Korean J Obstet Gynecol.
2012 Jul;55(7):485-492. 10.5468/KJOG.2012.55.7.485.
Resistance of uterine radial artery blood flow is positively correlated with peripheral blood NK cell fraction in patients with unexplained recurrent spontaneous abortion
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Cheil General Hospital and Women's Healthcare Center, Kwandong University College of Medicine, Seoul, Korea. km1yang@naver.com
- KMID: 1992558
- DOI: http://doi.org/10.5468/KJOG.2012.55.7.485
Abstract
OBJECTIVE
To evaluate whether increased peripheral blood inflammatory immune cell can induce decrease of uterine blood flow in patients with unexplained recurrent spontaneous abortion (RSA).
METHODS
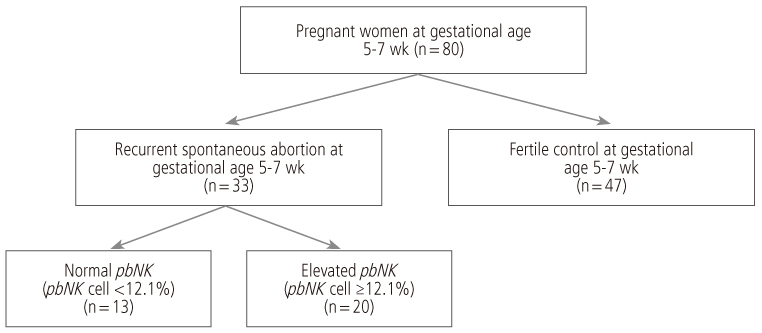
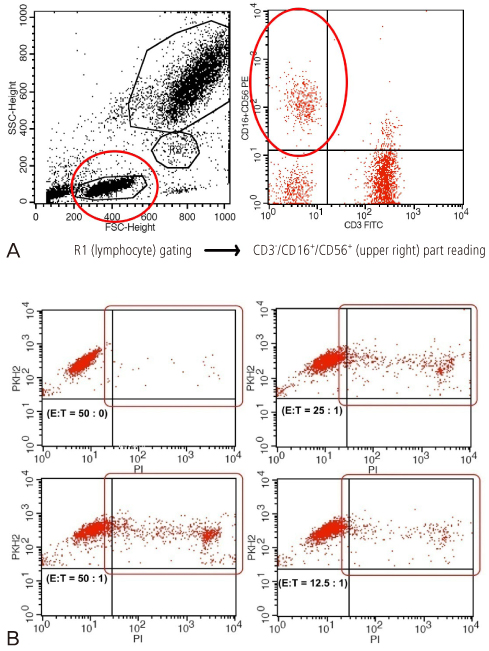
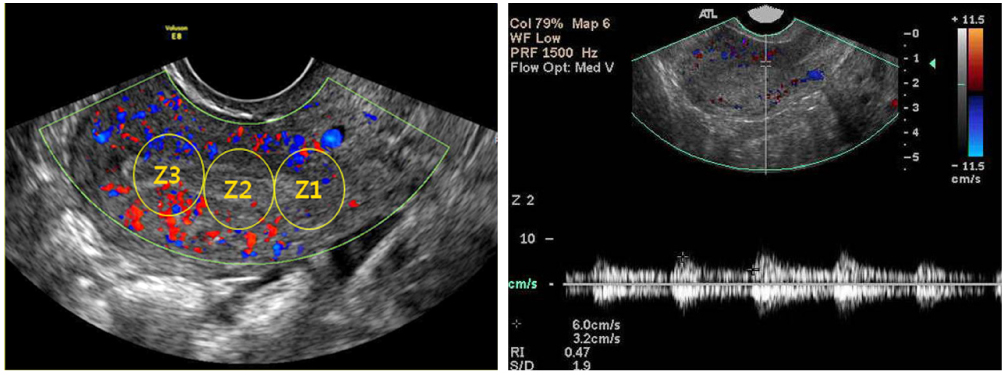
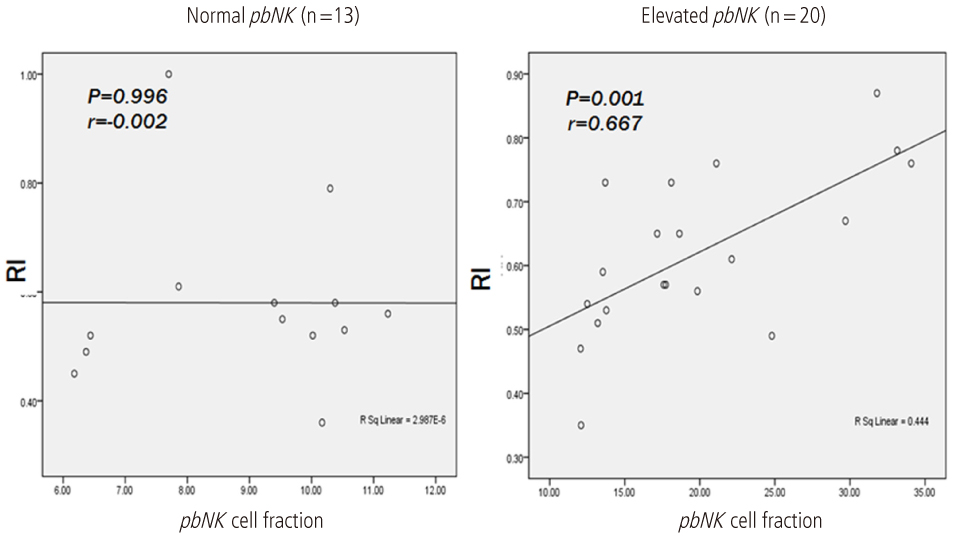
This study designed prospectively 33 pregnant women between 5 to 7 gestational weeks with a history of unexplained RSA included in this study. 47 normal pregnant women between 5 to 7 gestational weeks without history of infertility and/or RSA included as a control. Peripheral blood natural killer (pbNK) (CD3-/56+) fractions among peripheral blood monocyte (PBMC) were checked by flow cytometry. Uterine color-pulsed Doppler trans-vaginal ultrasound for evaluation of uterine radial artery RI was checked. Uterine radial artery resistance index (RI) compared between study and control group. After then, uterine radial artery RI was compared between high pbNK cell fraction above 12.1% among PBMC and normal pbNK cells fraction below 12.1%. Correlation between pbNK cell fraction to uterine radial artery RI was also evaluated.
RESULTS
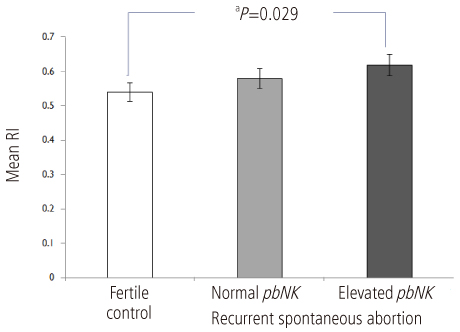
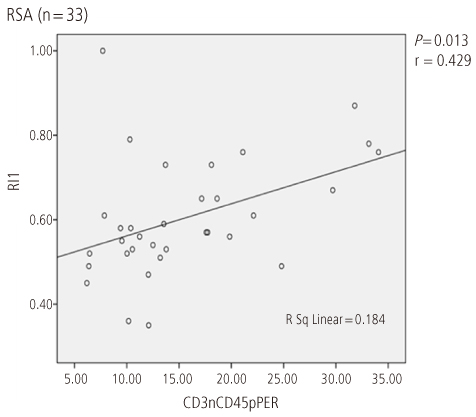
Uterine radial artery RI in early pregnancy was significantly higher in patients with RSA than that of normal control (0.60 +/- 0.14 vs. 0.54 +/- 0.12, P=0.039). Especially, the mean value of uterine radial artery RI in RSA patients with elevated pbNK cells was significantly increased than that of normal control (0.62 +/- 0.13 vs. 0.54 +/- 0.12, P=0.029). Otherwise, pbNK cell fractions among PBMC displayed strong positive correlation to uterine radial artery RI (Pearson's correlation coefficient P=0.001, r = 0.667).
CONCLUSION
Increased pbNK cells can evoke decreased uterine blood flow by their pro-inflammatory action on micro vascular structure such as uterine radial artery. This can be a one causative mechanism of inducing spontaneous abortion by increased NK cells. But, larger scaled study is needed for clarify our results.
MeSH Terms
Figure
Reference
-
1. Berek JS, Novak E. Berek & Novak's gynecology. 2007. 15th ed. Phiadelphia (PA): Linppincott williams & Wilkins.2. El-mashad AI, Mohamed MA, Farag MA, Ahmad MK, Ismail Y. Role of uterine artery Doppler velocimetry indices and plasma adrenomedullin level in women with unexplained recurrent pregnancy loss. J Obstet Gynaecol Res. 2011. 37:51–57.3. Ferreira AM, Pires CR, Moron AF, Araujo Júnior E, Traina E, Mattar R. Doppler assessment of uterine blood flow in recurrent pregnancy loss. Int J Gynaecol Obstet. 2007. 98:115–119.4. Beer AE, Kwak JY, Ruiz JE. Immunophenotypic profiles of peripheral blood lymphocytes in women with recurrent pregnancy losses and in infertile women with multiple failed in vitro fertilization cycles. Am J Reprod Immunol. 1996. 35:376–382.5. Perricone C, De Carolis C, Giacomelli R, Zaccari G, Cipriani P, Bizzi E, et al. High levels of NK cells in the peripheral blood of patients affected with anti-phospholipid syndrome and recurrent spontaneous abortion: a potential new hypothesis. Rheumatology (Oxford). 2007. 46:1574–1578.6. Choi JY, Hwang SJ, Han AR, Yoo JH, Park DW, Park CW, et al. Increased peripheral NK cell fraction and their cytolytic activity in patients with history of recurrent spontaneous abortion. Korean J Reprod Med. 2010. 37:115–124.7. Avril T, Jarousseau AC, Watier H, Boucraut J, Le Bouteiller P, Bardos P, et al. Trophoblast cell line resistance to NK lysis mainly involves an HLA class I-independent mechanism. J Immunol. 1999. 162:5902–5909.8. Kwak-Kim J, Park JC, Ahn HK, Kim JW, Gilman-Sachs A. Immunological modes of pregnancy loss. Am J Reprod Immunol. 2010. 63:611–623.9. Habara T, Nakatsuka M, Konishi H, Asagiri K, Noguchi S, Kudo T. Elevated blood flow resistance in uterine arteries of women with unexplained recurrent pregnancy loss. Hum Reprod. 2002. 17:190–194.10. Ghosh A, Ghosh M, Bhattacharya SM. Anti-phospholipid antibodies as a cause of recurrent pregnancy loss: a study in Calcutta, India. J Obstet Gynaecol. 2006. 26:407–410.11. Tamura H, Miwa I, Taniguchi K, Maekawa R, Asada H, Taketani T, et al. Different changes in resistance index between uterine artery and uterine radial artery during early pregnancy. Hum Reprod. 2008. 23:285–289.12. Keats TE, Sistrom C. Atlas de medidas radiológicas. 2002. Madrid, España: Elservier Science.13. Hui C, Lili M, Libin C, Rui Z, Fang G, Ling G, et al. Changes in coagulation and hemodynamics during pregnancy: a prospective longitudinal study of 58 cases. Arch Gynecol Obstet. 2012. 285:1231–1236.14. Steer CV, Tan SL, Mason BA, Campbell S. Midluteal-phase vaginal color Doppler assessment of uterine artery impedance in a subfertile population. Fertil Steril. 1994. 61:53–58.15. Goswamy RK, Williams G, Steptoe PC. Decreased uterine perfusion: a cause of infertility. Hum Reprod. 1988. 3:955–959.16. De Carolis C, Perricone C, Perricone R. NK cells, autoantibodies, and immunologic infertility: a complex interplay. Clin Rev Allergy Immunol. 2010. 39:166–175.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Elevated natural killer cell levels and autoimmunity synergistically decrease uterine blood flow during early pregnancy
- In vitro suppression of natural killer cell cytolytic activities by prednisolone in patients with history of unexplained recurrent spontaneous abortion and elevated peripheral blood NK cell fraction
- Changes of natural killer cell number and cytolytic activity during first trimester of pregnancy in recurrent spontaneous abortion patients and fertile control
- The Preconceptional Level of Peripheral Natural Killer Cells which was Expected to Bring Successful Treatment Outcome using Low-dose Intravenous Gamma Immunoglobulin (IVIg) Infusion in Patients with Recurrent Spontaneous Abortion
- A Change of Peripheral CD56+Natural Killer Cell Fraction in Recurrent Pregnancy Loss Patients who Successfully Treated with Intravenous Immunoglobulin in Previous Pregnancy