J Vet Sci.
2014 Jun;15(2):179-185. 10.4142/jvs.2014.15.2.179.
Effects of resveratrol on the insulin signaling pathway of obese mice
- Affiliations
-
- 1Department of Toxicology & Biochemistry, College of Veterinary Medicine, Jeju National University, Jeju 690-756, Korea. chhan@jejunu.ac.kr
- 2Department of Internal Medicine, School of Medicine, Institute of Medical Science, Jeju National University, Jeju 690-756, Korea.
- KMID: 1784637
- DOI: http://doi.org/10.4142/jvs.2014.15.2.179
Abstract
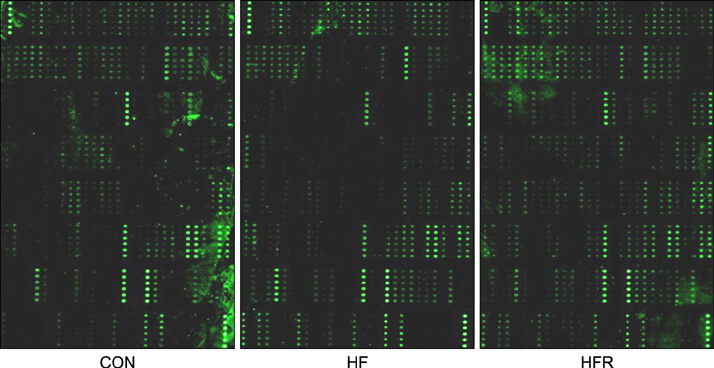
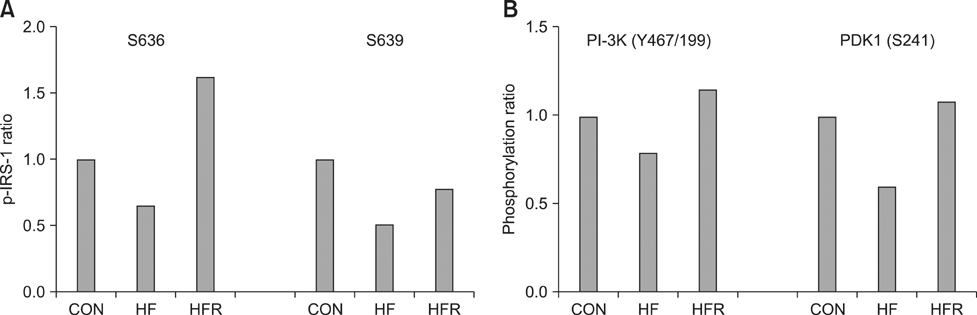
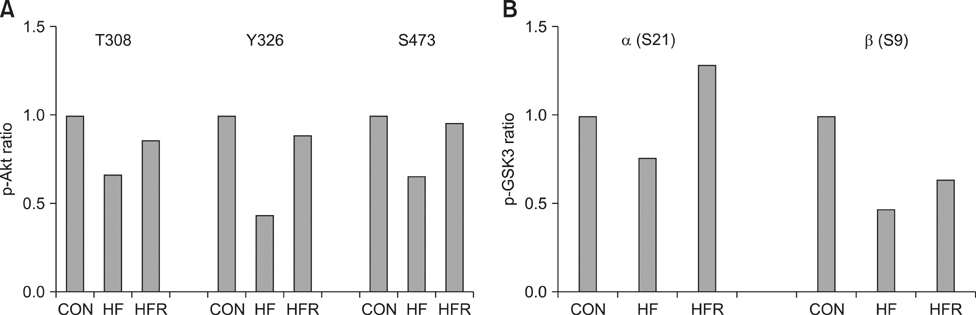
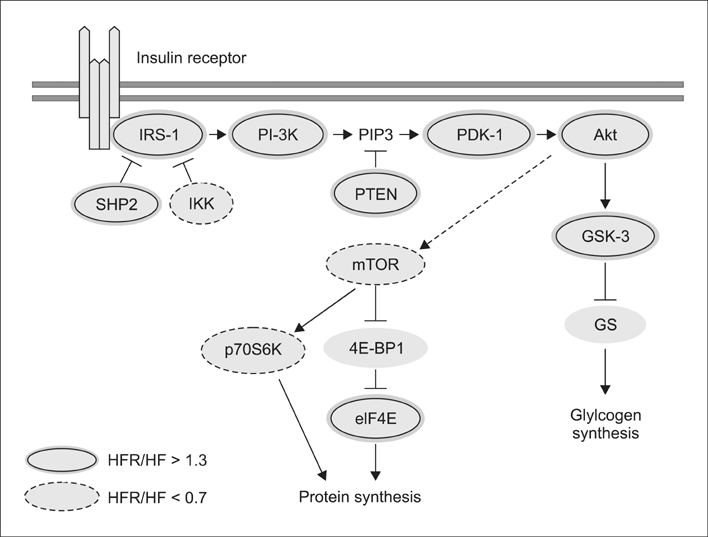
- The present study was conducted to investigate the effects of resveratrol on the insulin signaling pathway in the liver of obese mice. To accomplish this, we administered resveratrol to high fat diet-induced obese mice and examined the levels of protein phosphorylation in the liver using an antibody array. The phosphorylation levels of 10 proteins were decreased in the high fat diet and resveratrol (HFR) fed group relative to the levels in the high fat diet (HF) fed group. In contrast, the phosphorylation levels of more than 20 proteins were increased in the HFR group when compared with the levels of proteins in the HF group. Specifically, the phosphorylation levels of Akt (The308, Tyr326, Ser473) were restored to normal by resveratrol when compared with the levels in the HF group. In addition, the phosphorylation levels of IRS-1 (Ser636/Ser639), PI-3K p85-subunit alpha/gamma(Tyr467/Tyr199), PDK1 (Ser241), GSK-3alpha (S21) and GSK-3 (Ser9), which are involved in the insulin signaling pathway, were decreased in the HF group, whereas the levels were restored to normal in the HFR group. Overall, the results show that resveratrol restores the phosphorylation levels of proteins involved in the insulin signaling pathway, which were decreased by a high fat diet.
MeSH Terms
Figure
Reference
-
1. Alberti KG, Zimmet P, Shaw J. The metabolic syndrome-a new worldwide definition. Lancet. 2005; 366:1059–1062.
Article2. Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A, Prabhu VV, Allard JS, Lopez-Lluch G, Lewis K, Pistell PJ, Poosala S, Becker KG, Boss O, Gwinn D, Wang M, Ramaswamy S, Fishbein KW, Spencer RG, Lakatta EG, Le Couteur D, Shaw RJ, Navas P, Puigserver P, Ingram DK, de Cabo R, Sinclair DA. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006; 444:337–342.
Article3. Bhat KPL, Kosmeder JW 2nd, Pezzuto JM. Biological effects of resveratrol. Antioxid Redox Signal. 2001; 3:1041–1064.
Article4. Casamayor A, Morrice NA, Alessi DR. Phosphorylation of Ser-241 is essential for the activity of 3-phosphoinositide-dependent protein kinase-1: Identification of five sites of phosphorylation in vivo. Biochem J. 1999; 342:287–292.
Article5. Conus NM, Hannan KM, Cristiano BE, Hemmings BA, Pearsom RB. Direct identification of tyrosine 474 as a regulatory phosphorylation site for the Akt protein kinase. J Biol Chem. 2002; 277:38021–38028.
Article6. Duffy SJ, Vita JA. Effects of phenolics on vascular endothelial function. Curr Opin Lipidol. 2003; 14:21–27.
Article7. Fauconneau B, Waffo-Teguo P, Huguet F, Barrier L, Decendit A, Merillon JM. Comparative study of radical scavenger and antioxidant properties of phenolic compounds from Vitis vinifera cell cultures using in vitro tests. Life Sci. 1997; 61:2103–2110.
Article8. Frankel EN, Waterhouse AL, Kinsella JE. Inhibition of human LDL oxidation by resveratrol. Lancet. 1993; 341:1103–1104.
Article9. Hansen TK, Thiel S, Wouters PJ, Christiansen JS, Van den Berghe G. Intensive insulin therapy exerts antiinflammatory effects in critically ill patients and counteracts the adverse effect of low mannose-binding lectin levels. J Clin Endocrinol Metab. 2003; 88:1082–1088.
Article10. Hsieh T, Juan G, Darzynkiewicz Z, Wu JM. Resveratrol increases nitric oxide synthase, induces accumulation of p53 and p21WAF1/CIP1, and suppresses cultured bovine pulmonary artery endothelial cell proliferation by perturbing progression through S and G2. Cancer Res. 1999; 59:2596–2601.11. Kang W, Hong HJ, Guan J, Kim DG, Yang EJ, Koh G, Park D, Han CH, Lee YJ, Lee DH. Resveratrol improves insulin signaling in a tissue-specific manner under insulin-resistant conditions only: in vitro and in vivo experiments in rodents. Metabolism. 2012; 61:424–433.
Article12. Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P, Geny B, Laakso M, Puigserver P, Auwerx J. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell. 2006; 127:1109–1122.
Article13. Li X, Lu Y, Jin W, Liang K, Mills GB, Fan Z. Autophosphorylation of Akt at threonine 72 and serine 246. A potential mechanism of regulation of Akt kinase activity. J Biol Chem. 2006; 281:13837–13843.14. McManus EJ, Sakamoto K, Armit LJ, Ronaldson L, Shpiro N, Marquez R, Alessi DR. Role that phosphorylation of GSK3 plays in insulin and Wnt signalling defined by knockin analysis. EMBO J. 2005; 24:1571–1583.
Article15. Pace-Asciak CR, Rounova O, Hahn SE, Diamandis EP, Goldberg DM. Wines and grape juices as modulators of platelet aggregation in healthy human subjects. Clin Chim Acta. 1996; 246:163–182.
Article16. Postic C, Girard J. Contribution of de novo fatty acid synthesis to hepatic steatosis and insulin resistance: lessons from genetically engineered mice. J Clin Invest. 2008; 118:829–838.
Article17. Renaud S, de Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992; 339:1523–1526.
Article18. Sipula IJ, Brown NF, Perdomo G. Rapamycin-mediated inhibition of mammalian target of rapamycin in skeletal muscle cells reduces glucose utilization and increases fatty acid oxidation. Metabolism. 2006; 55:1637–1644.
Article19. Soleas GJ, Diamandis EP, Goldberg DM. The world of resveratrol. Adv Exp Med Biol. 2001; 492:159–182.
Article20. Thorburn AW, Gumbiner B, Bulacan F, Wallace P, Henry RR. Intracellular glucose oxidation and glycogen synthase activity are reduced in non-insulin-dependent (Type II) diabetes independent of impaired glucose uptake. J Clin Invest. 1990; 85:522–529.
Article21. Valenzano DR, Terzibasi E, Genade T, Cattaneo A, Domenici L, Cellerino A. Resveratrol prolongs lifespan and retards the onset of age-related markers in a short-lived vertebrate. Curr Biol. 2006; 16:296–300.
Article22. White MF. IRS proteins and the common path to diabetes. Am J Physiol Endocrinol Metab. 2002; 283:E413–E422.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory effects of resveratrol on hepatitis B virus X protein-induced hepatocellular carcinoma
- Effects of resveratrol on hepatic autophagy in high fat diet-induced obese mice
- Inhibitory Effects of Resveratrol on Airway Remodeling by Transforming Growth Factor-β/Smad Signaling Pathway in Chronic Asthma Model
- Resveratrol modulates the Akt/GSK-3β signaling pathway in a middle cerebral artery occlusion animal model
- Effect of resveratrol on the metastasis of 4T1 mouse breast cancer cells in vitro and in vivo