Korean J Physiol Pharmacol.
2013 Oct;17(5):455-461. 10.4196/kjpp.2013.17.5.455.
The Effect of Metformin Treatment on CRBP-I Level and Cancer Development in the Liver of HBx Transgenic Mice
- Affiliations
-
- 1Department of Pathology, Chonnam National University Hospital, Gwangju 501-757, Korea.
- 2Department of Medicine, Jeju National University School of Medicine, Jeju 690-756, Korea.
- 3Division of Food Bioscience, Konkuk University, Chungju 380-701, Korea.
- 4Department of Marine Life Sciences, Jeju National University, Jeju 690-756, Korea.
- 5The Disease Model Research Laboratory, Aging Research Center and World Class Institute, Korea Research Institute of Bioscience and Biotechnology, Daejeon 305-806, Korea.
- 6The Dongsuncheon Clinic, Suncheon 540-952, Korea.
- 7Department of Internal Medicine, Wonkwang University School of Medicine, Iksan 570-711, Korea. drhormone@naver.com
- KMID: 1500242
- DOI: http://doi.org/10.4196/kjpp.2013.17.5.455
Abstract
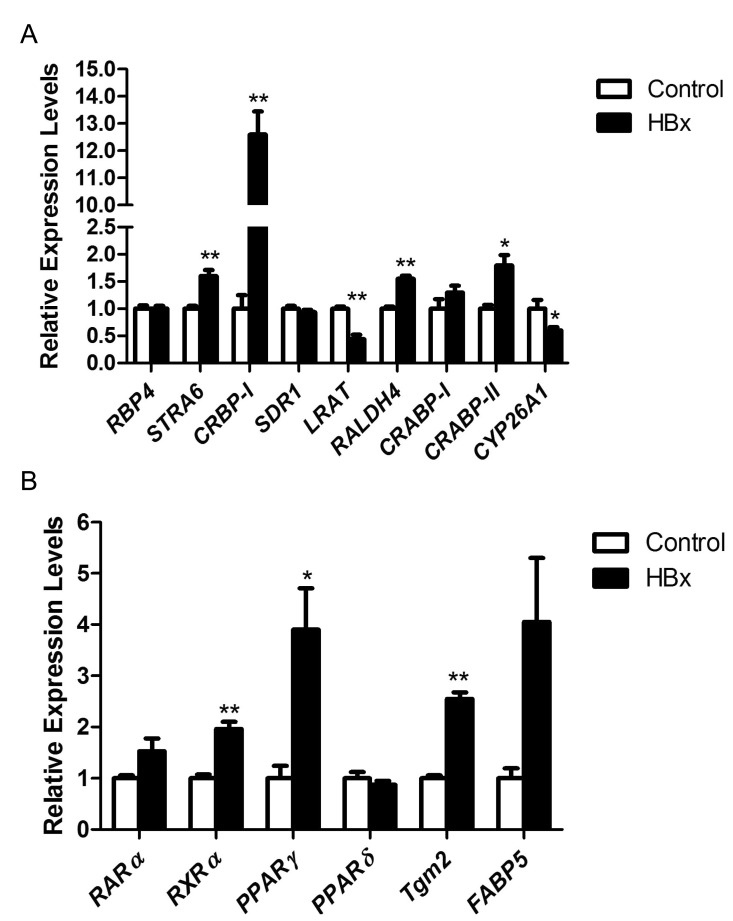
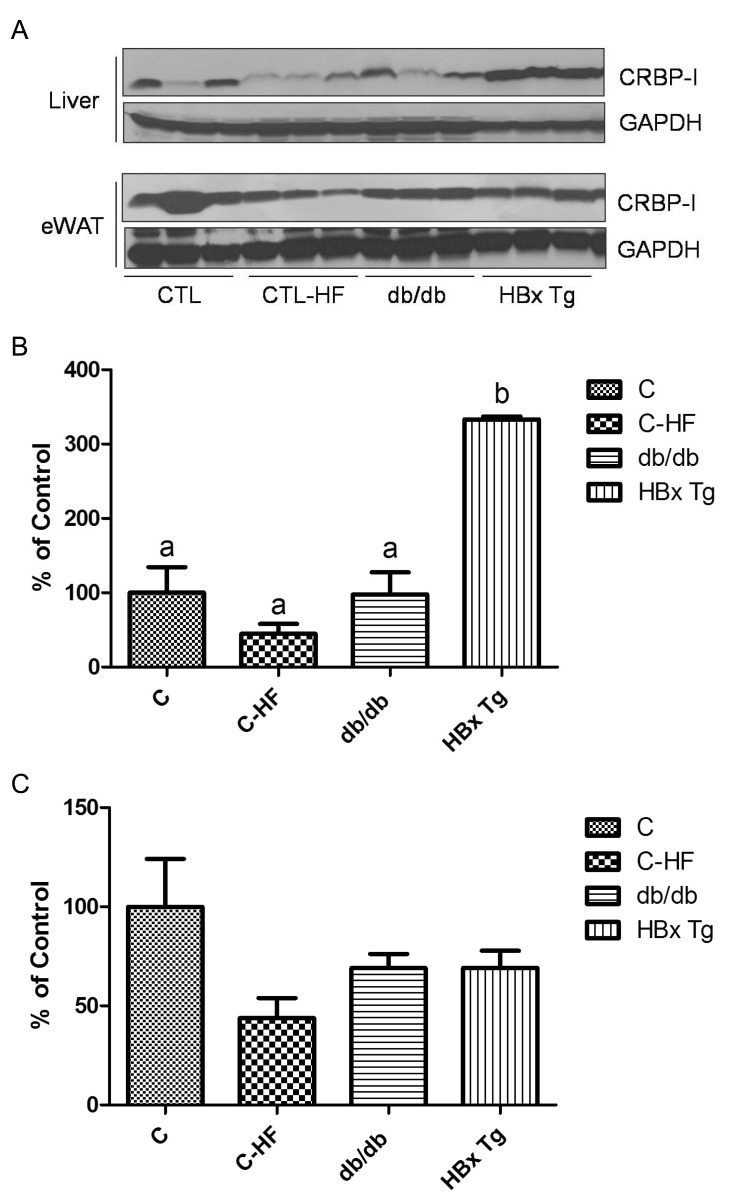
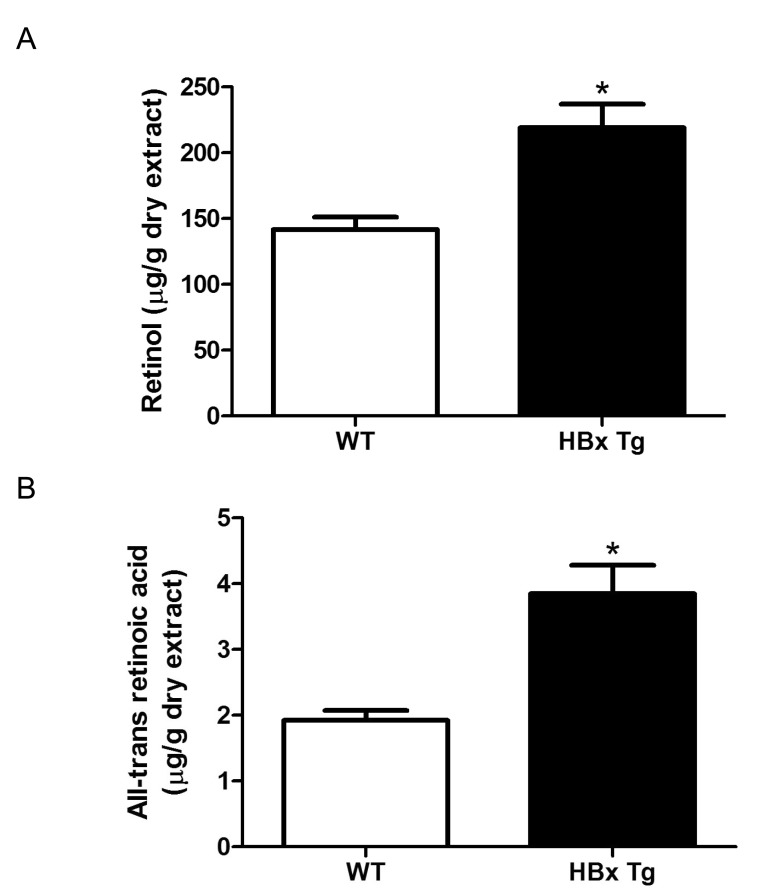
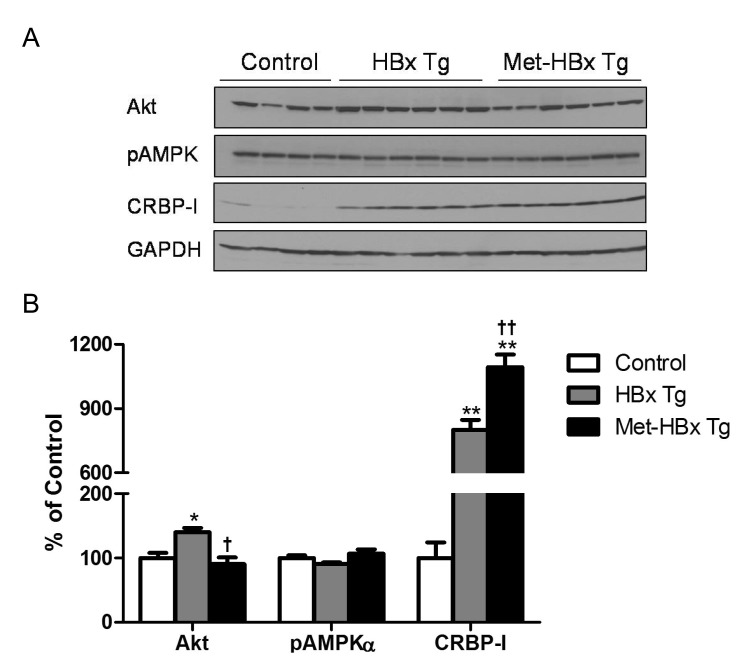
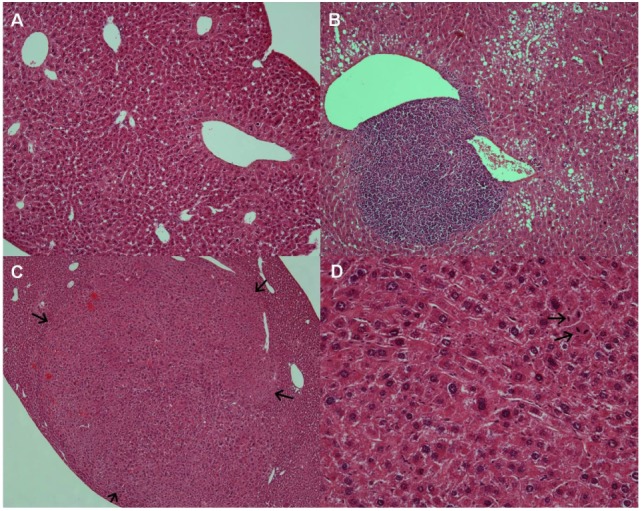
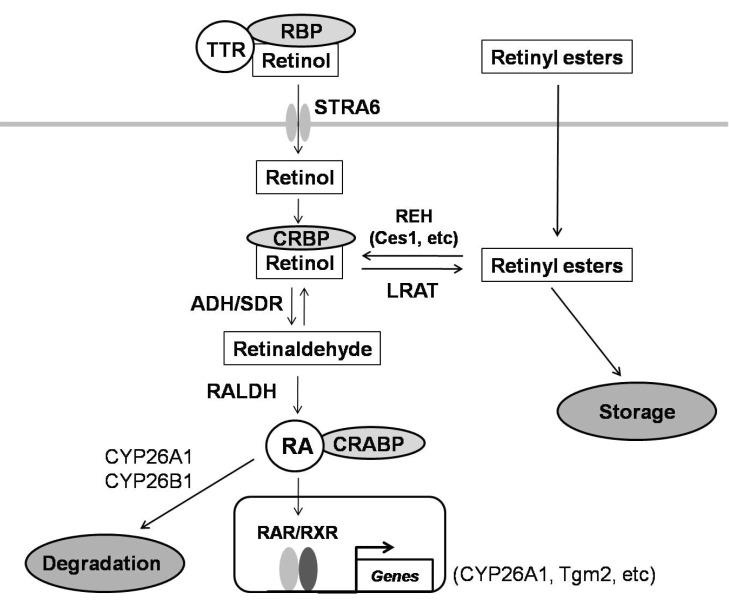
- Retinoids regulate not only various cell functions including proliferation and differentiation but also glucose and lipid metabolism. After we observed a marked up-regulation of cellular retinol-binding protein-I (CRBP-I) in the liver of hepatitis B virus x antigen (HBx)-transgenic (HBx Tg) mice which are prone to hepatocellular carcinoma (HCC) and fatty liver, we aimed to evaluate retinoid pathway, including genes for the retinoid physiology, CRBP-I protein expression, and retinoid levels, in the liver of HBx Tg mice. We also assessed the effect of chronic metformin treatment on HCC development in the mice. Many genes involved in hepatic retinoid physiology, including CRBP-I, were altered and the tissue levels of retinol and all-trans retinoic acid (ATRA) were elevated in the liver of HBx Tg mice compared to those of wild type (WT) control mice. CRBP-I protein expression in liver, but not in white adipose tissue, of HBx Tg mice was significantly elevated compared to WT control mice while CRBP-I protein expressions in the liver and WAT of high-fat fed obese and db/db mice were comparable to WT control mice. Chronic treatment of HBx Tg mice with metformin did not affect the incidence of HCC, but slightly increased hepatic CRBP-I level. In conclusion, hepatic CRBP-I level was markedly up-regulated in HCC-prone HBx Tg mice and neither hepatic CRBP-I nor the development of HCC was suppressed by metformin treatment.
Keyword
MeSH Terms
Figure
Reference
-
1. Custro N, Carroccio A, Ganci A, Scafidi V, Campagna P, Di Prima L, Montalto G. Glycemic homeostasis in chronic viral hepatitis and liver cirrhosis. Diabetes Metab. 2001; 27:476–481. PMID: 11547221.2. Shin HJ, Park YH, Kim SU, Moon HB, Park do S, Han YH, Lee CH, Lee DS, Song IS, Lee DH, Kim M, Kim NS, Kim DG, Kim JM, Kim SK, Kim YN, Kim SS, Choi CS, Kim YB, Yu DY. Hepatitis B virus X protein regulates hepatic glucose homeostasis via activation of inducible nitric oxide synthase. J Biol Chem. 2011; 286:29872–29881. PMID: 21690090.
Article3. Kim KH, Shin HJ, Kim K, Choi HM, Rhee SH, Moon HB, Kim HH, Yang US, Yu DY, Cheong J. Hepatitis B virus X protein induces hepatic steatosis via transcriptional activation of SREBP1 and PPARgamma. Gastroenterology. 2007; 132:1955–1967. PMID: 17484888.4. Kew MC. Hepatitis B virus x protein in the pathogenesis of hepatitis B virus-induced hepatocellular carcinoma. J Gastroenterol Hepatol. 2011; 26(Suppl 1):144–152. PMID: 21199526.
Article5. Yu DY, Moon HB, Son JK, Jeong S, Yu SL, Yoon H, Han YM, Lee CS, Park JS, Lee CH, Hyun BH, Murakami S, Lee KK. Incidence of hepatocellular carcinoma in transgenic mice expressing the hepatitis B virus X-protein. J Hepatol. 1999; 31:123–132. PMID: 10424292.
Article6. Starley BQ, Calcagno CJ, Harrison SA. Nonalcoholic fatty liver disease and hepatocellular carcinoma: a weighty connection. Hepatology. 2010; 51:1820–1832. PMID: 20432259.
Article7. Altucci L, Leibowitz MD, Ogilvie KM, de Lera AR, Gronemeyer H. RAR and RXR modulation in cancer and metabolic disease. Nat Rev Drug Discov. 2007; 6:793–810. PMID: 17906642.
Article8. Kluwe J, Wongsiriroj N, Troeger JS, Gwak GY, Dapito DH, Pradere JP, Jiang H, Siddiqi M, Piantedosi R, O'Byrne SM, Blaner WS, Schwabe RF. Absence of hepatic stellate cell retinoid lipid droplets does not enhance hepatic fibrosis but decreases hepatic carcinogenesis. Gut. 2011; 60:1260–1268. PMID: 21278145.
Article9. Yoon JS, Lee MY, Lee JS, Park CS, Youn HJ, Lee JH. Bis is involved in glial differentiation of p19 cells induced by retinoic Acid. Korean J Physiol Pharmacol. 2009; 13:251–256. PMID: 19885044.
Article10. Duong V, Rochette-Egly C. The molecular physiology of nuclear retinoic acid receptors. From health to disease. Biochim Biophys Acta. 2011; 1812:1023–1031. PMID: 20970498.
Article11. Noy N. Retinoid-binding proteins: mediators of retinoid action. Biochem J. 2000; 348(Pt 3):481–495. PMID: 10839978.
Article12. Zizola CF, Frey SK, Jitngarmkusol S, Kadereit B, Yan N, Vogel S. Cellular retinol-binding protein type I (CRBP-I) regulates adipogenesis. Mol Cell Biol. 2010; 30:3412–3420. PMID: 20498279.
Article13. Ashla AA, Hoshikawa Y, Tsuchiya H, Hashiguchi K, Enjoji M, Nakamuta M, Taketomi A, Maehara Y, Shomori K, Kurimasa A, Hisatome I, Ito H, Shiota G. Genetic analysis of expression profile involved in retinoid metabolism in non-alcoholic fatty liver disease. Hepatol Res. 2010; 40:594–604. PMID: 20618457.
Article14. Bonet ML, Ribot J, Palou A. Lipid metabolism in mammalian tissues and its control by retinoic acid. Biochim Biophys Acta. 2012; 1821:177–189. PMID: 21669299.
Article15. Ziouzenkova O, Orasanu G, Sharlach M, Akiyama TE, Berger JP, Viereck J, Hamilton JA, Tang G, Dolnikowski GG, Vogel S, Duester G, Plutzky J. Retinaldehyde represses adipogenesis and diet-induced obesity. Nat Med. 2007; 13:695–702. PMID: 17529981.
Article16. Chen HP, Shieh JJ, Chang CC, Chen TT, Lin JT, Wu MS, Lin JH, Wu CY. Metformin decreases hepatocellular carcinoma risk in a dose-dependent manner: population-based and in vitro studies. Gut. 2013; 62:606–615. PMID: 22773548.17. Zhang ZJ, Zheng ZJ, Shi R, Su Q, Jiang Q, Kip KE. Metformin for liver cancer prevention in patients with type 2 diabetes: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2012; 97:2347–2353. PMID: 22523334.
Article18. Kang W, Hong HJ, Guan J, Kim DG, Yang EJ, Koh G, Park D, Han CH, Lee YJ, Lee DH. Resveratrol improves insulin signaling in a tissue-specific manner under insulin-resistant conditions only: in vitro and in vivo experiments in rodents. Metabolism. 2012; 61:424–433. PMID: 21945106.19. Ribaya-Mercado JD, Holmgren SC, Fox JG, Russell RM. Dietary beta-carotene absorption and metabolism in ferrets and rats. J Nutr. 1989; 119:665–668. PMID: 2703921.20. Green MH, Snyder RW, Akohoue SA, Green JB. Increased rat mammary tissue vitamin A associated with increased vitamin A intake during lactation is maintained after lactation. J Nutr. 2001; 131:1544–1547. PMID: 11340113.21. Tang GW, Russell RM. 13-cis-retinoic acid is an endogenous compound in human serum. J Lipid Res. 1990; 31:175–182. PMID: 2324641.
Article22. Berry DC, Noy N. All-trans-retinoic acid represses obesity and insulin resistance by activating both peroxisome proliferation-activated receptor beta/delta and retinoic acid receptor. Mol Cell Biol. 2009; 29:3286–3296. PMID: 19364826.23. Muto Y, Omori M. A novel cellular retinoid-binding protein, F-type, in hepatocellular carcinoma. Ann N Y Acad Sci. 1981; 359:91–103. PMID: 6266314.
Article24. Dong D, Ruuska SE, Levinthal DJ, Noy N. Distinct roles for cellular retinoic acid-binding proteins I and II in regulating signaling by retinoic acid. J Biol Chem. 1999; 274:23695–23698. PMID: 10446126.
Article25. Jing Y, Waxman S, Mira-y-Lopez R. The cellular retinoic acid binding protein II is a positive regulator of retinoic acid signaling in breast cancer cells. Cancer Res. 1997; 57:1668–1672. PMID: 9135005.26. Theodosiou M, Laudet V, Schubert M. From carrot to clinic: an overview of the retinoic acid signaling pathway. Cell Mol Life Sci. 2010; 67:1423–1445. PMID: 20140749.
Article27. Sporn MB, Squire RA, Brown CC, Smith JM, Wenk ML, Springer S. 13-cis-retinoic acid: inhibition of bladder carcinogenesis in the rat. Science. 1977; 195:487–489. PMID: 835006.28. Hill DL, Grubbs CJ. Retinoids and cancer prevention. Annu Rev Nutr. 1992; 12:161–181. PMID: 1503802.
Article29. Muto Y, Moriwaki H, Ninomiya M, Adachi S, Saito A, Takasaki KT, Tanaka T, Tsurumi K, Okuno M, Tomita E, Nakamura T, Kojima T. Hepatoma Prevention Study Group. Prevention of second primary tumors by an acyclic retinoid, polyprenoic acid, in patients with hepatocellular carcinoma. N Engl J Med. 1996; 334:1561–1567. PMID: 8628336.
Article30. Sauvant P, Cansell M, Atgie C. Vitamin A and lipid metabolism: relationship between hepatic stellate cells (HSCs) and adipocytes. J Physiol Biochem. 2011; 67:487–496. PMID: 21626400.
Article31. O'Byrne SM, Wongsiriroj N, Libien J, Vogel S, Goldberg IJ, Baehr W, Palczewski K, Blaner WS. Retinoid absorption and storage is impaired in mice lacking lecithin:retinol acyltransferase (LRAT). J Biol Chem. 2005; 280:35647–35657. PMID: 16115871.32. Liu L, Gudas LJ. Disruption of the lecithin:retinol acyltransferase gene makes mice more susceptible to vitamin A deficiency. J Biol Chem. 2005; 280:40226–40234. PMID: 16174770.
Article33. McCormick DL, Hollister JL, Bagg BJ, Long RE. Enhancement of murine hepatocarcinogenesis by all-trans-retinoic acid and two synthetic retinamides. Carcinogenesis. 1990; 11:1605–1609. PMID: 2144795.
Article34. El-Serag HB, Tran T, Everhart JE. Diabetes increases the risk of chronic liver disease and hepatocellular carcinoma. Gastroenterology. 2004; 126:460–468. PMID: 14762783.
Article35. Muto Y, Omori M, Sugawara K. Demonstration of a novel cellular retinol-binding protein, F-type, in hepatocellular carcinoma. Gann. 1979; 70:215–222. PMID: 89058.36. Napoli JL. Interactions of retinoid binding proteins and enzymes in retinoid metabolism. Biochim Biophys Acta. 1999; 1440:139–162. PMID: 10521699.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relevance of reactive oxygen species in liver disease observed in transgenic mice expressing the hepatitis B virus X protein
- Pathologic Findings in the Liver of Hepatitis B Virus X Transgenic Mice
- A Study on the Expression of Proliferating Cell Nuclear Antigen and Apoptosis of the Hepatocellular Carcinoma in Human and Hepatitis B Virus X Transgenic Mice
- Hepatic STAMP2 decreases hepatitis B virus X protein-associated metabolic deregulation
- Usefulness of High-Resolution Sonography for Assessement of Hepatocellular Carcinoma in the Transgenic Mice Expressing Hepatitis B Virus X-protein: A Preliminary Study