J Vet Sci.
2011 Mar;12(1):1-6. 10.4142/jvs.2011.12.1.1.
Differential patterns of nestin and glial fibrillary acidic protein expression in mouse hippocampus during postnatal development
- Affiliations
-
- 1Department of Veterinary Anatomy, College of Veterinary Medicine, Chonnam National University, Gwangju 500-757, Korea. moonc@chonnam.ac.kr
- 2Department of Veterinary Toxicology, College of Veterinary Medicine, Chonnam National University, Gwangju 500-757, Korea.
- 3Department of Experimental Radiation Research, Research Center, Dongnam Institute of Radiological and Medical Sciences, Busan 619-753, Korea.
- 4Department of Veterinary Anatomy, College of Veterinary Medicine, Veterinary Medical Research Institute, Jeju National University, Jeju 690-756, Korea. shint@jejunu.ac.kr
- KMID: 1067330
- DOI: http://doi.org/10.4142/jvs.2011.12.1.1
Abstract
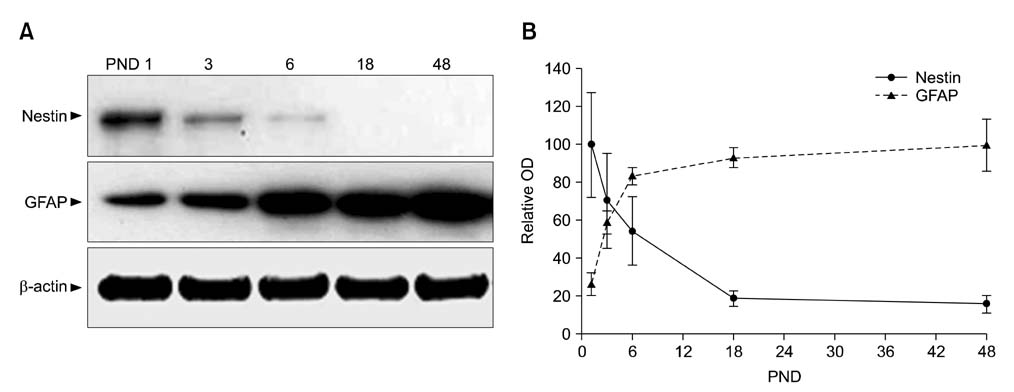
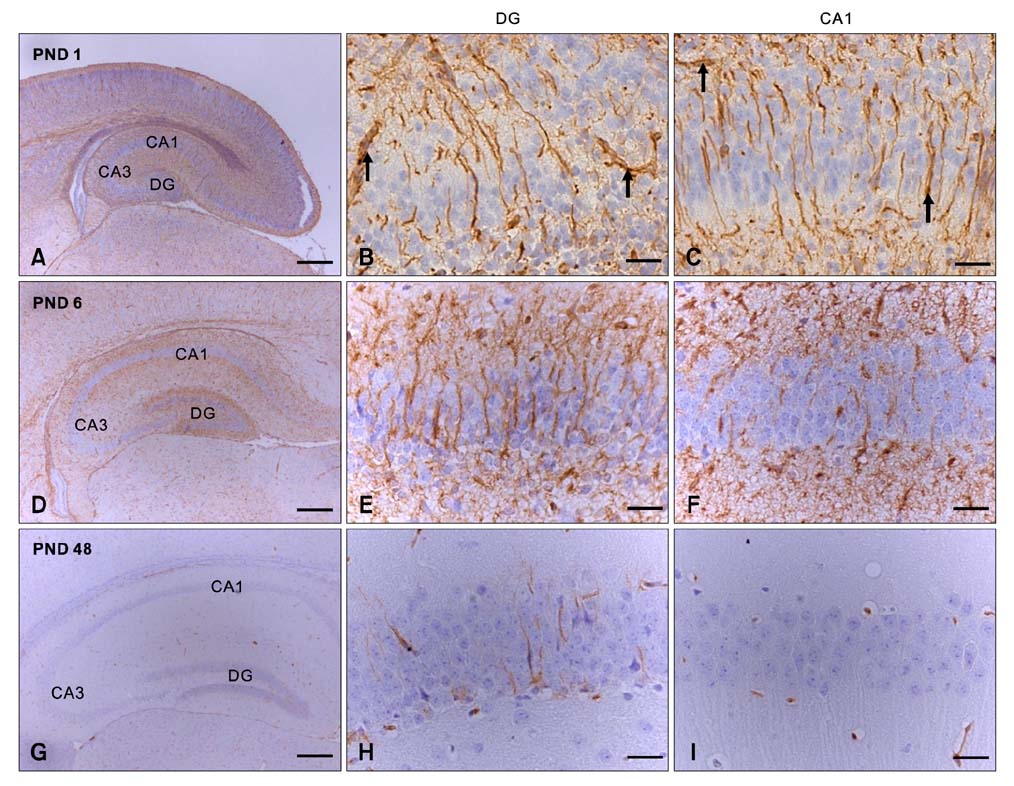
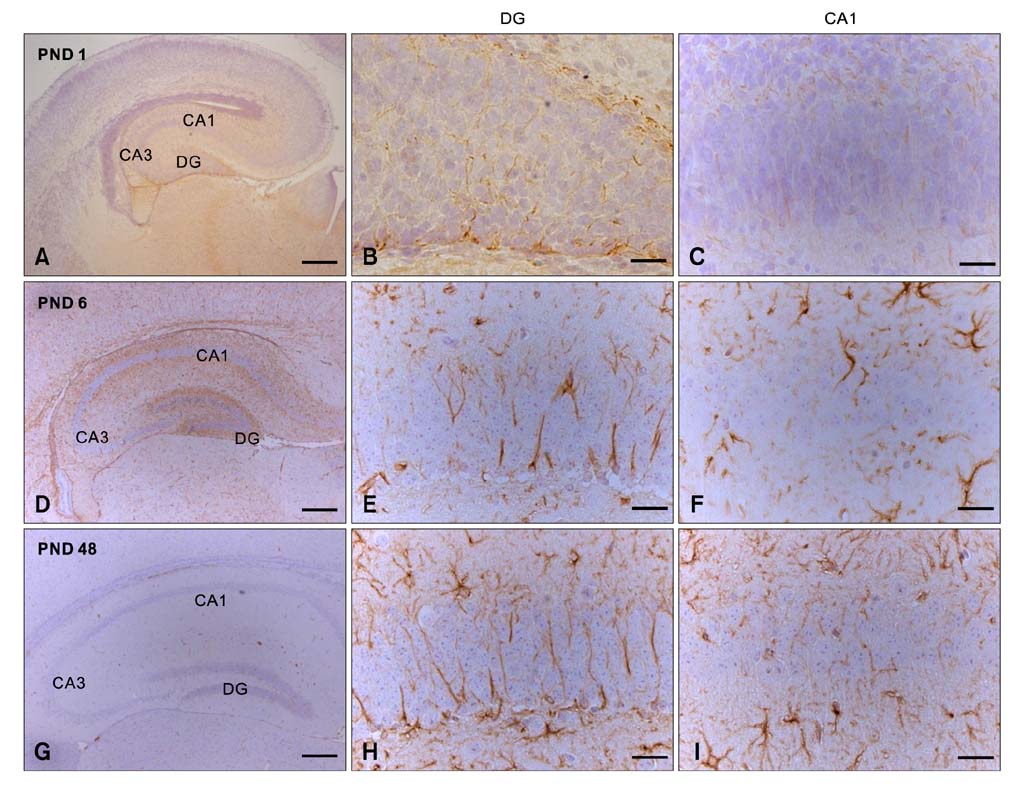
- Intermediate filaments, including nestin and glial fibrillary acidic protein (GFAP), are important for the brain to accommodate neural activities and changes during development. The present study examined the temporal changes of nestin and GFAP protein levels in the postnatal development of the mouse hippocampus. Mouse hippocampi were sampled on postnatal day (PND) 1, 3, 6, 18, and 48. Western blot analysis showed that nestin expression was high at PND 1 and markedly decreased until PND 18. Conversely, GFAP expression was acutely increased in the early phase of postnatal development. Nestin immunoreactivity was localized mainly in the processes of ramified cells at PND 1, but expression subsequently decreased. In contrast, GFAP was evident mainly in the marginal cells of the hippocampus at PND 1, but immunoreactivity revealed satellite, radial, or ramified shapes of the cells from PND 6-48. This study demonstrates that the opposing pattern of nestin and GFAP expressions in mouse hippocampus during postnatal development occur in the early development stage (PND 1-18), suggesting that the opposing change of nestin and GFAP in early postnatal development is important for neural differentiation and positioning in the mouse hippocampus.
Keyword
MeSH Terms
-
*Aging
Animals
Blotting, Western
Brain/cytology/growth & development
Female
*Gene Expression Regulation, Developmental
Glial Fibrillary Acidic Protein/genetics/*metabolism
Hippocampus/cytology/*growth & development/*metabolism
Immunohistochemistry
Intermediate Filament Proteins/genetics/*metabolism
Male
Mice
Mice, Inbred ICR
Nerve Tissue Proteins/genetics/*metabolism
Neurons/metabolism
Figure
Reference
-
1. Abdel-Rahman A, Rao MS, Shetty AK. Nestin expression in hippocampal astrocytes after injury depends on the age of the hippocampus. Glia. 2004. 47:299–313.
Article2. Bennett GS. Changes in intermediate filament composition during neurogenesis. Curr Top Dev Biol. 1987. 21:151–183.3. Compston A, Zajicek J, Sussman J, Webb A, Hall G, Muir D, Shaw C, Wood A, Scolding N. Glial lineages and myelination in the central nervous system. J Anat. 1997. 190:161–200.4. Davidoff MS, Middendorff R, Köfüncü E, Müller D, Jezek D, Holstein AF. Leydig cells of the human testis possess astrocyte and oligodendrocyte marker molecules. Acta Histochem. 2002. 104:39–49.
Article5. Eng LF, Ghirnikar RS, Lee YL. Glial fibrillary acidic protein: GFAP-thirty-one years (1969-2000). Neurochem Res. 2000. 25:1439–1451.6. Frisén J, Johansson CB, Török C, Risling M, Lendahl U. Rapid, widespread, and longlasting induction of nestin contributes to the generation of glial scar tissue after CNS injury. J Cell Biol. 1995. 131:453–464.
Article7. Fuchs E, Weber K. Intermediate filaments: structure, dynamics, function, and disease. Annu Rev Biochem. 1994. 63:345–382.
Article8. Gallo V, Armstrong RC. Developmental and growth factor-induced regulation of nestin in oligodendrocyte lineage cells. J Neurosci. 1995. 15:394–406.
Article9. Gates MA, Thomas LB, Howard EM, Laywell ED, Sajin B, Faissner A, Götz B, Silver J, Steindler DA. Cell and molecular analysis of the developing and adult mouse subventricular zone of the cerebral hemispheres. J Comp Neurol. 1995. 361:249–266.
Article10. Holmin S, Almqvist P, Lendahl U, Mathiesen T. Adult nestin-expressing subependymal cells differentiate to astrocytes in response to brain injury. Eur J Neurosci. 1997. 9:65–75.
Article11. Jin JK, Jeong BH, Na YJ, Kim YS, Carp RI, Wie MB, Moon C, Shin T. Increased expression of the embryonic intermediate filament, nestin, in the brains of scrapie-infected mice. Neurosci Lett. 2004. 367:254–258.
Article12. Kálmán M, Ajtai BM. A comparison of intermediate filament markers for presumptive astroglia in the developing rat neocortex: immunostaining against nestin reveals more detail, than GFAP or vimentin. Int J Dev Neurosci. 2001. 19:101–108.
Article13. Kempermann G, Kuhn HG, Gage FH. Genetic influence on neurogenesis in the dentate gyrus of adult mice. Proc Natl Acad Sci USA. 1997. 94:10409–10414.14. Kim JS, Lee HJ, Kim JC, Kang SS, Bae CS, Shin T, Jin JK, Kim SH, Wang H, Moon C. Transient impairment of hippocampus-dependent learning and memory in relatively low-dose of acute radiation syndrome is associated with inhibition of hippocampal neurogenesis. J Radiat Res (Tokyo). 2008. 49:517–526.
Article15. Kreplak L, Fudge D. Biomechanical properties of intermediate filaments: from tissues to single filaments and back. Bioessays. 2007. 29:26–35.
Article16. Lendahl U, Zimmerman LB, McKay RD. CNS stem cells express a new class of intermediate filament protein. Cell. 1990. 60:585–595.
Article17. Menet V, Giménez Y, Ribotta M, Sandillon F, Privat A. GFAP null astrocytes are a favorable substrate for neuronal survival and neurite growth. Glia. 2000. 31:267–272.
Article18. Miyaguchi K. Ultrastructure of intermediate filaments of nestin- and vimentin-immunoreactive astrocytes in organotypic slice cultures of hippocampus. J Struct Biol. 1997. 120:61–68.
Article19. Moon C, Ahn M, Kim S, Jin JK, Sim KB, Kim HM, Lee MY, Shin T. Temporal patterns of the embryonic intermediate filaments nestin and vimentin expression in the cerebral cortex of adult rats after cryoinjury. Brain Res. 2004. 1028:238–242.
Article20. Rutka JT, Murakami M, Dirks PB, Hubbard SL, Becker LE, Fukuyama K, Jung S, Tsugu A, Matsuzawa K. Role of glial filaments in cells and tumors of glial origin: a review. J Neurosurg. 1997. 87:420–430.
Article21. Seo HS, Yang M, Song MS, Kim JS, Kim SH, Kim JC, Kim H, Shin T, Wang H, Moon C. Toluene inhibits hippocampal neurogenesis in adult mice. Pharmacol Biochem Behav. 2010. 94:588–594.
Article22. Tardy M, Fages C, Le Prince G, Rolland B, Nunez J. Regulation of the glial fibrillary acidic protein (GFAP) and of its encoding mRNA in the developing brain and in cultured astrocytes. Adv Exp Med Biol. 1990. 265:41–52.
Article23. Wegner M, Stolt CC. From stem cells to neurons and glia: a Soxist's view of neural development. Trends Neurosci. 2005. 28:583–588.
Article24. Wei LC, Shi M, Chen LW, Cao R, Zhang P, Chan YS. Nestin-containing cells express glial fibrillary acidic protein in the proliferative regions of central nervous system of postnatal developing and adult mice. Brain Res Dev Brain Res. 2002. 139:9–17.
Article25. Wiese C, Rolletschek A, Kania G, Blyszczuk P, Tarasov KV, Tarasova Y, Wersto RP, Boheler KR, Wobus AM. Nestin expression--a property of multi-lineage progenitor cells? Cell Mol Life Sci. 2004. 61:2510–2522.26. Yang M, Kim JS, Song MS, Kim SH, Kang SS, Bae CS, Kim JC, Wang H, Shin T, Moon C. Cyclophosphamide impairs hippocampus-dependent learning and memory in adult mice: Possible involvement of hippocampal neurogenesis in chemotherapy-induced memory deficits. Neurobiol Learn Mem. 2010. 93:487–494.
Article27. Zerlin M, Levison SW, Goldman JE. Early patterns of migration, morphogenesis, and intermediate filament expression of subventricular zone cells in the postnatal rat forebrain. J Neurosci. 1995. 15:7238–7249.
Article28. Zimmerman L, Parr B, Lendahl U, Cunningham M, McKay R, Gavin B, Mann J, Vassileva G, McMahon A. Independent regulatory elements in the nestin gene direct transgene expression to neural stem cells or muscle precursors. Neuron. 1994. 12:11–24.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Nestin Expression in the Adult Mouse Retina with Pharmaceutically Induced Retinal Degeneration
- Immunohistochemical Changes of Nestin, BrdU, and GFAP in the Cells of Rat Spinal Ependymal Zone According to Their Developmental Stages
- Expression of Nestin on Endothelial Cells and Pericytes During Retinal Vascular Development in Mouse
- Time Course and Characteristics of Astrocyte Activation in the Rat Brain after Injury
- Vimentin and Survivin Expression Rates as Prognostic Factors in Medulloblastoma