J Periodontal Implant Sci.
2011 Oct;41(5):253-258. 10.5051/jpis.2011.41.5.253.
Secondary closure of an extraction socket using the double-membrane guided bone regeneration technique with immediate implant placement
- Affiliations
-
- 1Department of Dentistry, Inha University School of Medicine, Incheon, Korea. yunjh@inha.ac.kr
- KMID: 1783621
- DOI: http://doi.org/10.5051/jpis.2011.41.5.253
Abstract
- PURPOSE
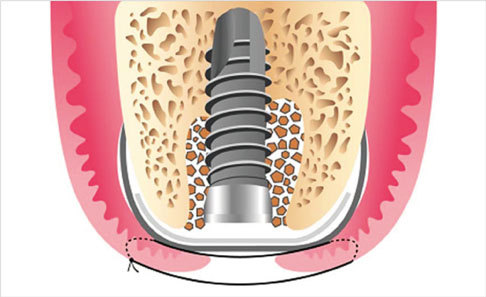
Immediate implantation presents challenges regarding site healing, osseointegration, and obtaining complete soft-tissue coverage of the extraction socket, especially in the posterior area. This last issue is addressed herein using the double-membrane (collagen membrane+high-density polytetrafluoroethylene [dPTFE] membrane) technique in two clinical cases of posterior immediate implant placement.
METHODS
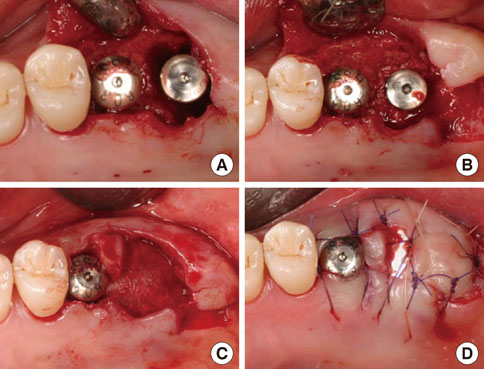
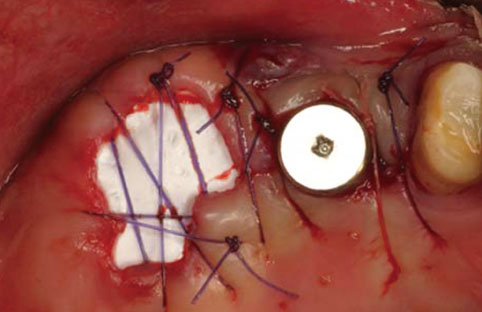
An implant was placed immediately after atraumatically extracting the maxillary posterior tooth. The gap between the coronal portion of the fixture and the adjacent bony walls was filled with allograft material. In addition, a collagen membrane (lower) and dPTFE membrane (upper) were placed in a layer-by-layer manner to enable the closure of the extraction socket without a primary flap closure, thus facilitating the preservation of keratinized mucosa. The upper dPTFE membrane was left exposed for 4 weeks, after which the membrane was gently removed using forceps without flap elevation.
RESULTS
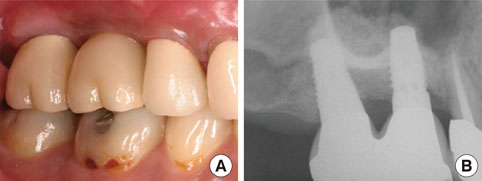
There was considerable plaque deposition on the outer surface of the dPTFE membrane but not on the inner surface. Moreover, scanning electron microscopy of the removed membrane revealed only a small amount of bacteria on the inner surface of the membrane. The peri-implant tissue was favorable both clinically and radiographically after a conventional dental-implant healing period.
CONCLUSIONS
Secondary closure of the extraction socket and immediate guided bone regeneration using the double-membrane technique may produce a good clinical outcome after immediate placement of a dental implant in the posterior area.
Keyword
MeSH Terms
Figure
Reference
-
1. Covani U, Cornelini R, Barone A. Bucco-lingual bone remodeling around implants placed into immediate extraction sockets: a case series. J Periodontol. 2003. 74:268–273.
Article2. Paolantonio M, Dolci M, Scarano A, d'Archivio D, di Placido G, Tumini V, et al. Immediate implantation in fresh extraction sockets. A controlled clinical and histological study in man. J Periodontol. 2001. 72:1560–1571.
Article3. Wilson TG Jr, Schenk R, Buser D, Cochran D. Implants placed in immediate extraction sites: a report of histologic and histometric analyses of human biopsies. Int J Oral Maxillofac Implants. 1998. 13:333–341.4. Bartee BK. The use of high-density polytetrafluoroethylene membrane to treat osseous defects: clinical reports. Implant Dent. 1995. 4:21–26.5. Bartee BK, Carr JA. Evaluation of a high-density polytetrafluoroethylene (n-PTFE) membrane as a barrier material to facilitate guided bone regeneration in the rat mandible. J Oral Implantol. 1995. 21:88–95.6. Crump TB, Rivera-Hidalgo F, Harrison JW, Williams FE, Guo IY. Influence of three membrane types on healing of bone defects. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996. 82:365–374.
Article7. Walters SP, Greenwell H, Hill M, Drisko C, Pickman K, Scheetz JP. Comparison of porous and non-porous teflon membranes plus a xenograft in the treatment of vertical osseous defects: a clinical reentry study. J Periodontol. 2003. 74:1161–1168.
Article8. Hoffmann O, Bartee BK, Beaumont C, Kasaj A, Deli G, Zafiropoulos GG. Alveolar bone preservation in extraction sockets using non-resorbabledPTFE membranes: a retrospective non-randomized study. J Periodontol. 2008. 79:1355–1369.
Article9. Irinakis T. Rationale for socket preservation after extraction of a single-rooted tooth when planning for future implant placement. J Can Dent Assoc. 2006. 72:917–922.10. Juodzbalys G, Wang HL. Soft and hard tissue assessment of immediate implant placement: a case series. Clin Oral Implants Res. 2007. 18:237–243.
Article11. Oh TJ, Meraw SJ, Lee EJ, Giannobile WV, Wang HL. Comparative analysis of collagen membranes for the treatment of implant dehiscence defects. Clin Oral Implants Res. 2003. 14:80–90.
Article12. Moses O, Pitaru S, Artzi Z, Nemcovsky CE. Healing of dehiscence-type defects in implants placed together with different barrier membranes: a comparative clinical study. Clin Oral Implants Res. 2005. 16:210–219.
Article13. Caffesse RG, Nasjleti CE, Morrison EC, Sanchez R. Guided tissue regeneration: comparison of bioabsorbable and non-bioabsorbable membranes. Histologic and histometric study in dogs. J Periodontol. 1994. 65:583–591.
Article14. Barber HD, Lignelli J, Smith BM, Bartee BK. Using a dense PTFE membrane without primary closure to achieve bone and tissue regeneration. J Oral Maxillofac Surg. 2007. 65:748–752.
Article15. Brägger U, Hämmerle CH, Lang NP. Immediate transmucosal implants using the principle of guided tissue regeneration (II). A cross-sectional study comparing the clinical outcome 1 year after immediate to standard implant placement. Clin Oral Implants Res. 1996. 7:268–276.
Article16. Lang NP, Brägger U, Hämmerle CH, Sutter F. Immediate transmucosal implants using the principle of guided tissue regeneration. I. Rationale, clinical procedures and 30-month results. Clin Oral Implants Res. 1994. 5:154–163.
Article17. Lekovic V, Camargo PM, Klokkevold PR, Weinlaender M, Kenney EB, Dimitrijevic B, et al. Preservation of alveolar bone in extraction sockets using bioabsorbable membranes. J Periodontol. 1998. 69:1044–1049.
Article18. Lekovic V, Kenney EB, Weinlaender M, Han T, Klokkevold P, Nedic M, et al. A bone regenerative approach to alveolar ridge maintenance following tooth extraction. Report of 10 cases. J Periodontol. 1997. 68:563–570.
Article19. Iasella JM, Greenwell H, Miller RL, Hill M, Drisko C, Bohra AA, et al. Ridge preservation with freeze-dried bone allograft and a collagen membrane compared to extraction alone for implant site development: a clinical and histologic study in humans. J Periodontol. 2003. 74:990–999.
Article20. Wang HL, Boyapati L. "PASS" principles for predictable bone regeneration. Implant Dent. 2006. 15:8–17.
Article21. Trombelli L, Farina R, Marzola A, Bozzi L, Liljenberg B, Lindhe J. Modeling and remodeling of human extraction sockets. J Clin Periodontol. 2008. 35:630–639.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Membrane Fixation using Healing Abutment during Guided Bone Regeneration: A Report of Two Cases
- The Sausage Technique using Collagen Membrane without Autogenous Bone Graft: A Case Report
- Free gingival graft for the increase of peri-implant attached keratinized mucosa decreased after guided bone regeneration
- Immediate implant placement in fresh extraction sockets
- Soft-tissue management for primary closure in immediate implant placement