Korean J Pain.
2010 Jun;23(2):116-123. 10.3344/kjp.2010.23.2.116.
The Neurological Safety of Epidural Pamidronate in Rats
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Jeju National University College of Medicine, Jeju, Korea. ppsshh11@daum.net
- 2Department of Anesthesiology and Pain Medicine, Seoul National University College of Medicine, Seoul, Korea.
- 3Department of Anesthesiology and Pain Medicine, Samsung Seoul Hospital, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 1770543
- DOI: http://doi.org/10.3344/kjp.2010.23.2.116
Abstract
- BACKGROUND
Pamidronate is a potent inhibitor of osteoclast-mediated bone resorption. Recently, the drug has been known to relieve bone pain. We hypothesized that direct epidural administration of pamidronate could have various advantages over oral administration with respect to dosage, side effects, and efficacy. Therefore, we evaluated the neuronal safety of epidurally-administered pamidronate.
METHODS
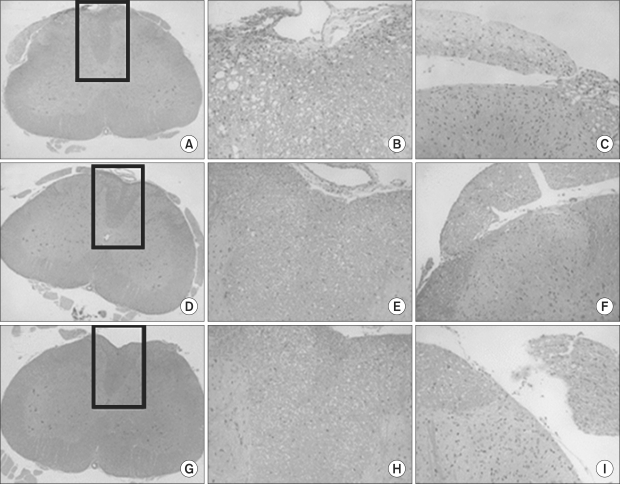
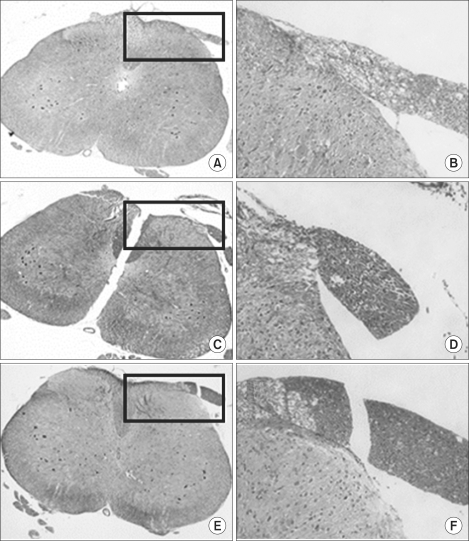
Twenty-seven rats weighing 250-350 g were equally divided into 3 groups. Each group received an epidural administration with either 0.3 ml (3.75 mg) of pamidronate (group P), 0.3 ml of 40% alcohol (group A), or 0.3 ml of normal saline (group N). A Pinch-toe test, motor function evaluation, and histopathologic examination of the spinal cord to detect conditions such as chromatolysis, meningeal inflammation, and neuritis, were performed on the 2nd, 7th, and 21st day following administration of each drug.
RESULTS
All rats in group A showed an abnormal response to the pinch-toe test and decreased motor function during the entire evaluation period. Abnormal histopathologic findings, including neuritis and meningeal inflammation were observed only in group A rats. Rats in group P, with the exception of 1, and group N showed no significant sensory/motor dysfunction over a 3-week observation period. No histopathologic changes were observed in groups P and N.
CONCLUSIONS
Direct epidural injection of pamidronate (about 12.5 mg/kg) showed no neurotoxic evidence in terms of sensory/motor function evaluation and histopathologic examination.
Keyword
MeSH Terms
Figure
Reference
-
1. Gangji V, Appelboom T. Analgesic effect of intravenous pamidronate on chronic back pain due to osteoporotic vertebral fractures. Clin Rheumatol. 1999; 18:266–267. PMID: 11206358.
Article2. Reid IR. Bisphosphonates: new indications and methods of administration. Curr Opin Rheumatol. 2003; 15:458–463. PMID: 12819475.
Article3. Bonabello A, Galmozzi MR, Bruzzese T, Zara GP. Analgesic effect of bisphosphonates in mice. Pain. 2001; 91:269–275. PMID: 11275384.
Article4. Bonabello A, Galmozzi MR, Canaparo R, Serpe L, Zara GP. Long-term analgesic effect of clodronate in rodents. Bone. 2003; 33:567–574. PMID: 14555260.
Article5. Cui JG, Holmin S, Mathiesen T, Meyerson BA, Linderoth B. Possible role of inflammatory mediators in tactile hypersensitivity in rat models of mononeuropathy. Pain. 2000; 88:239–248. PMID: 11068111.
Article6. Kawabata A, Kawao N, Hironaka Y, Ishiki T, Matsunami M, Sekiguchi F. Antiallodynic effect of etidronate, a bisphosphonate, in rats with adjuvant-induced arthritis: involvement of ATP-sensitive K+ channels. Neuropharmacology. 2006; 51:182–190. PMID: 16678221.
Article7. Sevcik MA, Luger NM, Mach DB, Sabino MA, Peters CM, Ghilardi JR, et al. Bone cancer pain: the effects of the bisphosphonate alendronate on pain, skeletal remodeling, tumor growth and tumor necrosis. Pain. 2004; 111:169–180. PMID: 15327821.
Article8. Walker K, Medhurst SJ, Kidd BL, Glatt M, Bowes M, Patel S, et al. Disease modifying and anti-nociceptive effects of the bisphosphonate, zoledronic acid in a model of bone cancer pain. Pain. 2002; 100:219–229. PMID: 12467993.
Article9. Yanow J, Pappagallo M, Pillai L. Complex regional pain syndrome (CRPS/RSD) and neuropathic pain: role of intravenous bisphosphonates as analgesics. ScientificWorld-Journal. 2008; 8:229–236. PMID: 18661047.
Article10. Hassenbusch SJ, Portenoy RK, Cousins M, Buchser E, Deer TR, Du Pen SL, et al. Polyanalgesic Consensus Conference 2003: an update on the management of pain by intraspinal drug delivery-- report of an expert panel. J Pain Symptom Manage. 2004; 27:540–563. PMID: 15165652.
Article11. Hayashi N, Weinstein JN, Meller ST, Lee HM, Spratt KF, Gebhart GF. The effect of epidural injection of betamethasone or bupivacaine in a rat model of lumbar radiculopathy. Spine. 1998; 23:877–885. PMID: 9580954.
Article12. Kawakami M, Matsumoto T, Hashizume H, Kuribayashi K, Tamaki T. Epidural injection of cyclooxygenase-2 inhibitor attenuates pain-related behavior following application of nucleus pulposus to the nerve root in the rat. J Orthop Res. 2002; 20:376–381. PMID: 11918320.
Article13. Bajrović F, Sketelj J. Extent of nociceptive dermatomes in adult rats is not primarily maintained by axonal competition. Exp Neurol. 1998; 150:115–121. PMID: 9514823.
Article14. Ochi T, Ohkubo Y, Mutoh S. FR143166 attenuates spinal pain transmission through activation of the serotonergic system. Eur J Pharmacol. 2002; 452:319–324. PMID: 12359273.
Article15. Sinnott CJ, Cogswell LP III, Johnson A, Strichartz GR. On the mechanism by which epinephrine potentiates lidocaine's peripheral nerve block. Anesthesiology. 2003; 98:181–188. PMID: 12502995.
Article16. Chatani K, Kawakami M, Weinstein JN, Meller ST, Gebhart GF. Characterization of thermal hyperalgesia, c-fos expression, and alterations in neuropeptides after mechanical irritation of the dorsal root ganglion. Spine. 1995; 20:277–289. PMID: 7537391.
Article17. Benford HL, McGowan NW, Helfrich MH, Nuttall ME, Rogers MJ. Visualization of bisphosphonate-induced caspase-3 activity in apoptotic osteoclasts in vitro. Bone. 2001; 28:465–473. PMID: 11344045.
Article18. Body JJ, Diel IJ, Lichinitser MR, Kreuser ED, Dornoff W, Gorbunova VA, et al. Intravenous ibandronate reduces the incidence of skeletal complications in patients with breast cancer and bone metastases. Ann Oncol. 2003; 14:1399–1405. PMID: 12954579.
Article19. Conte PF, Latreille J, Mauriac L, Calabresi F, Santos R, Campos D, et al. Delay in progression of bone metastases in breast cancer patients treated with intravenous pamidronate: results from a multinational randomized controlled trial. The Aredia Multinational Cooperative Group. J Clin Oncol. 1996; 14:2552–2559. PMID: 8823335.
Article20. Diener KM. Bisphosphonates for controlling pain from metastatic bone disease. Am J Health Syst Pharm. 1996; 53:1917–1927. PMID: 8862204.
Article21. Kubalek I, Fain O, Paries J, Kettaneh A, Thomas M. Treatment of reflex sympathetic dystrophy with pamidronate: 29 cases. Rheumatology (Oxford). 2001; 40:1394–1397. PMID: 11752511.
Article22. Robinson JN, Sandom J, Chapman PT. Efficacy of pamidronate in complex regional pain syndrome type I. Pain Med. 2004; 5:276–280. PMID: 15367305.
Article23. Abildgaard N, Glerup H, Rungby J, Bendix-Hansen K, Kassem M, Brixen K, et al. Biochemical markers of bone metabolism reflect osteoclastic and osteoblastic activity in multiple myeloma. Eur J Haematol. 2000; 64:121–129. PMID: 10997332.
Article24. Gallacher SJ, Ralston SH, Patel U, Boyle IT. Side-effects of pamidronate. Lancet. 1989; 2:42–43. PMID: 2567810.
Article25. Dodwell DJ, Howell A, Ford J. Reduction in calcium excretion in women with breast cancer and bone metastases using the oral bisphosphonate pamidronate. Br J Cancer. 1990; 61:123–125. PMID: 2297483.
Article26. Morton AR, Howell A. Bisphosphonates and bone metastases. Br J Cancer. 1988; 58:556–557. PMID: 3064797.
Article27. Harinck HI, Papapoulos SE, Blanksma HJ, Moolenaar AJ, Vermeij P, Bijvoet OL. Paget's disease of bone: early and late responses to three different modes of treatment with aminohydroxypropylidene bisphosphonate (APD). Br Med J (Clin Res Ed). 1987; 295:1301–1305.
Article28. Ralston SH, Gardner MD, Dryburgh FJ, Jenkins AS, Cowan RA, Boyle IT. Comparison of aminohydroxypropylidene diphosphonate, mithramycin, and corticosteroids/calcitonin in treatment of cancer-associated hypercalcaemia. Lancet. 1985; 2:907–910. PMID: 2865417.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Biomechanical Analysis of the Effect of Pamidronate on Prevention of Osteoporosis in Ovariectomized Rats
- Evaluation of the Neurological Safety of Epidural Milnacipran in Rats
- The Effect of Pamidronate on Prevention of Osteoporosis in Ovariectomized Rats
- Epidural hematoma treated by aspiration after transforaminal epidural steroid injection - A case report -
- High Concentrations of Pamidronate in Bone Weaken the Mechanical Properties of Intact Femora in a Rat Model