J Vet Sci.
2008 Sep;9(3):267-272. 10.4142/jvs.2008.9.3.267.
The inhibitory effect of quercitrin gallate on iNOS expression induced by lipopolysaccharide in Balb/c mice
- Affiliations
-
- 1College of Veterinary Medicine, Chungbuk National University, Cheongju 361-763, Korea. bwahn@cbu.ac.kr
- 2College of Pharmacy, Chungbuk National University, Cheongju 361-763, Korea.
- KMID: 1718579
- DOI: http://doi.org/10.4142/jvs.2008.9.3.267
Abstract
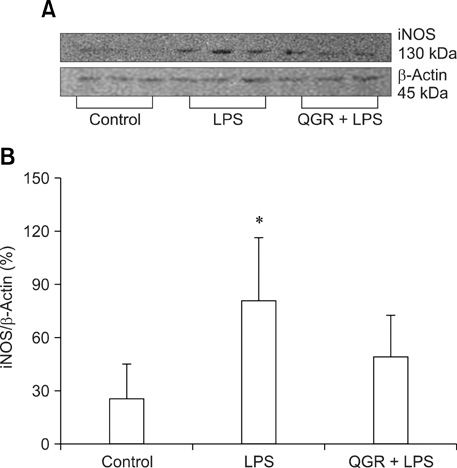
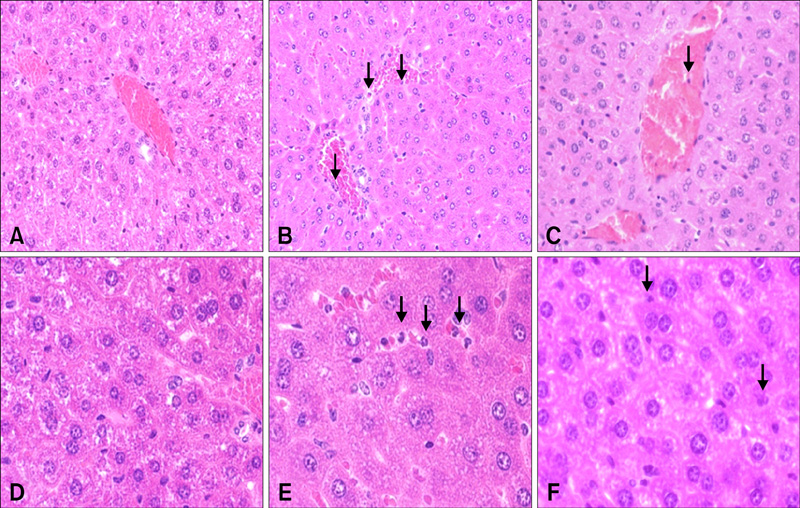
- Quercetin 3-O-beta-(2''-galloyl)-rhamnopyranoside (QGR) is a naturally occurring quercitrin gallate, which is a polyphenolic compound that was originally isolated from Persicaria lapathifolia (Polygonaceae). QGR has been shown to have an inhibitory effect on nitric oxide (NO) production in lipopolysaccharide (LPS)-stimulated macrophage RAW 264.7 cells. Therefore, this study was conducted to investigate the inhibitory effect of QGR on nitric oxide production and inducible nitric oxide synthases (iNOS) expression in LPS-stimulated Balb/c mice. To accomplish this, 10 mg/kg of QGR was administered via gavage once a day for 3 days. iNOS was then induced by intraperitoneal injection of LPS. Six hours after the LPS treatment the animals were sacrificed under ether anethesia. The serum levels of NO were then measured to determine if QGR exerted an inhibitory effect on NO production in vivo. LPS induced an approximately 6 fold increase in the expression of NO. However, oral administration of QGR reduced the LPS induced increase in NO by half. Furthermore, RT-PCR and western blot analysis revealed that the increased levels of iNOS expression that occurred in response to treatment with LPS were significantly attenuated in response to QGR pretreatment. Histologically, LPS induced the infiltration of polymorphonuclear neutrophils in portal veins and sinusoids and caused the formation of a large number of necrotic cells; however, pretreatment with QGR attenuated these LPS induced effects. Taken together, these results indicate that QGR inhibits iNOS expression in vivo as well as in vitro and has antiinflammatory potentials.
Keyword
MeSH Terms
-
Animals
DNA Primers
Gene Expression Regulation, Enzymologic/drug effects
Lipopolysaccharides/*pharmacology
Liver/drug effects/enzymology
Mice
Mice, Inbred BALB C
Nitric Oxide/blood
Nitric Oxide Synthase Type II/drug effects/*genetics
Quercetin/*analogs & derivatives/pharmacology
RNA, Messenger/genetics
Reverse Transcriptase Polymerase Chain Reaction
Figure
Reference
-
1. Akira S. Toll-like receptor and innate immunity. Adv Immunol. 2001. 78:1–56.2. Aktan F. iNOS-mediated nitric oxide production and its regulation. Life Sci. 2004. 75:639–653.
Article3. Aktan F, Henness S, Roufogalis BD, Ammit AJ. Gypenosides derived from Gynostemma pentaphyllum suppress NO synthesis in murine macrophages by inhibiting iNOS enzymatic activity and attenuating NF-B-mediated iNOS protein expression. Nitric Oxide. 2003. 8:235–242.
Article4. Alderton WK, Cooper CE, Knowles RG. Nitric oxide synthases: structure, function and inhibition. Biochem J. 2001. 357:593–615.
Article5. Bogdan C. Nitric oxide and the immune response. Nature Immunol. 2001. 2:907–916.
Article6. Buttery LD, Evans TJ, Springall DR, Carpenter A, Cohen J, Polak JM. Immunochemical localization of inducible nitric oxide synthase in endotoxin-treated rats. Lab Invest. 1994. 71:755–764.7. Grisham MB, Jourd'Heuil D, Wink DA. Nitric oxide. I. Physiological chemistry of nitric oxide and its metabolites: implications in inflammation. Am J Physiol. 1999. 276:G315–G321.8. Guha M, Mackman N. LPS induction of gene expression in human monocytes. Cell Signal. 2001. 13:85–94.
Article9. Haridas V, Arntzen CJ, Gutterman JU. Avicins, a family of triterpenoid saponins from Acacia victoriae (Bentham), inhibit activation of nuclear factor-kappaB by inhibiting both its nuclear localization and ability to bind DNA. Proc Natl Acad Sci USA. 2001. 98:11557–11562.
Article10. Hubbard AK, Giardina C. Regulation of ICAM-1 expression in mouse macrophages. Inflammation. 2000. 24:115–125.11. Hwang BY, Lee JH, Koo TH, Kim HS, Hong YS, Ro JS, Lee KS, Lee JJ. Kaurane diterpenes from Isodon japonicus inhibit nitric oxide and prostaglandin E2 production and NF-kappaB activation in LPS-stimulated macrophage RAW264.7 cells. Planta Med. 2001. 67:406–410.
Article12. Israël lA. The IKK complex: an integrator of all signals that activate NF-κB? Trends Cell Biol. 2000. 10:129–133.
Article13. Jin HZ, Hwang BY, Kim HS, Lee JH, Kim YH, Lee JJ. Antiinflammatory constituents of Celastrus orbiculatus inhibit the NF-kappaB activation and NO production. J Nat Prod. 2002. 65:89–91.
Article14. Kim BH, Cho SM, Reddy AM, Kim YS, Min KR, Kim Y. Down-regulatory effect of quercitrin gallate on nuclear factor-κB-dependent inducible nitric oxide synthase expression in lipopolysaccharide-stimulated macrophages RAW 264.7. Biochem Pharmacol. 2005. 69:1577–1583.
Article15. Kim Y, Jang DS, Park SH, Yun J, Min BK, Min KR, Lee HK. Flavonol glycoside gallate and ferulate esters from persicaria lapathifolia as inhibitors of superoxide production in human monocytes stimulated by unopsonized zymosan. Planta Med. 2000. 66:72–74.
Article16. Lee JH, Koo TH, Yoon H, Jung HS, Jin HZ, Lee K, Hong YS, Lee JJ. Inhibition of NF-kappa B activation through targeting I kappa B kinase by celastrol, a quinone methide triterpenoid. Biochem Pharmacol. 2006. 72:1311–1321.
Article17. Lin YL, Lin JK. (-)-Epigallocatechin-3-gallate blocks the induction of nitric oxide synthase by down-regulating lipopolysaccharide-induced activity of transcription factor nuclear factor-kappaB. Mol Pharmacol. 1997. 52:465–472.
Article18. Ma ZH, Ma QY, Sha HC, Wang LC. Effect of resveratrol on lipopolysaccharide-induced activation of rat peritoneal macrophages. Nan Fang Yi Ke Da Xue Xue Bao. 2006. 26:1363–1365.19. Magnani M, Crinelli R, Bianchi M, Antonelli A. The ubiquitin-dependent proteolytic system and other potential targets for the modulation of nuclear factor-κB (NF-κB). Curr Drug Targets. 2000. 1:387–399.
Article20. Manna SK, Sah NK, Newman RA, Cisneros A, Aggarwal BB. Oleandrin suppresses activation of nuclear transcription factor-kappaB, activator protein-1, and c-Jun NH2-terminal kinase. Cancer Res. 2000. 60:3838–3847.21. Mu MM, Chakravortty D, Sugiyama T, Koide N, Takahashi K, Mori I, Yoshida T, Yokochi T. The inhibitory action of quercetin on lipopolysaccharide-induced nitric oxide production in RAW 264.7 macrophage cells. J Endotoxin Res. 2001. 7:431–438.
Article22. Park HJ, Kim RG, Seo BR, Ha J, Ahn BT, Bok SH, Lee YS, Kim HJ, Lee KT. Saucernetin-7 and saucernetin-8 isolated from Saururus chinensis inhibit the LPS-induced production of nitric oxide and prostaglandin E2 in macrophage RAW264.7 cells. Planta Med. 2003. 69:947–950.
Article23. Peet GW, Li J. IκB kinases α and β show a random sequential kinetic mechanism and are inhibited by staurosporine and quercetin. J Biol Chem. 1999. 274:32655–32661.
Article24. Schmidt HH, Walter U. NO at work. Cell. 1994. 78:919–925.
Article25. Suzuki H, Chiba T, Kobayashi M, Takeuchi M, Furuichi K, Tanaka K. In vivo and in vitro recruitment of an IκBα-ubiquitin ligase to IKappaBalpha phosphorylated by IKK, leading to ubiquitination. Biochem Biophys Res Commun. 1999. 256:121–126.
Article26. Tian B, Brasier AR. Identification of a nuclear factor kappa B-dependent gene network. Recent Prog Horm Res. 2003. 58:95–130.
Article27. Wadsworth TL, Koop DR. Effects of the wine polyphenolics quercetin and resveratrol on pro-inflammatory cytokine expression in RAW 264.7 macrophages. Biochem Pharmacol. 1999. 57:941–949.
Article28. Xie QW, Cho HJ, Calaycay J, Mumford RA, Swiderek KM, Lee TD, Ding A, Troso T, Nathan C. Cloning and characterization of inducible nitric oxide synthase from mouse macrophages. Science. 1992. 256:225–228.
Article29. Xie QW, Kashiwabara Y, Nathan C. Role of transcription factor NF-κB/Rel in induction of nitric oxide synthase. J Biol Chem. 1994. 269:4705–4708.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Insulin Enhances Suppressive Effect of Lipopolysaccharide on Glucose-induced Proliferation of Vascular Smooth Muscle Cells
- Epigallocatechin-3-gallate Regulates Inducible Nitric Oxide Synthase Expression in Human Umbilical Vein Endothelial Cells
- Effect of Epigallocatechin-3-Gallate on Expression of iNOS mRNA and Production of NO in HaCaT Cell
- Diclofenac inhibits IFN-gamma plus lipopolysaccharide-induced iNOS gene expression via suppression of NF-kappaB activation in RAW 264.7 macrophages
- Inhibitory effects of tilianin on the expression of inducible nitric oxide synthase in low density lipoprotein receptor deficiency mice