Lab Anim Res.
2015 Mar;31(1):13-23. 10.5625/lar.2015.31.1.13.
Beneficial effects of ethanol extracts of Red Liriope platyphylla on vascular dysfunction in the aorta of spontaneously hypertensive rats
- Affiliations
-
- 1Department of Biomaterials Science, College of Natural Resources & Life Science/Life and Industry Convergence Institute, Pusan National University, Miryang, Korea. dyhwang@pusan.ac.kr
- 2Department of Life Science and Environmental Biochemistry, College of Natural Resources & Life Science, Pusan National University, Miryang, Korea.
- 3Department of Experimental Animal Research, Biomedical Research Institute, Seoul National University Hospital, Seoul, Korea.
- KMID: 1787647
- DOI: http://doi.org/10.5625/lar.2015.31.1.13
Abstract
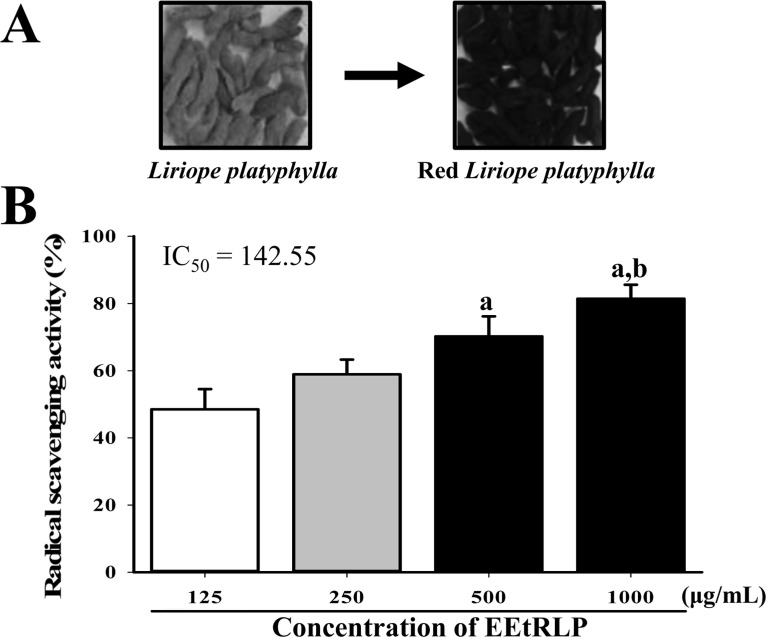
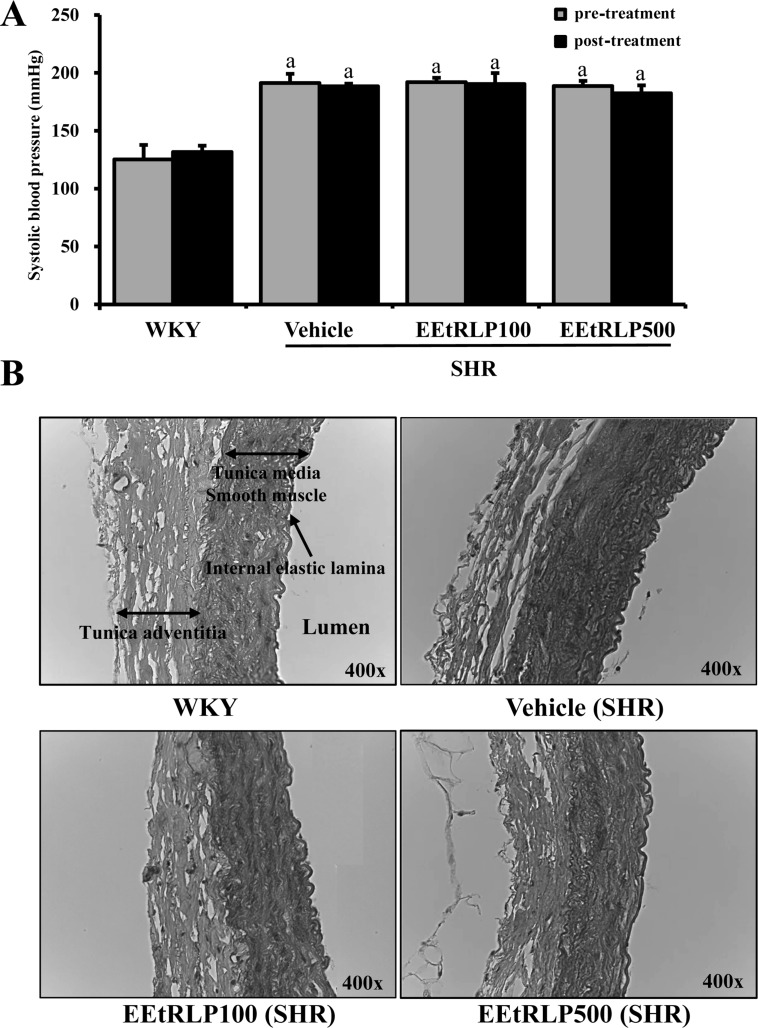
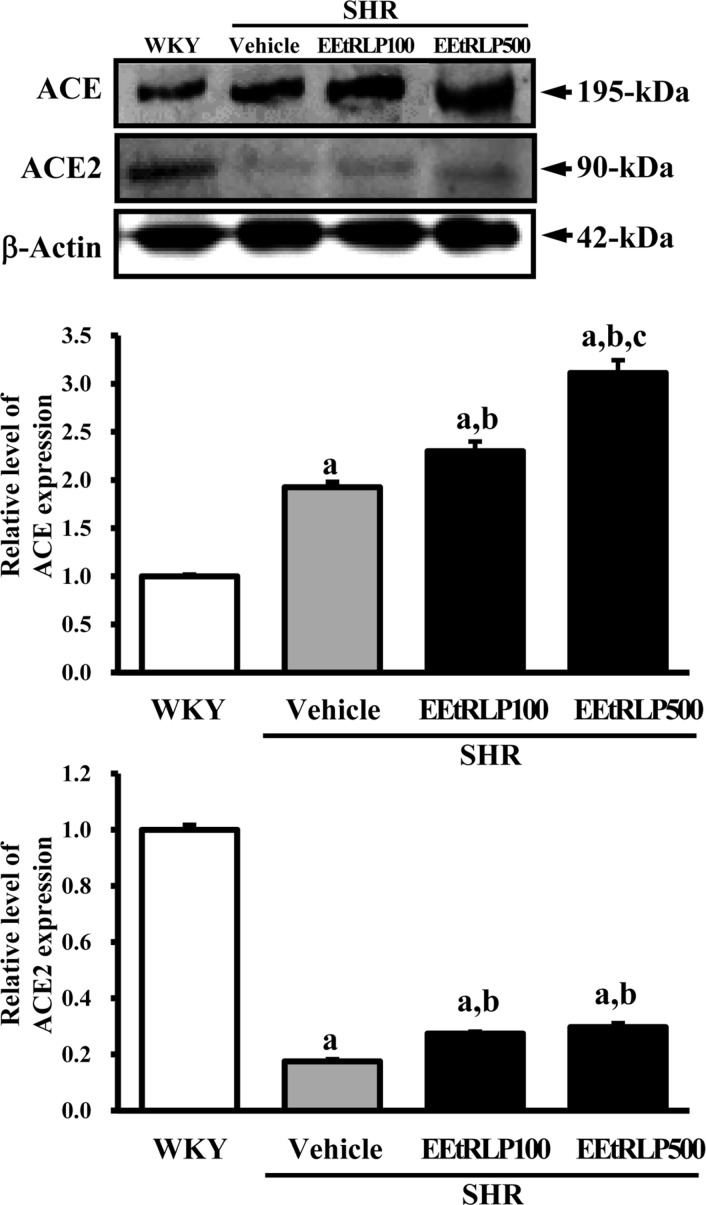
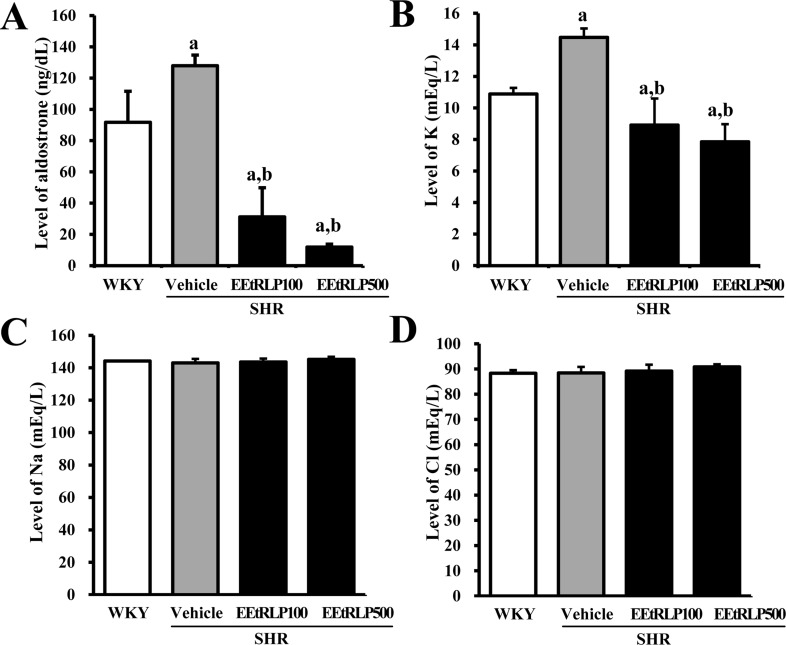
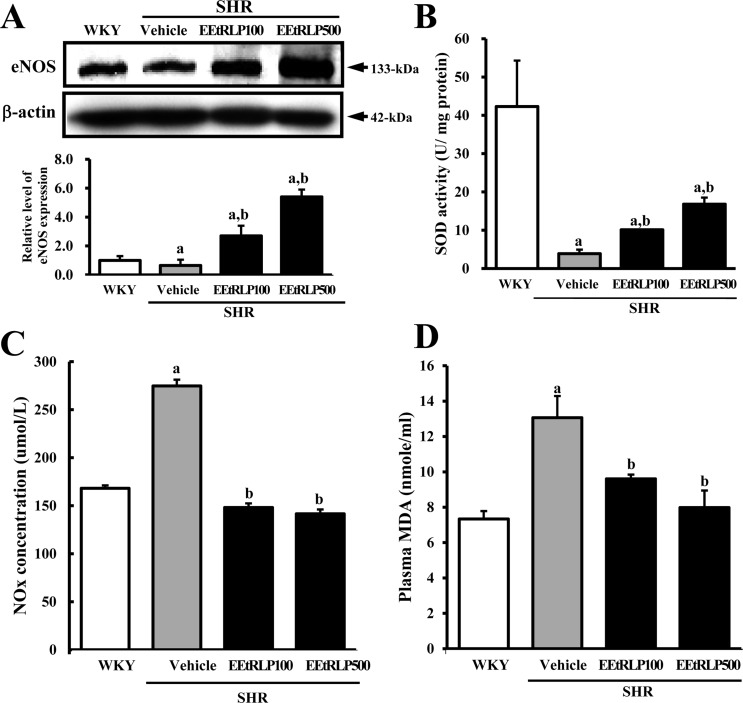
- Some biological effects of Red Liriope platyphylla (RLP) on various chronic diseases including Alzheimer's disease, diabetes and obesity were suggested after a report of the production from Liriope platyphylla (L. platyphylla, LP) roots using a steaming process. To examine the beneficial effects of ethanol extracts RLP (EEtRLP) on the vascular dysfunction of hypertension, alterations in key factors related to vascular regulation and antioxidant conditions were investigated in spontaneously hypertensive rats (SHR) after EEtRLP treatment for 2 weeks. High levels of 2,2-diphenyl-1-picrylhydrazyl (DPPH) scavenging activity were detected in 500 or 1,000 mg/mL EEtRLP. Although no significant improvement of systolic blood pressure or aortic wall thickness were observed in the EEtRLP treated group, the expression level of angiotensin converting enzyme (ACE) and ACE2 increased significantly after EEtRLP treatment. Moreover, the concentration of aldosterone and K ion in serum rapidly recovered in the EEtRLP treated group relative to the vehicle treated group. Furthermore, the endothelial nitric oxide synthase (eNOS) expression and superoxide dismutase (SOD) activity were significantly increased in the EEtRLP treated group relative to the vehicle treated group, while the level of malondialdehyde (MDA) and NOx in the serum of the same group were recovered to the level of Wistar Kyoto (WKY) rats. Overall, the results presented herein provide novel evidence that EEtRLP treatment may improve vascular dysfunction in the aorta of the SHR through up regulation of the antioxidant state and down regulation of aldosterone and K ion concentration. These results also suggest that EEtRLP may be a potential candidate for treatment of various chronic diseases showing vascular dysfunction.
Keyword
MeSH Terms
-
Aldosterone
Alzheimer Disease
Animals
Aorta*
Blood Pressure
Chronic Disease
Down-Regulation
Ethanol*
Hypertension
Malondialdehyde
Nitric Oxide Synthase Type III
Obesity
Peptidyl-Dipeptidase A
Rats
Rats, Inbred SHR*
Steam
Superoxide Dismutase
Up-Regulation
Aldosterone
Ethanol
Malondialdehyde
Nitric Oxide Synthase Type III
Peptidyl-Dipeptidase A
Steam
Superoxide Dismutase
Figure
Reference
-
1. Carretero OA, Oparil S. Essential hypertension. Part I: definition and etiology. Circulation. 2000; 101(3):329–335. PMID: 10645931.2. Fisher DL, Williams GH. Hypertensive vascular disease. Harrisons principles of internal medicine. 16th Edition. 2005. (2):p. 1463–1480. Chapter 230.3. Lüscher TF, Aarhus LL, Vanhoutte PM. Indomethacin improves the impaired endothelium-dependent relaxations in small mesenteric arteries of the spontaneously hypertensive rat. Am J Hypertens. 1990; 3(1):55–58. PMID: 2302329.
Article4. Taddei S, Virdis A, Ghiadoni L, Sudano I, Salvetti A. Endothelial dysfunction in hypertension. J Cardiovasc Pharmacol. 2001; 38(Suppl 2):S11–S14. PMID: 11811368.
Article5. Taddei S, Ghiadoni L, Virdis A, Versari D, Salvetti A. Mechanisms of endothelial dysfunction: clinical significance and preventive non-pharmacological therapeutic strategies. Curr Pharm Des. 2003; 9(29):2385–2402. PMID: 14529554.
Article6. Mihailovic-Stanojevic N, Belšèak-Cvitanoviæ A, Grujiæ-Milanoviæ J, Ivanov M, Jovoviæ Dj, Bugarski D, Miloradoviæ Z. Antioxidant and antihypertensive activity of extract from Thymus serpyllum L. in experimental hypertension. Plant Foods Hum Nutr. 2013; 68(3):235–240. PMID: 23828496.7. Zhang JX, Yang JR, Chen GX, Tang LJ, Li WX, Yang H, Kong X. Sesamin ameliorates arterial dysfunction in spontaneously hypertensive rats via downregulation of NADPH oxidase subunits and upregulation of eNOS expression. Acta Pharmacol Sin. 2013; 34(7):912–920. PMID: 23624755.
Article8. Zhou F, Xue Z, Wang J. Antihypertensive effects of silk fibroin hydrolysate by alcalase and purification of an ACE inhibitory dipeptide. J Agric Food Chem. 2010; 58(11):6735–6740. PMID: 20481470.
Article9. Yuan W, Wang J, Zhou F. In vivo hypotensive and physiological effects of a silk fibroin hydrolysate on spontaneously hypertensive rats. Biosci Biotechnol Biochem. 2012; 76(10):1987–1989. PMID: 23047112.10. Hashimoto R, Umemoto S, Guo F, Umeji K, Itoh S, Kishi H, Kobayashi S, Matsuzaki M. Nifedipine activates PPARgamma and exerts antioxidative action through Cu/ZnSOD independent of blood-pressure lowering in SHRSP. J Atheroscler Thromb. 2010; 17(8):785–795. PMID: 20460829.11. Choi SI, Kim JE, Hwang IS, Lee HR, Lee YJ, Kwak MH, Son HJ, Lee HS, Lee JS, Kang BC, Hwang DY. Toxicity of red Liriope platyphylla manufactured by steaming process on liver and kidney organs of ICR mice. Lab Anim Res. 2012; 28(4):229–238. PMID: 23326283.12. Lee HR, Kim JE, Lee YJ, Kwak MH, Im DS, Hwang DY. Red Liriope platyphylla stimulated the insulin secretion through the regulation of calcium concentration in rat insulinoma cells and animal models. Lab Anim Res. 2013; 29(2):84–95. PMID: 23825481.13. Lee HR, Kim JE, Goo JS, Choi SI, Hwang IS, Lee YJ, Son HJ, Lee HS, Lee JS, Hwang DY. Red Liriope platyphylla contains a large amount of polyphenolic compounds which stimulate insulin secretion and suppress fatty liver formation through the regulation of fatty acid oxidation in OLETF rats. Int J Mol Med. 2012; 30(4):905–913. PMID: 22842959.14. Choi SI, Goo JS, Kim JE, Nam SH, Hwang IS, Lee HR, Lee YJ, Son HJ, Lee HS, Lee JS, Kim HJ, Hwang DY. Differential effects of the steaming time and frequency for manufactured red Liriope platyphylla on nerve growth factor secretion ability, nerve growth factor receptor signaling pathway and regulation of calcium concentration. Mol Med Rep. 2012; 6(5):1160–1170. PMID: 22895564.15. Choi SI, Goo JS, Kim JE, Hwang IS, Lee HR, Lee YJ, Son HJ, Lee HS, Lee JS, Hwang DY. Effects of Red Liriope platyphylla on NGF secretion ability, NGF receptor signaling pathway and γ-secretase components in NSE/hAPPsw transgenic mice expressing Alzheimer's Disease. Lab Anim Res. 2012; 28(3):155–163. PMID: 23091515.16. Kwak MH, Kim JE, Hwang IS, Lee YJ, An BS, Hong JT, Lee SH, Hwang DY. Quantitative evaluation of therapeutic effect of Liriope platyphylla on phthalic anhydride-induced atopic dermatitis in IL-4/Luc/CNS-1 Tg mice. J Ethnopharmacol. 2013; 148(3):880–889. PMID: 23726789.17. Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic. 1965; 16(3):144–158.18. Zhishen J, Mengcheng T, Jianming W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999; 64:555–559.
Article19. Oh H, Ko EK, Kim DH, Jang KK, Park SE, Lee HS, Kim YC. Secoiridoid glucosides with free radical scavenging activity from the leaves of Syringa dilatata. Phytother Res. 2003; 17(4):417–419. PMID: 12722154.20. Oh KS, Han W, Wang MH, Lee BH. The effects of chronic treatment with Morus bombycis KOIDZUMI in spontaneously hypertensive rats. Biol Pharm Bull. 2007; 30(7):1278–1283. PMID: 17603167.21. Rao MS. Inhibition of the renin angiotensin aldosterone system: focus on aliskiren. J Assoc Physicians India. 2010; 58:102–108. PMID: 20653151.22. Jang HD, Chang KS, Huang YS, Hsu CL, Lee SH, Su MS. Principal phenolic phytochemicals and antioxidant activities of three Chinese medicinal plants. Food Chem. 2007; 103(3):749–756.
Article23. Kassim NK, Rahmani M, Ismail A, Sukari MA, Ee GC, Nasir NM, Awang K. Antioxidant activity-guided separation of coumarins and lignan from Melicope glabra (Rutaceae). Food Chem. 2013; 139(1-4):87–92. PMID: 23561082.24. Zhao W, Yu J, Su Q, Liang J, Zhao L, Zhang Y, Sun W. Antihypertensive effects of extract from Picrasma quassiodes (D. Don) Benn. in spontaneously hypertensive rats. J Ethnopharmacol. 2013; 145(1):187–192. PMID: 23142488.
Article25. Zhao F, Gao Z, Jiao W, Chen L, Chen L, Yao X. In vitro anti-inflammatory effects of beta-carboline alkaloids, isolated from Picrasma quassioides, through inhibition of the iNOS pathway. Planta Med. 2012; 78(18):1906–1911. PMID: 23115019.26. Jiao WH, Gao H, Zhao F, He F, Zhou GX, Yao XS. A new neolignan and a new sesterterpenoid from the stems of Picrasma quassioides Bennet. Chem Biodivers. 2011; 8(6):1163–1169. PMID: 21674788.27. Zhai H, Yang Y, Ma Y, Li X, Chen B, Xiang Y, Liu F, Yu Z, Guo J. Role of oxidized low-density lipoprotein in NF-κB signal pathway. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2013; 29(12):1254–1257. PMID: 24321067.28. Touyz RM, Schiffrin EL. Reactive oxygen species in vascular biology: implications in hypertension. Histochem Cell Biol. 2004; 122(4):339–352. PMID: 15338229.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antihypertensive effect of ethanol extracts of Aralia elata in spontaneously hypertensive rats
- Precautionary effects of Red Liriope platyphylla on NGF secretion and Abeta42 deposition under the preclinical stage of Alzheimer's disease in Tg2576 mice
- Effect of ethanol extracts from red pepper seeds on antioxidative defense system and oxidative stress in rats fed high-fat, high-cholesterol diet
- Laxative effects of Liriope platyphylla are tightly correlated with suppression of endoplasmic reticulum stress in loperamide-induced constipation of SD rats
- Effect of Thiopental and Thiamylal on Isolated Thoracic Aorta in Normotensive and Spontaneously Hypertensive Rats