J Clin Neurol.
2007 Mar;3(1):38-44. 10.3988/jcn.2007.3.1.38.
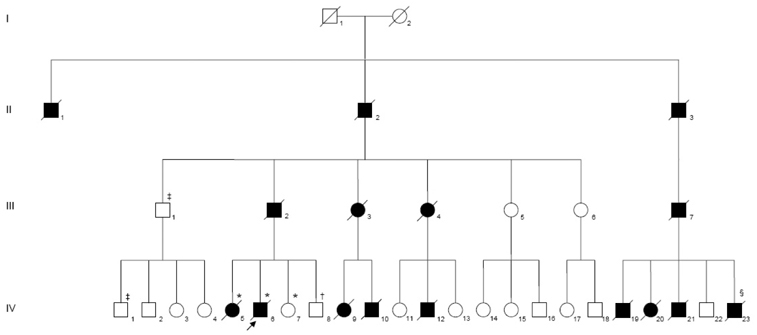
Anticipation and Phenotypic Heterogeneity in Korean Familial Amyotrophic Lateral Sclerosis with Superoxide Dismutase 1 Gene Mutation
- Affiliations
-
- 1Department of Neurology, The Catholic University of Korea, Seoul, Korea. neuronet@catholic.ac.kr
- 2Catholic Neuroscience Center, The Catholic University of Korea, Seoul, Korea.
- 3Department of Pharmacology, The Catholic University of Korea, Seoul, Korea.
- KMID: 1700685
- DOI: http://doi.org/10.3988/jcn.2007.3.1.38
Abstract
- BACKGROUND AND PURPOSE
Different mutations in the Cu/Zn superoxide dismutase 1 (SOD1) gene have been reported in approximately 10% of cases of familial amyotrophic lateral sclerosis (ALS). The aim of this study was to analyze for mutations in the SOD1 gene and clinical characteristics in Korean family of ALS.
METHODS
A subpopulation of the family reported here has been described previously. In the present study, we analyzed the SOD1 gene in the proband and his immediate family members, who were not reported on previously. Genomic DNA was isolated from the leukocytes of whole blood samples and the coding region of the SOD1 gene was analyzed by PCR and direct sequencing.
RESULTS
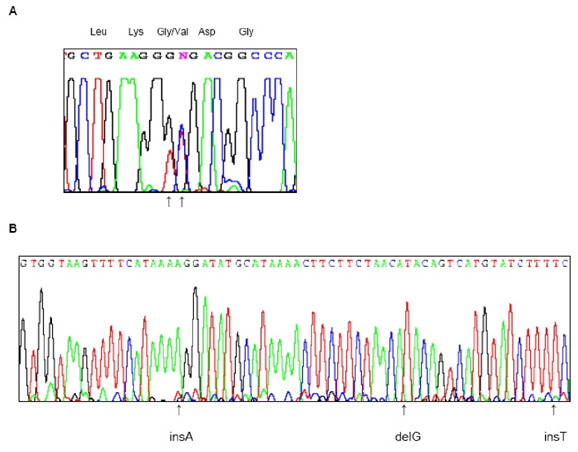
The genetic alterations were a GGC-to-GTT transition at codon 10 in exon 1 and [IVS4+15_16insA; IVS4+42delG; IVS4+59_60insT] in intron 4. Patients with these mutations exhibit diverse clinical onset symptoms and acceleration of the age at onset in successive generations, which is called anticipation.
CONCLUSIONS
We have described a family with familial ALS that showed autosomal-dominant inheritance and two distinct genetic alterations in Cu/Zn-SOD1. The affected family members had different phenotypes and anticipation.
MeSH Terms
Figure
Cited by 2 articles
-
Motor Unit Number Estimation in Evaluating Disease Progression in Patients with Amyotrophic Lateral Sclerosis
Suk-Won Ahn, Su-Hyun Kim, Dong-Hoon Oh, Sung-Min Kim, Kyung Seok Park, Yoon-Ho Hong, Oh-Sang Kwon, Jung-Joon Sung, Kwang-Woo Lee
J Korean Med Sci. 2010;25(9):1359-1363. doi: 10.3346/jkms.2010.25.9.1359.A Novel F45S SOD1 Mutation in Amyotrophic Lateral Sclerosis Coexisting with Bullous Pemphigoid
Seong-il Oh, Jeong Ho Hong, Byung Woo Choi, Ki-Wook Oh, Chan Kum Park, Min-Jung Kwon, Chang-Seok Ki, Joo Yeon Ko, Seung Hyun Kim
J Clin Neurol. 2015;11(4):390-394. doi: 10.3988/jcn.2015.11.4.390.
Reference
-
1. Rowland LP, Shneider NA. Amyotrophic lateral sclerosis. N Engl J Med. 2001. 344:1688–1700.
Article2. Mulder DW, Kurland LT, Offord KP, Beard CM. Familial adult motor neuron disease: amyotrophic lateral sclerosis. Neurology. 1986. 36:511–517.
Article3. Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature. 1993. 362:59–62.
Article4. Gros-Louis F, Gaspar C, Rouleau GA. Genetics of familial and sporadic amyotrophic lateral sclerosis. Biochim Biophys Acta. 2006. 1762:956–972.
Article5. Radunovic A, Leigh PN. Cu/Zn superoxide dismutase gene mutations in amyotrophic lateral sclerosis: correlation between genotype and clinical features. J Neurol Neurosurg Psychiatry. 1996. 61:565–572.
Article6. Nakashima K, Watanabe Y, Kuno N, Nanba E, Takahashi K. Abnormality of Cu/Zn superoxide dismutase (SOD1) activity in Japanese familial amyotrophic lateral sclerosis with two base pair deletion in the SOD1 gene. Neurology. 1995. 45:1019–1020.7. Pramatarova A, Goto J, Nanba E, Nakashima K, Takahashi K, Takagi A, et al. A two basepair deletion in the SOD 1 gene causes familial amyotrophic lateral sclerosis. Hum Mol Genet. 1994. 3:2061–2062.8. Zu JS, Deng HX, Lo TP, Mitsumoto H, Ahmed MS, Hung WY, et al. Exon 5 encoded domain is not required for the toxic function of mutant SOD1 but essential for the dismutase activity: identification and characterization of two new SOD1 mutations associated with familial amyotrophic lateral sclerosis. Neurogenetics. 1997. 1:65–71.
Article9. Andersen PM, Nilsson P, Keranen ML, Forsgren L, Hagglund J, Karlsborg M, et al. Phenotypic heterogeneity in motor neuron disease patients with CuZn-superoxide dismutase mutations in Scandinavia. Brain. 1997. 120:1723–1737.
Article10. Orrell RW, Habgood JJ, Gardiner I, King AW, Bowe FA, Hallewell RA, et al. Clinical and functional investigation of 10 missense mutations and a novel frameshift insertion mutation of the gene for copper-zinc superoxide dismutase in UK families with amyotrophic lateral sclerosis. Neurology. 1997. 48:746–751.
Article11. Sapp PC, Rosen DR, Hosler BA, Esteban J, McKenna-Yasek D, O'Regan JP, et al. Identification of three novel mutations in the gene for Cu/Zn superoxide dismutase in patients with familial amyotrophic lateral sclerosis. Neuromuscul Disord. 1995. 5:353–357.
Article12. Jackson M, Al-Chalabi A, Enayat ZE, Chioza B, Leigh PN, Morrison KE. Copper/zinc superoxide dismutase 1 and sporadic amyotrophic lateral sclerosis: analysis of 155 cases and identification of a novel insertion mutation. Ann Neurol. 1997. 42:803–807.
Article13. Hosler BA, Nicholson GA, Sapp PC, Chin W, Orrell RW, de Belleroche JS, et al. Three novel mutations and two variants in the gene for Cu/Zn superoxide dismutase in familial amyotrophic lateral sclerosis. Neuromuscul Disord. 1996. 6:361–366.
Article14. Watanabe Y, Adachi Y, Nakashima K. Japanese familial amyotrophic lateral sclerosis family with a two-base deletion in the superoxide dismutase-1 gene. Neuropathology. 2001. 21:61–66.
Article15. Kim NH, Kim HJ, Hong YH, Chun JU, Sung JJ, Kwon OY, et al. Korean familial amyotrophic lateral sclerosis family with a novel Gly10Val mutation in the SOD1 gene. J Korean Neurol Assoc. 2002. 20:379–384.16. Wilbourn AJ. Clinical neurophysiology in the diagnosis of amyotrophic lateral sclerosis: the Lambert and the El Escorial criteria. J Neurol Sci. 1998. 160:S25–S29.
Article17. Aoki M, Abe K, Houi K, Ogasawara M, Matsubara Y, Kobayashi T, et al. Variance of age at onset in a Japanese family with amyotrophic lateral sclerosis associated with a novel Cu/Zn superoxide dismutase mutation. Ann Neurol. 1995. 37:676–679.
Article18. Ceroni M, Malaspina A, Poloni TE, Alimonti D, Rognoni F, Habgood J, et al. Clustering of ALS patients in central Italy due to the occurrence of the L84F SOD1 gene mutation. Neurology. 1999. 53:1064–1071.
Article19. Iwai K, Yamamoto M, Yoshihara T, Sobue G. Anticipation in familial amyotrophic lateral sclerosis with SOD1-G93S mutation. J Neurol Neurosurg Psychiatry. 2002. 72:819–820.
Article20. Hart PJ. Pathogenic superoxide dismutase structure, folding, aggregation and turnover. Curr Opin Chem Biol. 2006. 10:131–138.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Impairment of Neurite Formation in Familial ALS-associated Cu, Zn-Superoxide Dismutase Mutant Cells
- Korean Familial Amyotrophic Lateral Sclerosis Family with a Novel Gly10Val Mutation in the SOD1 Gene
- A Novel F45S SOD1 Mutation in Amyotrophic Lateral Sclerosis Coexisting with Bullous Pemphigoid
- Extracellular Toxicity of Motor Neuronal Cells Expressing Mutant Cu/Zn Superoxide Dismutase in Familial ALS Cell Line Model
- The Effect of Calcium Modulators on Motoneuron Cells Which Express Mutated Cu / Zn Superoxide Dismutase