J Korean Med Sci.
2013 Dec;28(12):1729-1733. 10.3346/jkms.2013.28.12.1729.
Application of the Multiplex Cytokine Analysis to Monitor Xenogeneic Immune Responses to the Porcine Islet Graft in Non-Human Primate
- Affiliations
-
- 1Department of Microbiology and Immunology, Seoul National University College of Medicine, Seoul, Korea. chgpark@snu.ac.kr
- 2Translational Xenotransplantation Research Center, Seoul National University College of Medicine, Seoul, Korea.
- 3Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- 4Institute of Endemic Diseases, Seoul National University College of Medicine, Seoul, Korea.
- 5Organ Transplantation Center, Myong-Ji Hospital, Goyang, Korea.
- KMID: 1779412
- DOI: http://doi.org/10.3346/jkms.2013.28.12.1729
Abstract
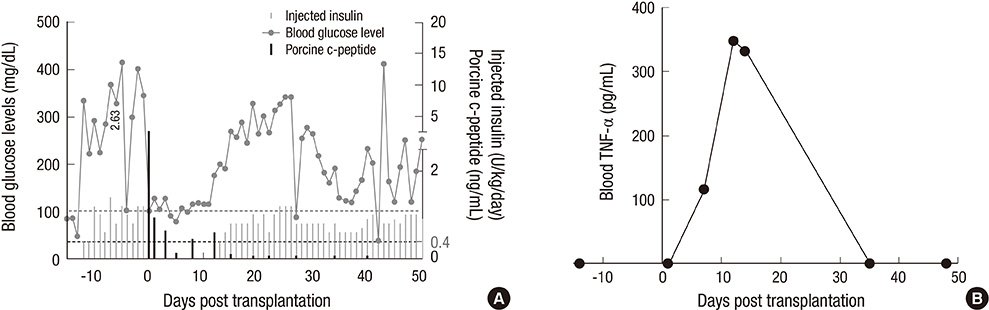
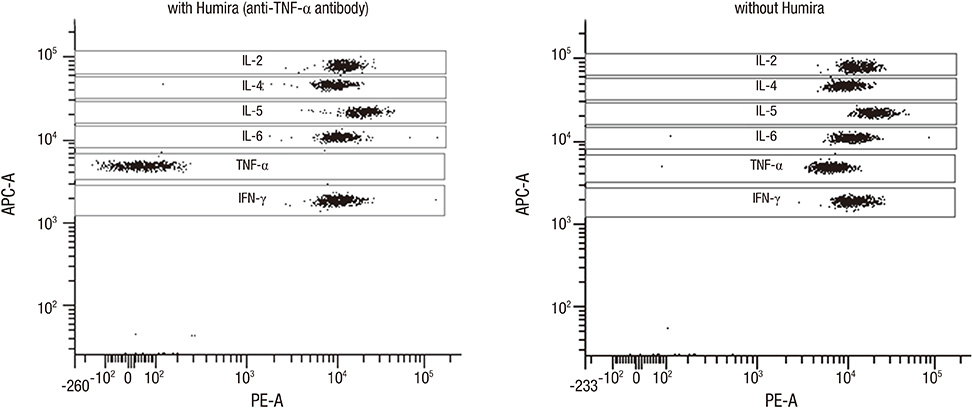
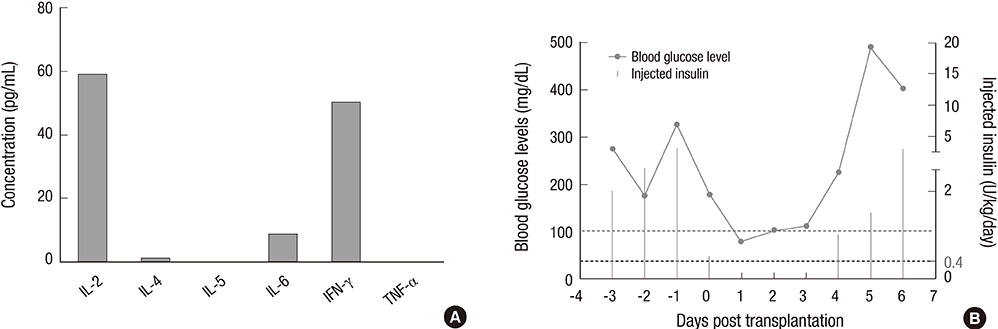
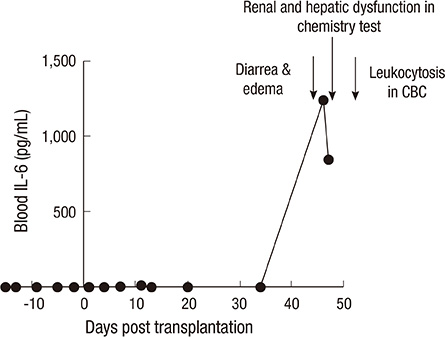
- Non-human primate studies must be conducted prior to the clinical trial of xenotransplantation. In order to develop clinically applicable immune-modulatory regimen through non-human primate studies, close monitoring of xenogeneic immune responses is required. We adopted multiplex cytokine analysis in assessment of the immune status during the course of pig-to-non-human primate islet transplantation. This study aimed to assess the feasibility of this multiplex cytokine assay in the development of immune-modulatory regimen. Using this assay, we were able to detect different cytokines with a minimal usage of blood samples, and this allowed us to detect various immunological situations in the recipients. Detection of TNF-alpha surge (347.8 pg/mL) guided us to block TNF-alpha in the early phase of transplantation. Supportive information for in vivo efficacy of cytokine neutralizing antibody could be speculated by in vitro neutralization assay (1,250 pg/mL --> 0 pg/mL). In addition, periodic monitoring of cytokines in peripheral blood allowed the detection of the infection episode prior to other routine assays. These benefits of multiplex cytokine assay may be generally applied to other pre-clinical research, which is a prerequisite for clinical trials.
MeSH Terms
-
Animals
Antibodies, Neutralizing/immunology
Blood Cell Count
Cytokines/*blood/immunology
Immunoassay/*methods
Interleukin-6/blood
Islets of Langerhans Transplantation/*immunology
Macaca mulatta
Swine
Transplantation, Heterologous
Tumor Necrosis Factor-alpha/blood/immunology
Antibodies, Neutralizing
Cytokines
Interleukin-6
Tumor Necrosis Factor-alpha
Figure
Reference
-
1. First WHO global consultation on regulatory requirements for xenotransplantation clinical trials: Changsha, China, 19-21 November 2008: the Changsha Communiqué. Xenotransplantation. 2009; 16:61–63.2. Cooper DK, Casu A. The International Xenotransplantation Association consensus statement on conditions for undertaking clinical trials of porcine islet products in type 1 diabetes: chapter 4: pre-clinical efficacy and complication data required to justify a clinical trial. Xenotransplantation. 2009; 16:229–238.3. Marigliano M, Bertera S, Grupillo M, Trucco M, Bottino R. Pig-to-nonhuman primates pancreatic islet xenotransplantation: an overview. Curr Diab Rep. 2011; 11:402–412.4. Van der Windt DJ, Bottino R, Kumar G, Wijkstrom M, Hara H, Ezzelarab M, Ekser B, Phelps C, Murase N, Casu A, et al. Clinical islet xenotransplantation: how close are we? Diabetes. 2012; 61:3046–3055.5. Jin SM, Kim KS, Lee SY, Gong CH, Park SK, Yu JE, Yeom SC, Yoon TW, Ha J, Park CG, et al. Enhanced prediction of porcine islet yield and posttransplant outcome using a combination of quantitative histomorphometric parameters and flow cytometry. Cell Transplant. 2010; 19:299–311.6. Atalar K, Afzali B, Lord G, Lombardi G. Relative roles of Th1 and Th17 effector cells in allograft rejection. Curr Opin Organ Transplant. 2009; 14:23–29.7. Pujol-Borrell R, Todd I, Doshi M, Bottazzo GF, Sutton R, Gray D, Adolf GR, Feldmann M. HLA class II induction in human islet cells by interferon-gamma plus tumour necrosis factor or lymphotoxin. Nature. 1987; 326:304–306.8. Sato N, Goto T, Haranaka K, Satomi N, Nariuchi H, Mano-Hirano Y, Sawasaki Y. Actions of tumor necrosis factor on cultured vascular endothelial cells: morphologic modulation, growth inhibition, and cytotoxicity. J Natl Cancer Inst. 1986; 76:1113–1121.9. Ranges GE, Figari IS, Espevik T, Palladino MA Jr. Inhibition of cytotoxic T cell development by transforming growth factor beta and reversal by recombinant tumor necrosis factor alpha. J Exp Med. 1987; 166:991–998.10. Philip R, Epstein LB. Tumour necrosis factor as immunomodulator and mediator of monocyte cytotoxicity induced by itself, gamma-interferon and interleukin-1. Nature. 1986; 323:86–89.11. Lee JR, Seok CJ, Kim JS, Chang JM, Seo JW. Expression of NF-kappaB and cytokines in chronic rejection of transplanted murine heart. J Korean Med Sci. 2001; 16:397–406.12. Abdallah AN, Billes MA, Attia Y, Doutremepuich C, Cassaigne A, Iron A. Evaluation of plasma levels of tumour necrosis factor alpha and interleukin-6 as rejection markers in a cohort of 142 heart-grafted patients followed by endomyocardial biopsy. Eur Heart J. 1997; 18:1024–1029.13. Speier S, Nyqvist D, Cabrera O, Yu J, Molano RD, Pileggi A, Moede T, Köhler M, Wilbertz J, Leibiger B, et al. Noninvasive in vivo imaging of pancreatic islet cell biology. Nat Med. 2008; 14:574–578.14. Speier S, Nyqvist D, Köhler M, Caicedo A, Leibiger IB, Berggren PO. Noninvasive high-resolution in vivo imaging of cell biology in the anterior chamber of the mouse eye. Nat Protoc. 2008; 3:1278–1286.15. Abdulreda MH, Faleo G, Molano RD, Lopez-Cabezas M, Molina J, Tan Y, Echeverria OA, Zahr-Akrawi E, Rodriguez-Diaz R, Edlund PK, et al. High-resolution, noninvasive longitudinal live imaging of immune responses. Proc Natl Acad Sci U S A. 2011; 108:12863–12868.16. Perez VL, Caicedo A, Berman DM, Arrieta E, Abdulreda MH, Rodriguez-Diaz R, Pileggi A, Hernandez E, Dubovy SR, Parel JM, et al. The anterior chamber of the eye as a clinical transplantation site for the treatment of diabetes: a study in a baboon model of diabetes. Diabetologia. 2011; 54:1121–1126.17. Behndig A, Markström K. Determination of the aqueous humor volume by three-dimensional mapping of the anterior chamber. Ophthalmic Res. 2005; 37:13–16.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Experimental Pancreas Islet Isolation in Non- Human Primate
- Current Status and Future Perspectives of Xenotransplantation
- Effect of Human or Mouse IL-7 on the Homeostasis of Porcine T Lymphocytes
- Influence of Interferon-gamma Deficiency in Immune Tolerance Induced by Male Islet Transplantation
- The profile of cytokine mRNA expression and immune cell infiltration in the rejection of allo- and xenogeneic skin transplantation