Korean J Physiol Pharmacol.
2012 Oct;16(5):297-303. 10.4196/kjpp.2012.16.5.297.
High K(+)-Induced Relaxation by Nitric Oxide in Human Gastric Fundus
- Affiliations
-
- 1Department of Surgery, Chungbuk National University College of Medicine, Cheongju 361-763, Korea. yunhyo@chungbuk.ac.kr
- 2Department of Physiology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea. physiokyc@chungbuk.ac.kr
- 3Department of Pharmacology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 4Department of Pathology, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 5Department of Preventing Medicine, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 6Department of Internal Medicine, Chungbuk National University College of Medicine, Cheongju 361-763, Korea.
- 7Department of Physiology, College of Medcine, Shanghai Jiaotong University, Shanghai 200240, China.
- KMID: 1493961
- DOI: http://doi.org/10.4196/kjpp.2012.16.5.297
Abstract
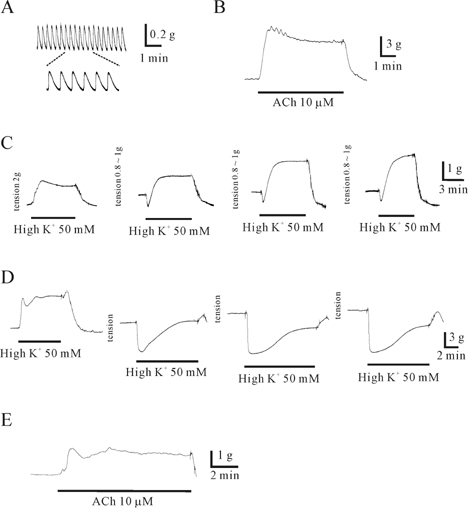
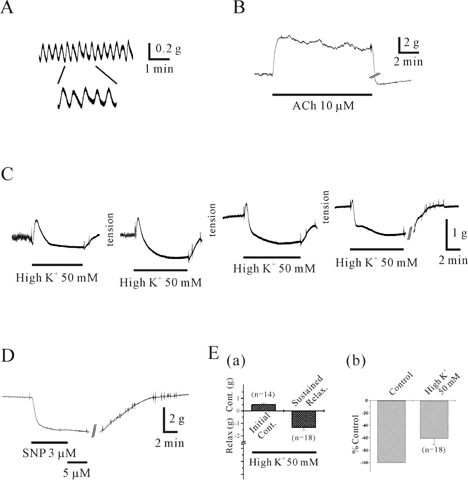
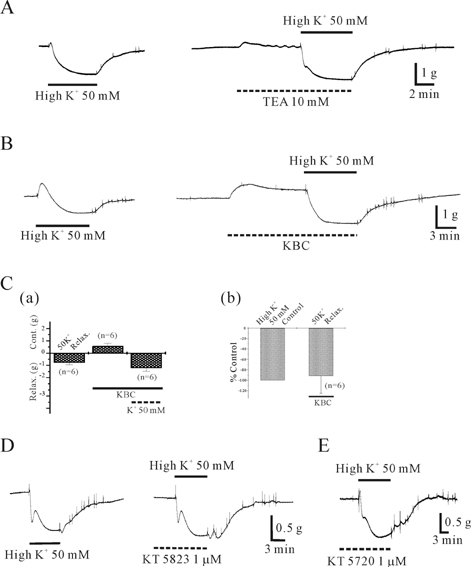
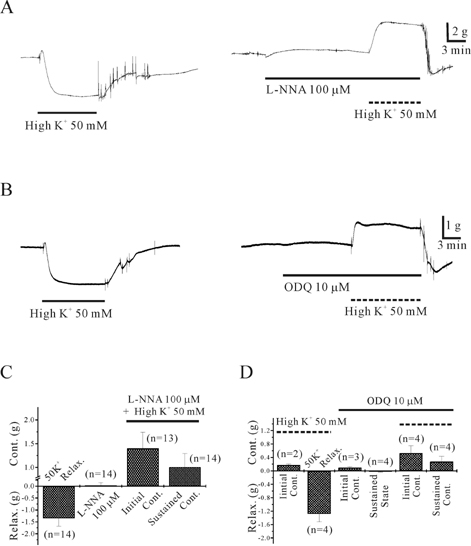
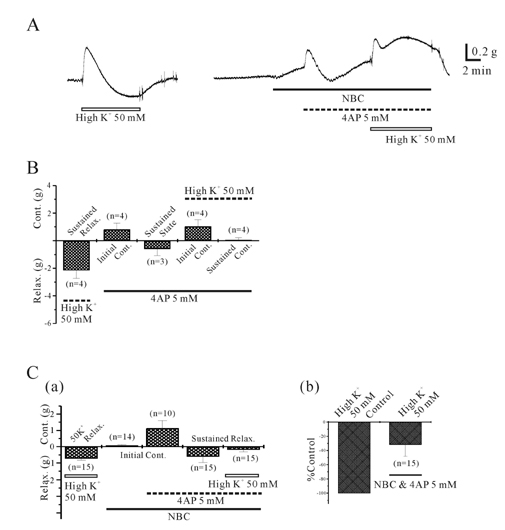
- This study was designed to elucidate high K(+)-induced relaxation in the human gastric fundus. Circular smooth muscle from the human gastric fundus greater curvature showed stretch-dependent high K+ (50 mM)-induced contractions. However, longitudinal smooth muscle produced stretch-dependent high K(+)-induced relaxation. We investigated several relaxation mechanisms to understand the reason for the discrepancy. Protein kinase inhibitors such as KT 5823 (1 microM) and KT 5720 (1 microM) which block protein kinases (PKG and PKA) had no effect on high K(+)-induced relaxation. K+ channel blockers except 4-aminopyridine (4-AP), a voltage-dependent K+ channel (KV) blocker, did not affect high K(+)-induced relaxation. However, N(G)-nitro-L-arginine and 1H-(1,2,4)oxadiazolo (4,3-A)quinoxalin-1-one, an inhibitors of soluble guanylate cyclase (sGC) and 4-AP inhibited relaxation and reversed relaxation to contraction. High K(+)-induced relaxation of the human gastric fundus was observed only in the longitudinal muscles from the greater curvature. These data suggest that the longitudinal muscle of the human gastric fundus greater curvature produced high K(+)-induced relaxation that was activated by the nitric oxide/sGC pathway through a KV channel-dependent mechanism.
MeSH Terms
Figure
Reference
-
1. Cannon WB, Lieb CW. The receptive relaxation of the stomach. Am J Physiol. 1911. 29:267–273.2. Paton WD, Vane JR. Analysis of the responses of the isolated stomach to electrical stimulation and to drugs. J Physiol. 1963. 165:10–46.3. Hennig GW, Brookes SJ, Costa M. Excitatory and inhibitory motor reflexes in the isolated guinea-pig stomach. J Physiol. 1997. 501:197–212.4. Desai KM, Sessa WC, Vane JR. Involvement of nitric oxide in the reflex relaxation of the stomach to accommodate food or fluid. Nature. 1991. 351:477–479.5. Abrahamsson H. Studies on the inhibitory nervous control of gastric motility. Acta Physiol Scand Suppl. 1973. 390:1–38.6. Barbier AJ, Lefebvre RA. Involvement of the L-arginine: nitric oxide pathway in nonadrenergic noncholinergic relaxation of the cat gastric fundus. J Pharmacol Exp Ther. 1993. 266:172–178.7. Lefebvre RA, Smits GJ, Timmermans JP. Study of NO and VIP as non-adrenergic non-cholinergic neurotransmitters in the pig gastric fundus. Br J Pharmacol. 1995. 116:2017–2026.8. Meulemans AL, Helsen LF, Schuurkes JA. Role of NO in vagally-mediated relaxations of guinea-pig stomach. Naunyn Schmiedebergs Arch Pharmacol. 1993. 347:225–230.9. Yano S, Kiyota Y, Yamamoto M, Watanabe K. Pharmacological features of non-adrenergic non-cholinergic (NANC) relaxation induced by electrical vagal stimulation in isolated mouse stomach. Jpn J Pharmacol. 1995. 69:9–15.10. Tonini M, De Giorgio R, De Ponti F, Sternini C, Spelta V, Dionigi P, Barbara G, Stanghellini V, Corinaldesi R. Role of nitric oxide- and vasoactive intestinal polypeptide-containing neurones in human gastric fundus strip relaxations. Br J Pharmacol. 2000. 129:12–20.11. Kim SJ, Ahn SC, Kim JK, Kim YC, So I, Kim KW. Changes in intracellular Ca2+ concentration induced by L-type Ca2+ channel current in guinea pig gastric myocytes. Am J Physiol. 1997. 273:C1947–C1956.12. Kuriyama H, Mishima K, Suzuki H. Some differences in contractile responses of isolated longitudinal and circular muscle from the guinea-pig stomach. J Physiol. 1975. 251:317–331.13. Kim YC, Choi W, Yun HY, Sung R, Yoo RY, Park SM, Yun SJ, Kim MJ, Song YJ, Xu WX, Lee SJ. Nitric oxide-mediated relaxation by high K in human gastric longitudinal smooth muscle. Korean J Physiol Pharmacol. 2011. 15:405–413.14. Schubert ML, Makhlouf GM. Gastrin secretion induced by distention is mediated by gastric cholinergic and vasoactive intestinal peptide neurons in rats. Gastroenterology. 1993. 104:834–839.15. Shafik A, El Sibai O, Shafik AA, Shafik IA. Mechanism of gastric emptying through the pyloric sphincter: a human study. Med Sci Monit. 2007. 13:CR24–CR29.16. Burnstock G. The non-adrenergic non-cholinergic nervous system. Arch Int Pharmacodyn Ther. 1986. 280:2 Suppl. 1–15.17. Derici K, Samsar U, Demirel-Yilmaz E. Nitric oxide effects depend on different mechanisms in different regions of the rat heart. Heart Vessels. 2012. 27:89–97.18. Dixit D, Zarate N, Liu LW, Boreham DR, Huizinga JD. Interstitial cells of Cajal and adaptive relaxation in the mouse stomach. Am J Physiol Gastrointest Liver Physiol. 2006. 291:G1129–G1136.19. Sung R, Kim YC, Yun HY, Choi W, Kim HS, Kim H, Lee KJ, You RY, Park SM, Youn SJ, Kim MJ, Kim WS, Song YJ, Kim SY, Xu WX, Lee SJ. Interstitial cells of Cajal (ICC)-like-c-Kit positive cells are involved in gastritis and carcinogenesis in human stomach. Oncol Rep. 2011. 26:33–42.20. Yun HY, Sung R, Kim YC, Choi W, Kim HS, Kim H, Lee GJ, You RY, Park SM, Yun SJ, Kim MJ, Kim WS, Song YJ, Xu WX, Lee SJ. Regional distribution of interstitial cells of Cajal (ICC) in Human Stomach. Korean J Physiol Pharmacol. 2010. 14:317–324.21. Cousins HM, Edwards FR, Hickey H, Hill CE, Hirst GD. Electrical coupling between the myenteric interstitial cells of Cajal and adjacent muscle layers in the guinea-pig gastric antrum. J Physiol. 2003. 550:829–844.22. Osa T, Kuriyama H. The membrane properties and decremental conduction of excitation in the fundus of the guinea-pig stomach. Jpn J Physiol. 1970. 20:626–639.23. Connor C, Prosser CL. Comparison of ionic effects on longitudinal and circular muscle of cat jejunum. Am J Physiol. 1974. 226:1212–1218.24. Somlyo AP, Somlyo AV. Signal transduction and regulation in smooth muscle. Nature. 1994. 372:231–236.25. Hagiwara S, Byerly L. Membrane biophysics of calcium currents. Fed Proc. 1981. 40:2220–2225.26. Barajas-López C, Huizinga JD. Different mechanisms of contraction generation in circular muscle of canine colon. Am J Physiol. 1989. 256:G570–G580.27. Kelly KA, Code CF. Canine gastric pacemaker. Am J Physiol. 1971. 220:112–118.28. Xu WX, Kim SJ, So I, Kim KW. Role of actin microfilament in osmotic stretch-induced increase of voltage-operated calcium channel current in guinea-pig gastric myocytes. Pflugers Arch. 1997. 434:502–504.29. Xu WX, Li Y, Wu LR, Li ZL. Effects of different kinds of stretch on voltage-dependent calcium current in antrial circular smooth muscle cells of the guinea-pig. Sheng Li Xue Bao. 2000. 52:69–74.30. Yuan XJ, Tod ML, Rubin LJ, Blaustein MP. NO hyperpolarizes pulmonary artery smooth muscle cells and decreases the intracellular Ca2+ concentration by activating voltage-gated K+ channels. Proc Natl Acad Sci USA. 1996. 93:10489–10494.31. Yu YC, Guo HS, Li Y, Piao L, Li L, Li ZL, Xu WX. Role of calcium mobilization in sodium nitroprusside-induced increase of calcium-activated potassium currents in gastric antral circular myocytes of guinea pig. Acta Pharmacol Sin. 2003. 24:819–825.32. Ibba Manneschi L, Pacini S, Corsani L, Bechi P, Faussone-Pellegrini MS. Interstitital cells of Cajal in the human stomach: distribution and relationship with enteric innervation. Histol Histopathol. 2004. 19:1153–1164.33. Iino S, Horiguchi K, Nojyo Y. Interstitial cells of Cajal are innervated by nitrergic nerves and express nitric oxide-sensitive guanylate cyclase in the guinea-pig gastrointestinal tract. Neuroscience. 2008. 152:437–448.34. Nemeth L, Puri P. Three-dimensional morphology of c-Kit-positive cellular network and nitrergic innervation in the human gut. Arch Pathol Lab Med. 2001. 125:899–904.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effects of Nitric Oxide and Vasoactive Intestinal Peptide on the Smooth Muscle Relaxation of a Guinea Pig's Gastric Fundus
- Impaired endothelium-dependent relaxation is mediated by reduced production of nitric oxide in the streptozotocin-induced diabetic rats
- Role of 5-Hydroxytryptamine in Non-Adrenergic Non-cholinergic Relaxation of the Circcular Muscle from Rat Stomach Fundus
- Expression of Constitutive Nitric Oxide Synthase by Gastrointestinal Epithelial Cells
- Protective mechanism of nitric oxide and mucus against ischemia/reperfusion-induced gastric mucosal injury