Korean J Ophthalmol.
2008 Jun;22(2):123-129. 10.3341/kjo.2008.22.2.123.
Short-term Efficacy of Topical Immunosuppressive Agents on the Survival of Cultivated Allo-Conjunctival Equivalents
- Affiliations
-
- 1Department of Ophthalmology, Dongguk University College of Medicine, Goyang, Korea.
- 2Department of Ophthalmology, Seoul National University College of Medicine, Seoul, Korea. kmk9@snu.ac.kr
- 3Artificial Eye Center, Clinical Research institute Seoul National University Hospital, Seoul, Korea.
- 4Valued eye clinic, Taegeon, Korea.
- 5Woori eye clinic, Taegeon, Korea.
- 6Korea Eye Center Hospital, Seoul, Korea.
- KMID: 1099007
- DOI: http://doi.org/10.3341/kjo.2008.22.2.123
Abstract
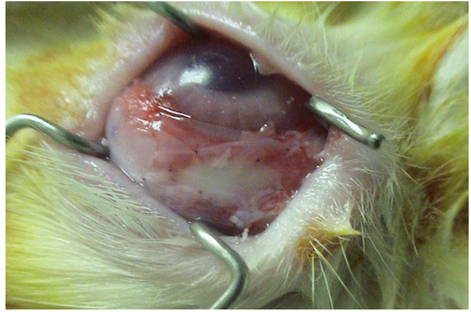
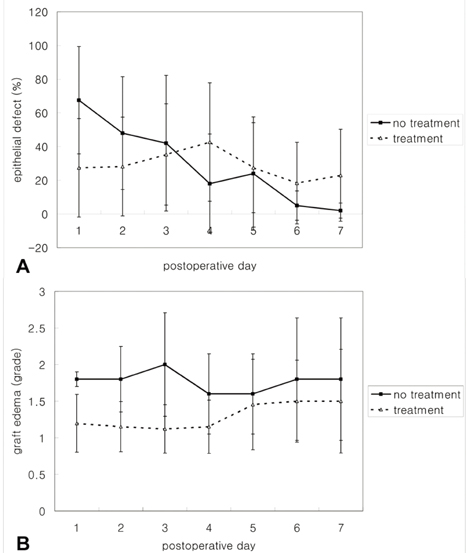
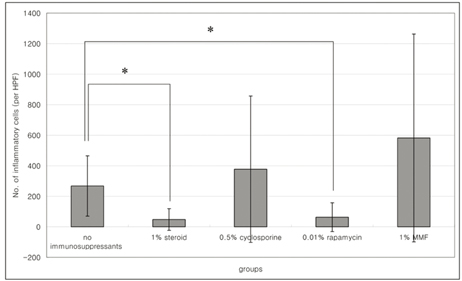
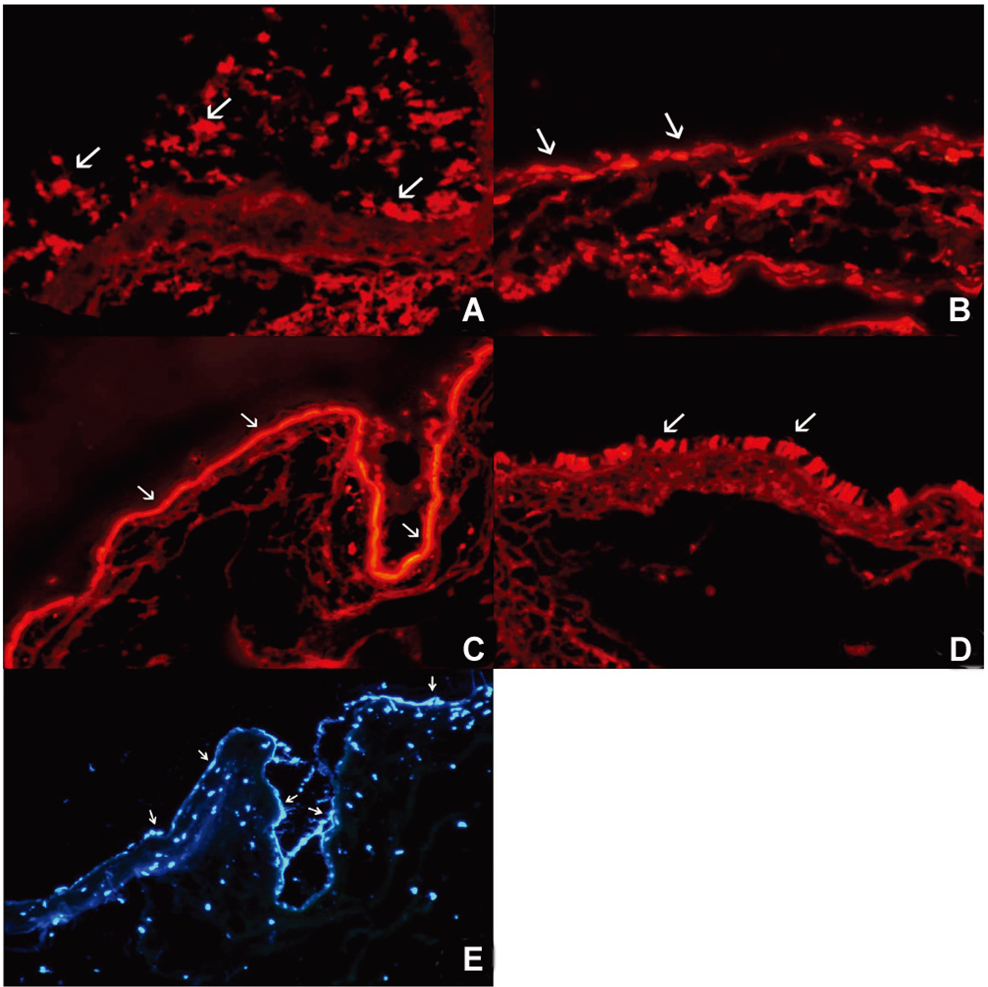
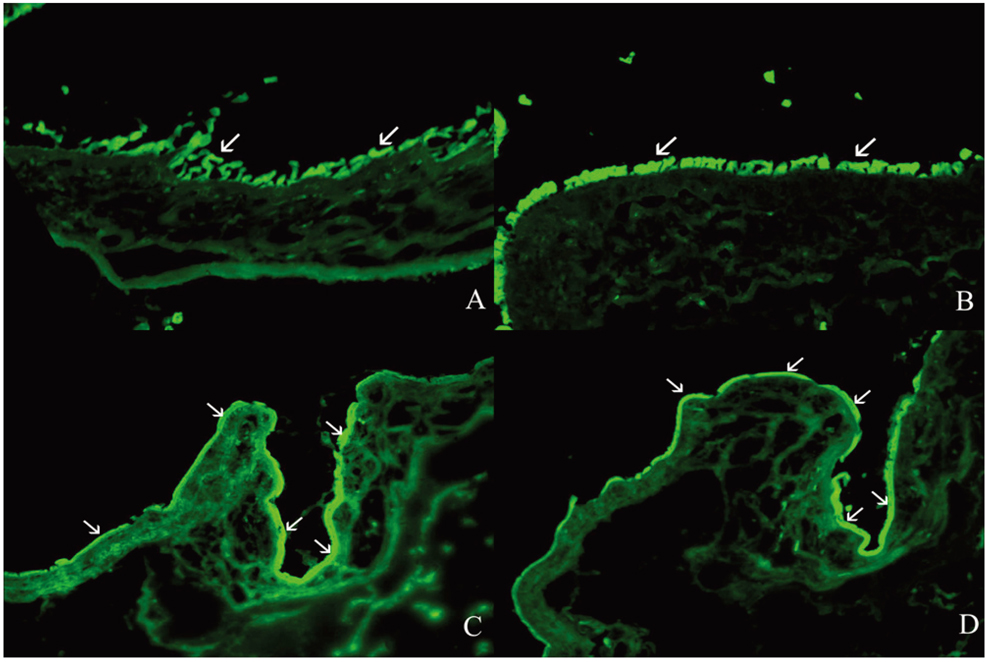
- PURPOSE: To investigate the short-term efficacy of topical immunosuppressive agents on the survival of cultivated allo-conjunctival equivalents. METHODS: Twenty-five eyes of New Zealand white rabbits were included. Temporal conjunctivae were trephined to a diameter of 7.5 mm, and then cultured allo-conjunctival epithelial cells on amniotic membrane were transplanted onto them. Various immunosuppressants including steroid, cyclosporine, and rapamycin were applied topically four times a day for a week. Epithelial defects and graft edema were graded daily. Numbers of inflammatory cells were measured in H&E. PKH26 and cytokeratin 4 and 7 were immunostained. RESULTS: Earlier epithelialization was observed in 1% steroid-treated eyes and defects persisted significantly in 0.5% CsA applied eyes. In histology, PKH26 positive cells considered as donor cells were only found in 1% steroid or 0.01% rapamycin applied eyes. 1% steroid- or 0.01% rapamycin-applied eyes both showed positive staining for keratin-4 and -7. Inflammatory cells were less found in 1% steroid or 0.01% rapamycin treated eyes. CONCLUSIONS: Topical steroid or rapamycin can help to suppress acute inflammation and enhance the acute survival of transplanted conjunctival cells.
MeSH Terms
-
Administration, Topical
Animals
Cell Count
*Cell Transplantation
Cells, Cultured
Conjunctiva/*cytology
Cyclosporine/pharmacology
Epithelial Cells/metabolism/*transplantation
Female
Fluorescent Antibody Technique, Indirect
Graft Survival/*drug effects
Immunosuppressive Agents/*pharmacology
Keratin-4/metabolism
Keratin-7/metabolism
Male
Organic Chemicals/metabolism
Prednisone/pharmacology
Rabbits
Sirolimus/pharmacology
Transplantation, Homologous
Figure
Reference
-
1. Thoft RA. Conjunctival transplantation. Arch Ophthalmol. 1977. 95:1425–1427.2. Kwitko S, Marinho D, Barcaro S, et al. Allograft conjunctival transplantation for bilateral ocular surface disorders. Ophthalmology. 1995. 102:1020–1025.3. Järplid B, Schantz B. Rejection of skin and conjunctival allografts in the dog after prolonged graft survival. Acta Chir Scand. 1973. 139:517–521.4. Obel N, Schantz B. Transplantation of allogeneic conjunctiva as a complement to allogeneic skin grafting when studying immunosuppressive procedures in the dog. Acta Chir Scand. 1972. 138:21–24.5. Nussenblatt RB, Whitcup SM, Palestine AG. Uveiti. 1996. 2nd ed. St.Louis: Mosby;106–122.6. Dios E, Herreras JM, Mayo A, Blanco G. Efficacy of systemic cyclosporine A and amniotic membrane on rabbit conjunctival limbal allograft rejection. Cornea. 2005. 24:182–188.7. Kwon YS, Kim JC. Inhibition of corneal neovascularization by rapamycin. Exp Mol Med. 2006. 38:173–179.8. Kwon YS, Hong HS, Kim JC, et al. Inhibitory effect of rapamycin on corneal neovascularization in vitro and in vivo. Invest Ophthalmol Vis Sci. 2005. 46:454–460.9. Ebrahem Q, Minamoto A, Hoppe G, et al. Triamcinolone acetonide inhibits IL-6- and VEGF-induced angiogenesis downstream of the IL-6 and VEGF receptors. Invest Ophthalmol Vis Sci. 2006. 47:4935–4941.10. Benelli U, Ross JR, Nardi M, Klintworth GK. Corneal neovascularization induced by xenografts or chemical cautery. Inhibition by cyclosporin A. Invest Ophthalmol Vis Sci. 1997. 38:274–282.11. De Saint Jean M, Baudouin C, Di Nolfo M, et al. Comparison of morphological and functional characteristics of primary-cultured human conjunctival epithelium and of Wong-Kilbourne derivative of Chang conjunctival cell line. Exp Eye Res. 2004. 78:257–274.12. Bell TA, Hunnisett AG. Cyclosporin A: tissue levels following topical and systemic administration to rabbits. Br J Ophthalmol. 1986. 70:852–855.13. Wiederholt M, Kossendrup D, Schulz W, Hoffmann F. Pharmacokinetic of topical cyclosporin A in the rabbit eye. Invest Ophthalmol Vis Sci. 1986. 27:519–524.14. Kaswan RL. Intraocular penetration of topically applied cyclosporine. Transplant Proc. 1988. 20:650–655.15. Stamer L, Bohnke M, Vogelberg K, Arndt R. Tissue levels of locally applied cyclosporin A in the rabbit eye. Fortschr Ophthalmol. 1989. 86:540–542.16. Mosteller MW, Gebhardt BM, Hamilton AM, Kaufman HE. Penetration of topical cyclosporine into the rabbit cornea, aqueous humor, and serum. Arch Ophthalmol. 1985. 103:101–102.17. Okada K, Sakata H, Minamoto A, Fujihara M. Effect of FK 506 administered topically versus intramuscularly on suppression of the corneal immune reaction in rats. Ophthalmologica. 1996. 210:175–179.18. Benelli U, Lepri A, Del Tacca M, Nardi M. FK-506 delays corneal graft rejection in a model of corneal xenotransplantation. J Ocul Pharmacol Ther. 1996. 12:425–431.19. Hikita N, Chan CC, Whitcup SM, et al. Effects of topical FK506 on endotoxin-induced uveitis (EIU) in the Lewis rat. Curr Eye Res. 1995. 14:209–214.20. Hikita N, Lopez JS, Chan CC, et al. Use of topical FK506 in a corneal graft rejection model in Lewis rats. Invest Ophthalmol Vis Sci. 1997. 38:901–909.21. Ang LP, Tan DT, Beuerman RW, Lavker RM. Development of a conjunctival epithelial equivalent with improved proliferative properties using a multistep serumfree culture system. Invest Ophthalmol Vis Sci. 2004. 45:1789–1795.22. Ang LP, Tan DT, Phan TT, et al. The in vitro and in vivo proliferative capacity of serum-free cultivated human conjunctival epithelial cells. Curr Eye Res. 2004. 28:307–317.23. Meller D, Tseng SC. Conjunctival epithelial cell differentiation on amniotic membrane. Invest Ophthalmol Vis Sci. 1999. 40:878–886.24. Ang LP, Cheng ZY, Beuerman RW, et al. The development of a serum-free derived bioengineered conjunctival epithelial equivalent using an ultrathin poly (epsilon-caprolactone) membrane substrate. Invest Ophthalmol Vis Sci. 2006. 47:105–112.25. Ang LP, Tan DT. Autologous cultivated conjunctival transplantation for recurrent viral papillomata. Am J Ophthalmol. 2005. 140:136–138.26. Nishida K, Yamato M, Hayashida Y, et al. Functional bioengineered corneal epithelial sheet grafts from corneal stem cells expanded ex vivo on a temperature-responsive cell culture surface. Transplantation. 2004. 77:379–385.27. Adinolfi M, Akle CA, McColl I, et al. Expression of HLA antigens, β2-microglobulin and enzymes by human amniotic epithelial cells. Nature. 1982. 295:325–327.28. Akle CA, Adinolfi M, Welsh KI, et al. Immunogenicity of human amniotic epithelial cells after transplantation into volunteers. Lancet. 1981. 2:1003–1005.29. Horan PK, Slezak SE. Stable cell membrane labelling. Nature. 1989. 340:167–168.30. Horan PK, Melnicoff MJ, Jensen BD, Slezak SE. Fluorescent cell labeling for in vivo and in vitro cell tracking. Methods Cell Biol. 1990. 33:469–490.31. Slezak SE, Horan PK. Fluorescent in vivo tracking of hematopoietic cells. Part I. Technical considerations. Blood. 1989. 74:2172–2177.32. Slezak SE, Muirhead KA. Radioactive cell membrane labelling. Nature. 1991. 352:261–262.33. Teare GF, Horan PK, Slezak SE, et al. Long-term tracking of lymphocytes in vivo: the migration of PKH-labeled lymphocytes. Cell Immunol. 1991. 134:157–170.34. Bertelmann E, Pleyer U. Immunomodulatory therapy in ophthalmology - is there a place for topical application? Ophthalmologica. 2004. 218:359–367.35. Claman HN. Corticosteroids as immunomodulators. Ann N Y Acad Sci. 1993. 685:288–292.36. Nussenblatt RB, Whitcup SM, Palestine AG. Uveitis. 1996. 2nd ed. St.Louis: Mosby;99–106.37. Olsen TW, Benegas NM, Joplin AC, et al. Rapamycin inhibits corneal allograft rejection and neovascularization. Arch Ophthalmol. 1994. 112:1471–1475.38. Ohia EO, Mancino M, Kulkarni PS. Effects of steroids and immunosuppressive drugs on endotoxin-uveitis in rabbits. J Ocul Pharmacol. 1992. 8:295–307.39. Roberge FG, Xu D, Chan CC, et al. Treatment of autoimmune uveoretinitis in the rat with rapamycin, an inhibitor of lymphocyte growth factor signal transduction. Curr Eye Res. 1993. 12:197–203.40. Sehgal SN. Rapamune (RAPA, rapamycin, sirolimus): mechanism of action immunosuppressive effect results from blockade of signal transduction and inhibition of cell cycle progression. Clin Biochem. 2006. 39:484–489.41. Sehgal SN. Rapamune (RAPA, rapamycin, sirolimus): mechanism of action immunosuppressive effect results from blockade of signal transduction and inhibition of cell cycle progression. Clin Biochem. 1998. 31:335–340.42. Garweg JG, Wegmann-Burns M, Goldberg D. Effects of daunorubicin, mitomycin C, azathiprine and cyclosporine A on human retinal pigmented epithelial, corneal endothelial and conjunctival cell lines. Graefes Arch Clin Exp Ophthalmol. 2006. 244:382–389.43. Nelson JD, Cameron JD. Krachmer JH, Mannis MJ, Holland EJ, editors. The Conjunctiva: anatomy and physiology. Cornea. 2005. v. 2. Philadelphia: Elsevier Mosby;chap. 3.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Three Cases of Oral Lichen Planus Treated with Topical Tacrolimus
- Co-cultivation of Adenovirus with Cultivated Conjunctival Epithelial Cell: Morphological Changes in Infected Cell
- Mechanisms and clinical applications of immunosuppressive medications
- A Case of Chronic Actinic Dermatitis Treated with 0.03% Topical Tacrolimus
- Long Term Treatment Concepts and Proactive Therapy for Atopic Eczema