J Korean Med Sci.
2014 Mar;29(3):438-440. 10.3346/jkms.2014.29.3.438.
Intratracheal Administration of Umbilical Cord Blood-Derived Mesenchymal Stem Cells in a Patient with Acute Respiratory Distress Syndrome
- Affiliations
-
- 1Department of Pulmonary and Critical Care Medicine, Asan Medical Center, College of Medicine, University of Ulsan, Seoul, Korea. sbhong@amc.seoul.kr
- KMID: 1734934
- DOI: http://doi.org/10.3346/jkms.2014.29.3.438
Abstract
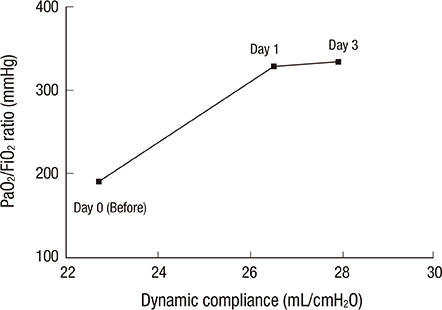
- Umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) have been introduced as a possible therapy in acute lung injury and acute respiratory distress syndrome (ARDS). This case history is reported of a 59-yr-old man who was treated with MSCs in the course of ARDS and subsequent pulmonary fibrosis. He received a long period of mechanical ventilation and weaning proved difficult. On hospital day 114, he underwent the intratracheal administration of UCB-derived MSCs at a dose of 1 x 10(6)/kg. After cell infusion, an immediate improvement was shown in his mental status, his lung compliance (from 22.7 mL/cmH2O to 27.9 mL/cmH2O), PaO2/FiO2 ratio (from 191 mmHg to 334 mmHg) and his chest radiography over the course of three days. Even though he finally died of repeated pulmonary infection, our current findings suggest the possibility of using MSCs therapy in an ARDS patient. It is the first clinical case of UCB-derived MSCs therapy ever reported.
MeSH Terms
-
Bacterial Infections/diagnosis
Drug Resistance, Multiple, Bacterial
Fetal Blood/*cytology
Humans
Male
*Mesenchymal Stem Cell Transplantation
Mesenchymal Stromal Cells/*cytology
Middle Aged
Respiratory Distress Syndrome, Adult/complications/radiography/*surgery
Seizures/etiology
Shock, Septic/diagnosis
Tomography, X-Ray Computed
Treatment Outcome
Figure
Reference
-
1. Loebinger MR, Janes SM. Stem cells for lung disease. Chest. 2007; 132:279–285.2. Lee JW, Fang X, Krasnodembskaya A, Howard JP, Matthay MA. Concise review: mesenchymal stem cells for acute lung injury: role of paracrine soluble factors. Stem Cells. 2011; 29:913–919.3. Matthay MA, Goolaerts A, Howard JP, Lee JW. Mesenchymal stem cells for acute lung injury: preclinical evidence. Crit Care Med. 2010; 38:10 Suppl. S569–S573.4. Mac Sweeney R, McAuley DF. Mesenchymal stem cell therapy in acute lung injury: is it time for a clinical trial? Thorax. 2012; 67:475–476.5. Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, Lanino E, Sundberg B, Bernardo ME, Remberger M, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet. 2008; 371:1579–1586.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Differentiation of Osteoblast Progenitor Cells from Human Umbilical Cord Blood
- Differential Potential of Stem Cells Following Their Origin: Subacromial Bursa, Bone Marrow, Umbilical Cord Blood
- Percutaneous transplantation of human umbilical cord-derived mesenchymal stem cells in a dog suspected to have fibrocartilaginous embolic myelopathy
- Difference in HLA-DR Expression of Human Umbilical Cord Blood Derived Mesenchymal Stem Cells after Tri-lineage Differentiation
- Endothelial progenitor cells and mesenchymal stem cells from human cord blood