Anat Cell Biol.
2015 Jun;48(2):85-94. 10.5115/acb.2015.48.2.85.
Improved adipogenic in vitro differentiation: comparison of different adipogenic cell culture media on human fat and bone stroma cells for fat tissue engineering
- Affiliations
-
- 1Department of Oral and Maxillofacial Surgery, Christian-Albrechts-University, Kiel, Germany. amiralexanderghoniem@gmail.com
- KMID: 1845276
- DOI: http://doi.org/10.5115/acb.2015.48.2.85
Abstract
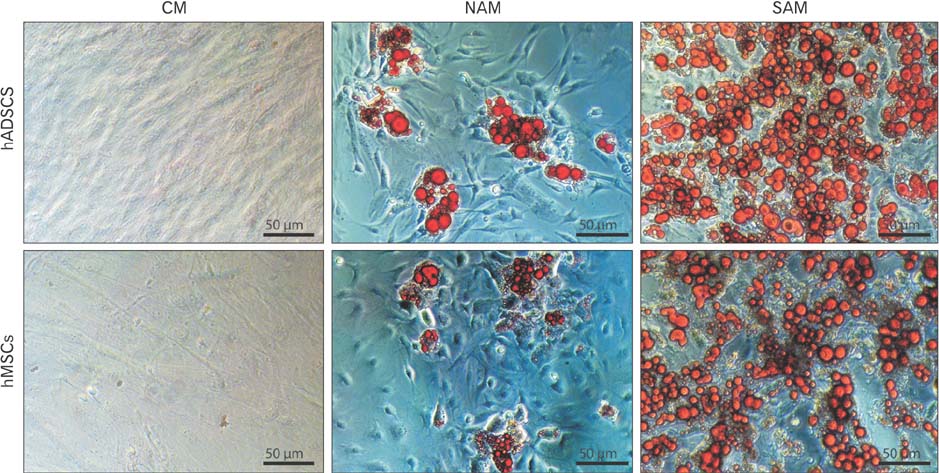
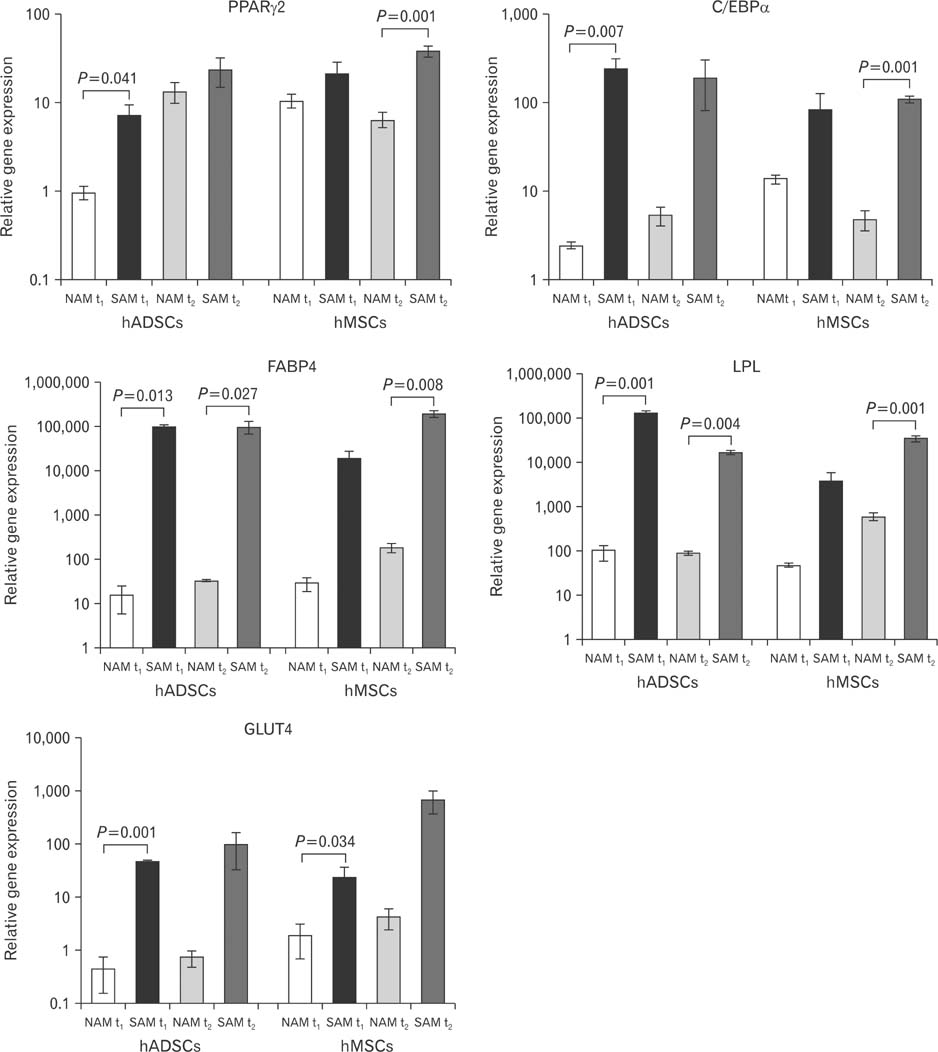
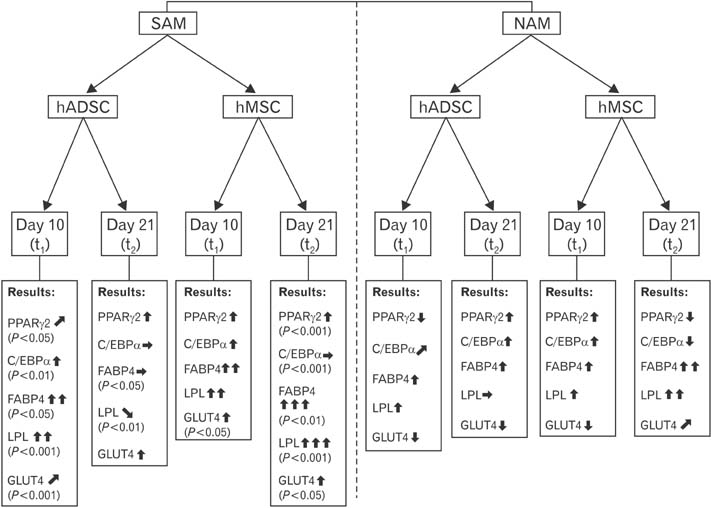
- To date there is no sufficient in vitro fat tissue engineering and a protocol has not been well established for this purpose. Therefore, we evaluated the in vitro influence of two different adipogenic growth media for their stimulation potential on different cell lineages to clearly define the most potent adipogenic growth media for future in vitro tissue engineering approaches. The samples for differentiation were composed of human adipogenic-derived stroma cells (hADSCs) and human bone marrow mesenchymal stroma cells (hMSCs). A normal adipogenic medium (NAM) and a specific adipogenic medium (SAM) were tested for their adipogenic stimulation potential. After 10 days and 21 days the relative gene expression was measured for the adipogenic marker genes PPARgamma2, C/EBPalpha, FABP4, LPL, and GLUT4 detected through real time reverse transcriptase polymease chain reaction (RT-PCR). Other study variables were the comparison between NAM and SAM and between the used cells hADSCs and hMSCs. Additionally an Oil-Red staining was performed after 21 days. Our results revealed that only SAM was significantly (P<0.05) superior in the differentiation process in contrast to NAM for 10 days and 21 days. As well was SAM superior to differentiate the used cell lineages. This was evaluated by the detected marker genes PPARgamma2, C/EBPalpha, FABP4, LPL, and GLUT4 through real time RT-PCR and by Oil-Red staining. In addition, the hMSCs proofed to be equal donor cells for adipogenic differentiation especially when stimulated by SAM. The results suggest that the SAM should be established as a new standard medium for a more promising in vitro adipogenic differentiation.
Keyword
MeSH Terms
Figure
Reference
-
1. Patrick CW Jr. Engineering adipose tissue for regenerative and reparative therapies. Semin Plast Surg. 2005; 19:207–215.2. Matsumoto D, Sato K, Gonda K, Takaki Y, Shigeura T, Sato T, Aiba-Kojima E, Iizuka F, Inoue K, Suga H, Yoshimura K. Cell-assisted lipotransfer: supportive use of human adipose-derived cells for soft tissue augmentation with lipoinjection. Tissue Eng. 2006; 12:3375–3382.3. Aimaiti A, Saiwulaiti Y, Saiyiti M, Wang YH, Cui L, Yusufu A. Therapeutic effect of osteogenically induced adipose derived stem cells on vascular deprivation-induced osteonecrosis of the femoral head in rabbits. Chin J Traumatol. 2011; 14:215–220.4. Pak J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: a case series. J Med Case Rep. 2011; 5:296.5. Yoshimura K, Sato K, Aoi N, Kurita M, Inoue K, Suga H, Eto H, Kato H, Hirohi T, Harii K. Cell-assisted lipotransfer for facial lipoatrophy: efficacy of clinical use of adipose-derived stem cells. Dermatol Surg. 2008; 34:1178–1185.6. Yuksel E, Choo J, Wettergreen M, Liebschner M. Challenges in soft tissue engineering. Semin Plast Surg. 2005; 19:261–270.7. Yoshimura H, Muneta T, Nimura A, Yokoyama A, Koga H, Sekiya I. Comparison of rat mesenchymal stem cells derived from bone marrow, synovium, periosteum, adipose tissue, and muscle. Cell Tissue Res. 2007; 327:449–462.8. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006; 8:315–317.9. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001; 7:211–228.10. Torii I, Morikawa S, Nakano A, Morikawa K. Establishment of a human preadipose cell line, HPB-AML-I: refractory to PPARgamma-mediated adipogenic stimulation. J Cell Physiol. 2003; 197:42–52.11. Sekiya I, Larson BL, Vuoristo JT, Cui JG, Prockop DJ. Adipogenic differentiation of human adult stem cells from bone marrow stroma (MSCs). J Bone Miner Res. 2004; 19:256–264.12. Mauney JR, Volloch V, Kaplan DL. Matrix-mediated retention of adipogenic differentiation potential by human adult bone marrow-derived mesenchymal stem cells during ex vivo expansion. Biomaterials. 2005; 26:6167–6175.13. Skurk T, Ecklebe S, Hauner H. A novel technique to propagate primary human preadipocytes without loss of differentiation capacity. Obesity (Silver Spring). 2007; 15:2925–2931.14. Lee MJ, Wu Y, Fried SK. A modified protocol to maximize differentiation of human preadipocytes and improve metabolic phenotypes. Obesity (Silver Spring). 2012; 20:2334–2340.15. Lequeux C, Auxenfans C, Mojallal A, Sergent M, Damour O. Optimization of a culture medium for the differentiation of preadipocytes into adipocytes in a monolayer. Biomed Mater Eng. 2009; 19:283–291.16. Gerhold DL, Liu F, Jiang G, Li Z, Xu J, Lu M, Sachs JR, Bagchi A, Fridman A, Holder DJ, Doebber TW, Berger J, Elbrecht A, Moller DE, Zhang BB. Gene expression profile of adipocyte differentiation and its regulation by peroxisome proliferator-activated receptor-gamma agonists. Endocrinology. 2002; 143:2106–2118.17. Hanks CT. Insulin and hydrocortisone influences on cultured rat tongue epithelium. Arch Oral Biol. 1979; 24:765–776.18. Garofalo RS, Orena SJ, Rafidi K, Torchia AJ, Stock JL, Hildebrandt AL, Coskran T, Black SC, Brees DJ, Wicks JR, McNeish JD, Coleman KG. Severe diabetes, age-dependent loss of adipose tissue, and mild growth deficiency in mice lacking Akt2/PKB beta. J Clin Invest. 2003; 112:197–208.19. Kim JE, Chen J. Regulation of peroxisome proliferator-activated receptor-gamma activity by mammalian target of rapamycin and amino acids in adipogenesis. Diabetes. 2004; 53:2748–2756.20. Ortega FJ, Moreno-Navarrete JM, Ribas V, Esteve E, Rodriguez-Hermosa JI, Ruiz B, Peral B, Ricart W, Zorzano A, Fernandez-Real JM. Subcutaneous fat shows higher thyroid hormone receptor-alpha1 gene expression than omental fat. Obesity (Silver Spring). 2009; 17:2134–2141.21. Lefterova MI, Lazar MA. New developments in adipogenesis. Trends Endocrinol Metab. 2009; 20:107–114.22. Hasegawa N, Kawaguchi H, Hirachi A, Takeda K, Mizuno N, Nishimura M, Koike C, Tsuji K, Iba H, Kato Y, Kurihara H. Behavior of transplanted bone marrow-derived mesenchymal stem cells in periodontal defects. J Periodontol. 2006; 77:1003–1007.23. Caprio M, Fève B, Claës A, Viengchareun S, Lombès M, Zennaro MC. Pivotal role of the mineralocorticoid receptor in corticosteroid-induced adipogenesis. FASEB J. 2007; 21:2185–2194.24. Tang QQ, Gronborg M, Huang H, Kim JW, Otto TC, Pandey A, Lane MD. Sequential phosphorylation of CCAAT enhancer-binding protein beta by MAPK and glycogen synthase kinase 3beta is required for adipogenesis. Proc Natl Acad Sci U S A. 2005; 102:9766–9771.25. Wang GL, Shi X, Salisbury E, Sun Y, Albrecht JH, Smith RG, Timchenko NA. Cyclin D3 maintains growth-inhibitory activity of C/EBPalpha by stabilizing C/EBPalpha-cdk2 and C/EBPalpha-Brm complexes. Mol Cell Biol. 2006; 26:2570–2582.26. Rosen ED, MacDougald OA. Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol. 2006; 7:885–896.27. Bernlohr DA, Coe NR, LiCata VJ. Fatty acid trafficking in the adipocyte. Semin Cell Dev Biol. 1999; 10:43–49.28. Maeda K, Cao H, Kono K, Gorgun CZ, Furuhashi M, Uysal KT, Cao Q, Atsumi G, Malone H, Krishnan B, Minokoshi Y, Kahn BB, Parker RA, Hotamisligil GS. Adipocyte/macrophage fatty acid binding proteins control integrated metabolic responses in obesity and diabetes. Cell Metab. 2005; 1:107–119.29. Trigatti BL, Gerber GE. A direct role for serum albumin in the cellular uptake of long-chain fatty acids. Biochem J. 1995; 308(Pt 1):155–159.30. Berenguer M, Le Marchand-Brustel Y, Govers R. GLUT4 molecules are recruited at random for insertion within the plasma membrane upon insulin stimulation. FEBS Lett. 2010; 584:537–542.31. Martinez L, Berenguer M, Bruce MC, Le Marchand-Brustel Y, Govers R. Rosiglitazone increases cell surface GLUT4 levels in 3T3-L1 adipocytes through an enhancement of endosomal recycling. Biochem Pharmacol. 2010; 79:1300–1309.32. Dicker A, Le Blanc K, Aström G, van Harmelen V, Götherström C, Blomqvist L, Arner P, Rydén M. Functional studies of mesenchymal stem cells derived from adult human adipose tissue. Exp Cell Res. 2005; 308:283–290.33. Sobh MA. Adipogenesis of Sprague Dawely rats mesenchymal stem cells: a morphological, immunophenotyping and gene expression follow-up study. Anat Cell Biol. 2014; 47:83–90.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Dipogenic and Osteogenic Potentials of Visceral and Subcutaneous Adipose Stem Cells of Human
- Plasticity of Cloned Human Marrow Adipocytes between Osteogenesis and Adipogenesis
- Differential Potential of Stem Cells Following Their Origin: Subacromial Bursa, Bone Marrow, Umbilical Cord Blood
- Effects of nanoscale ridge/groovepattern arrayed surface on in vitro differentiation of multi-potent pulp cells derived from human supernumerary teeth
- Optimization of adipogenic differentiation conditions for canine adipose-derived stem cells