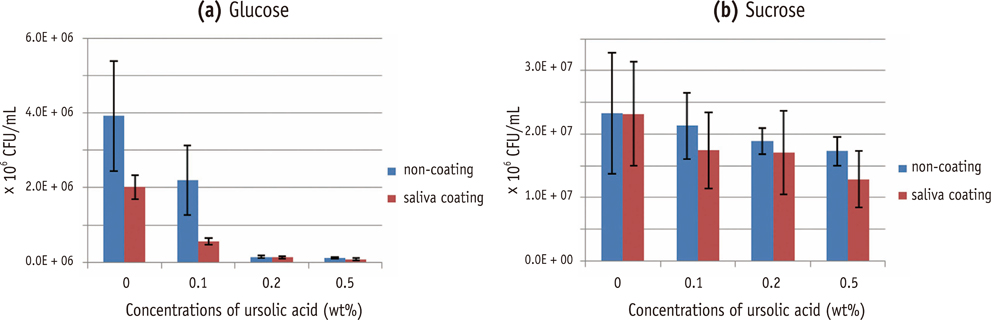
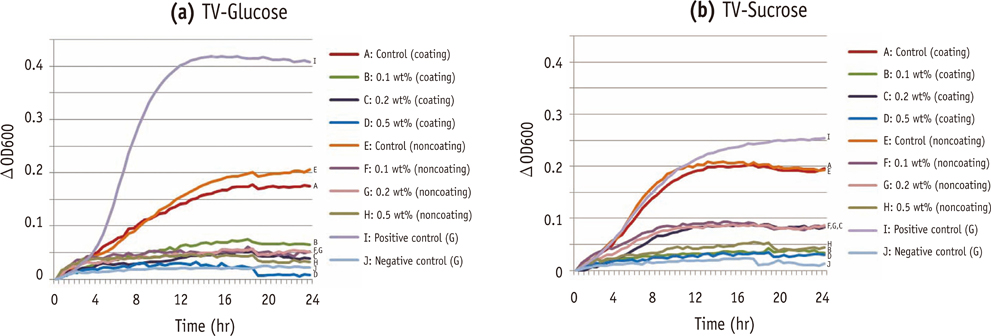
Composite resins with five different concentrations (0.04, 0.1, 0.2, 0.5, and 1.0 wt%) of UA (U6753, Sigma Aldrich) were prepared, and their flexural strengths were measured according to ISO 4049. To evaluate the effect of carbohydrate source on biofilm formation, either glucose or sucrose was used as a nutrient source, and to investigate the effect of saliva treatment, the specimen were treated with either unstimulated whole saliva or phosphate-buffered saline (PBS). For biofilm assay, composite disks were transferred to S. mutans suspension and incubated for 24 hr. Afterwards, the specimens were rinsed with PBS and sonicated. The colony forming units (CFU) of the disrupted biofilm cultures were enumerated. For growth inhibition test, the composites were placed on a polystyrene well cluster, and S. mutans suspension was inoculated. The optical density at 600 nm (OD600) was recorded by Infinite F200 pro apparatus (TECAN). One-way ANOVA and two-way ANOVA followed by Bonferroni correction were used for the data analyses.
RESULTS
The flexural strength values did not show significant difference at any concentration (p > 0.01). In biofilm assay, the CFU score decreased as the concentration of UA increased. The influence of saliva pretreatment was conflicting. The sucrose groups exhibited higher CFU score than glucose group (p < 0.05). In bacterial growth inhibition test, all experimental groups containing UA resulted in complete inhibition.
CONCLUSIONS
Within the limitations of the experiments, UA included in the composite showed inhibitory effect on S. mutans biofilm formation and growth.
MeSH Terms
Figure
Cited by 3 articles
-
Antibacterial properties of composite resins incorporating silver and zinc oxide nanoparticles on Streptococcus mutans and Lactobacillus
Shahin Kasraei, Lida Sami, Sareh Hendi, Mohammad-Yousef AliKhani, Loghman Rezaei-Soufi, Zahra Khamverdi
Restor Dent Endod. 2014;39(2):109-114. doi: 10.5395/rde.2014.39.2.109.Synergistic effect of xylitol and ursolic acid combination on oral biofilms
Yunyun Zou, Yoon Lee, Jinyoung Huh, Jeong-Won Park
Restor Dent Endod. 2014;39(4):288-295. doi: 10.5395/rde.2014.39.4.288.Antibacterial effect of self-etching adhesive systems on Streptococcus mutans
Seung-Ryong Kim, Dong-Hoon Shin
Restor Dent Endod. 2014;39(1):32-38. doi: 10.5395/rde.2014.39.1.32.
Reference
-
1. Haj-Ali R, Walker MP, Williams K. Survey of general dentists regarding posterior restorations, selection criteria, and associated clinical problems. Gen Dent. 2005; 53:369–375.2. Opdam NJ, Bronkhorst EM, Roeters JM, Loomans BA. A retrospective clinical study on longevity of posterior composite and amalgam restorations. Dent Mater. 2007; 23:2–8.
Article3. Beyth N, Yudovin-Farber I, Bahir R, Domb AJ, Weiss EI. Antibacterial activity of dental composites containing quaternary ammonium polyethylenimine nanoparticles against Streptococcus mutans. Biomaterials. 2006; 27:3995–4002.
Article4. Leung D, Spratt DA, Pratten J, Gulabivala K, Mordan NJ, Young AM. Chlorhexidine-releasing methacrylate dental composite materials. Biomaterials. 2005; 26:7145–7153.
Article5. Stobie N, Duffy B, McCormack DE, Colreavy J, Hidalgo M, McHale P, Hinder SJ. Prevention of Staphylococcus epidermidis biofilm formation using a low-temperature processed silver-doped phenyltriethoxysilane sol-gel coating. Biomaterials. 2008; 29:963–969.
Article6. Imazato S, Kinomoto Y, Tarumi H, Torii M, Russell RR, McCabe JF. Incorporation of antibacterial monomer MDPB into dentin primer. J Dent Res. 1997; 76:768–772.
Article7. Fan C, Chu L, Rawls HR, Norling BK, Cardenas HL, Whang K. Development of an antimicrobial resin-a pilot study. Dent Mater. 2011; 27:322–328.
Article8. Nohr RS, Macdonald JG. New biomaterials through surface segregation phenomenon: new quaternary ammonium compounds as antibacterial agents. J Biomater Sci Polym Ed. 1994; 5:607–619.
Article9. Liu J. Oleanolic acid and ursolic acid: research perspectives. J Ethnopharmacol. 2005; 100:92–94.
Article10. Liu J. Pharmacology of oleanolic acid and ursolic acid. J Ethnopharmacol. 1995; 49:57–68.
Article11. Fontanay S, Grare M, Mayer J, Finance C, Duval RE. Ursolic, oleanolic and betulinic acids: antibacterial spectra and selectivity indexes. J Ethnopharmacol. 2008; 120:272–276.
Article12. Takahashi N, Nyvad B. The role of bacteria in the caries process: ecological perspectives. J Dent Res. 2011; 90:294–303.13. Costerton JW, Lewandowski Z, DeBeer D, Caldwell D, Korber D, James G. Biofilms, the customized microniche. J Bacteriol. 1994; 176:2137–2142.
Article14. Shapiro S, Giertsen E, Guggenheim B. An in vitro oral biofilm model for comparing the efficacy of antimicrobial mouthrinses. Caries Res. 2002; 36:93–100.
Article15. Hardt M, Witkowska HE, Webb S, Thomas LR, Dixon SE, Hall SC, Fisher SJ. Assessing the effects of diurnal variation on the composition of human parotid saliva: quantitative analysis of native peptides using iTRAQ reagents. Anal Chem. 2005; 77:4947–4954.
Article16. Allaker RP, Douglas CW. Novel anti-microbial therapies for dental plaque-related diseases. Int J Antimicrob Agents. 2009; 33:8–13.
Article17. Jeon JG, Rosalen PL, Falsetta ML, Koo H. Natural products in caries research: current (limited) knowledge, challenges and future perspective. Caries Res. 2011; 45:243–263.
Article18. Kim MJ, Kim CS, Park JY, Lim YK, Park SN, Ahn SJ, Jin DC, Kim TH, Kook JK. Antimicrobial effects of ursolic acid against mutans streptococci isolated from Koreans. Int J Oral Biol. 2011; 36:7–11.19. Cowan MM. Plant products as antimicrobial agents. Clin Microbiol Rev. 1999; 12:564–582.
Article20. Kurek A, Grudniak AM, Szwed M, Klicka A, Samluk L, Wolska KI, Janiszowska W, Popowska M. Oleanolic acid and ursolic acid affect peptidoglycan metabolism in Listeria monocytogenes. Antonie Van Leeuwenhoek. 2010; 97:61–68.
Article21. Kohda H, Kozai K, Nagasaka N, Miyake Y, Suginaka H, Hidaka K, Yamasaki K. Prevention of dental caries by Oriental folk medicines-active principles of Zizyphi Fructus for inhibition of insoluble glucan formation by cariogenic bacterium Streptococcus mutans. Planta Med. 1986; (2):119–120.22. Ooshima T, Matsumura M, Hoshino T, Kawabata S, Sobue S, Fujiwara T. Contributions of three glycosyltransferases to sucrose-dependent adherence of Streptococcus mutans. J Dent Res. 2001; 80:1672–1677.
Article23. Gibbons RJ. Role of adhesion in microbial colonization of host tissues: a contribution of oral microbiology. J Dent Res. 1996; 75:866–870.
Article24. Cross SE, Kreth J, Zhu L, Sullivan R, Shi W, Qi F, Gimzewski JK. Nanomechanical properties of glucans and associated cell-surface adhesion of Streptococcus mutans probed by atomic force microscopy under in situ conditions. Microbiology. 2007; 153:3124–3132.
Article25. Schilling KM, Bowen WH. Glucans synthesized in situ in experimental salivary pellicle function as specific binding sites for Streptococcus mutans. Infect Immun. 1992; 60:284–295.
Article26. Ren D, Zuo R, González Barrios AF, Bedzyk LA, Eldridge GR, Pasmore ME, Wood TK. Differential gene expression for investigation of Escherichia coli biofilm inhibition by plant extract ursolic acid. Appl Environ Microbiol. 2005; 71:4022–4034.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory effect of 18β-glycyrrhetinic acid on the biofilm formation of Streptococcus mutans
- Synergistic effect of xylitol and ursolic acid combination on oral biofilms
- Inhibitory effect on Streptococcus mutans and mechanical properties of the chitosan containing composite resin
- Antimicrobial Effect of Polyphenon 60 against Streptococcus mutans and Streptococcus sobrinus
- Inhibitory effects of Coptis chinensis extract on the growth and biofilm formation of Streptococcus mutans and Streptococcus sobrinus