Yonsei Med J.
2008 Jun;49(3):486-495. 10.3349/ymj.2008.49.3.486.
Protective Effect of Ultra Low Molecular Weight Heparin on Glutamate-Induced Apoptosis in Cortical Cells
- Affiliations
-
- 1Pharmacological Institute of New Drugs, School of Pharmacy, Shandong University, 44 Wen Hua Xi Road, Jinan, P.R. China. sdzhangqz@163.com
- 2Institute of Biochemical and Biotechnological Drugs, School of Pharmacy, Shandong University, 44 Wen Hua Xi Road, Jinan, P.R. China.
- 3Institute of Materia Medica, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, P.R. China.
- KMID: 724266
- DOI: http://doi.org/10.3349/ymj.2008.49.3.486
Abstract
- PURPOSE
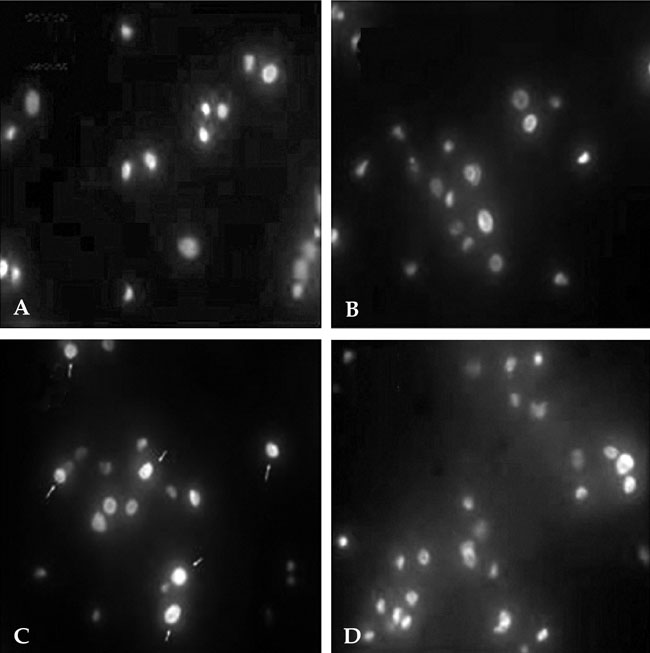
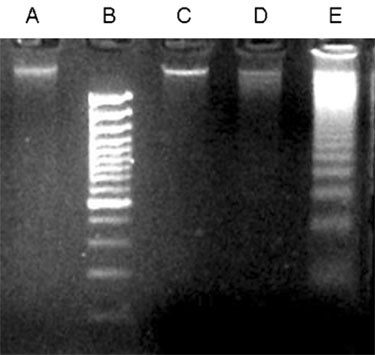
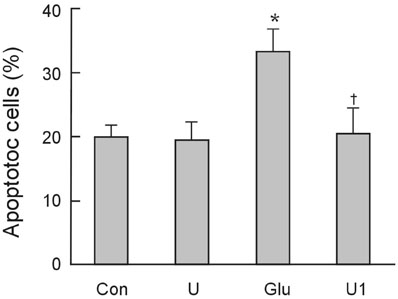
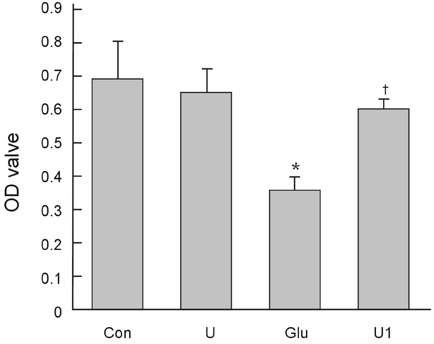
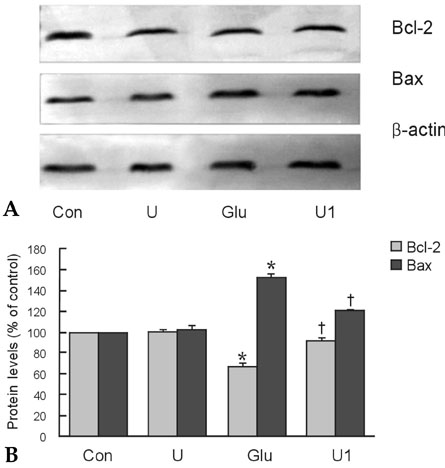
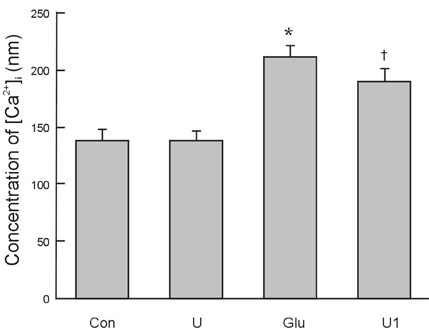
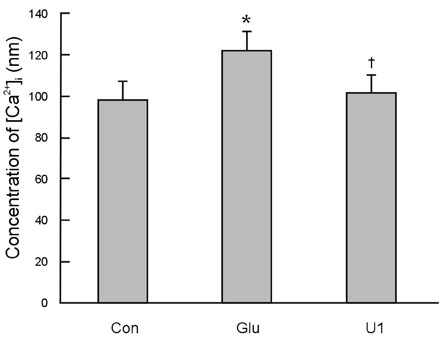
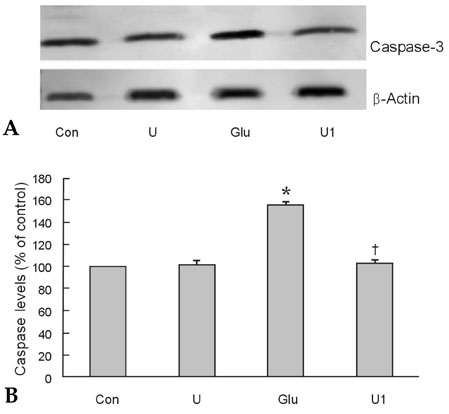
To investigate the effect of ultra low molecular weight heparin (ULMWH) on glutamate induced apoptosis in rat cortical cells and to explore the possible mechanisms. MATERIALS AND METHODS: Cell viability was measured using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. Apoptosis was first analyzed with Hoechst 33258 and then confirmed by DNA fragmentation. The concentration of free intracellular calcium ([Ca2+](i)) was determined with fura-2/AM fluorometry. The expression of Bcl-2 family protein and caspase-3 were evaluated with Western blot. RESULTS: Typical apoptotic morphological change in rat cortical cells treated with 100micromol/L glutamate for 24h was detected by Hoechst 33258 staining, which was then confirmed by the DNA ladder of agarose gel electrophoresis. The apoptotic rate of the glutamate treated cells was up to 33.21%, and 24 h of treatment with glutamate increased [Ca2+](i), down-regulated Bcl-2 expression, up-regulated Bax expression, and increased caspase-3 activation in rat cortical cells. Our research demonstrated that ULMWH pretreatment can prevent the glutamate- induced apoptosis, attenuate the increase of [Ca2+](i) not only in medium containing Ca2+ but also in Ca2+-free medium, up-regulate the expression of Bcl-2, down-regulate the expression of Bax, and decrease caspase-3 activation. CONCLUSION: ULMWH has neuroprotective capacity to antagonize glutamate-induced apoptosis in cortical cells, through decrease of Ca2+ release and modulation of apoptotic processes.
MeSH Terms
-
Animals
Apoptosis/*drug effects
Blotting, Western
Calcium/metabolism
Caspase 3/metabolism
Cell Survival/drug effects
Cells, Cultured
Cerebral Cortex/cytology
DNA Fragmentation/drug effects
Glutamic Acid/*pharmacology
Heparin, Low-Molecular-Weight/*pharmacology
Proto-Oncogene Proteins c-bcl-2/metabolism
Rats
Rats, Wistar
bcl-2-Associated X Protein/metabolism
Figure
Reference
-
1. Bergamaschini L, Rossi E, Storini C, Pizzimenti S, Distaso M, Perego C, et al. Peripheral treatment with enoxaparin, a low molecular weight heparin, reduces plaques and beta-amyloid accumulation in a mouse model of Alzheimer's disease. J Neurosci. 2004. 24:4181–4186.
Article2. Zhu H, Yu J, Kindy MS. Inhibition of amyloidosis using low-molecular-weight heparins. Mol Med. 2001. 7:517–522.
Article3. Jonas S, Sugimori M, Llinás R. Is low molecular weight heparin a neuroprotectant? Ann N Y Acad Sci. 1997. 825:389–393.
Article4. Ma Q, Dudas B, Hejna M, Cornelli U, Lee JM, Lorens S, et al. The blood-brain barrier accessibility of a heparin-derived oligosaccharides C3. Thromb Res. 2002. 105:447–453.
Article5. Zhang ZG, Zhang QZ, Cheng YN, Ji SL, Du GH. Antagonistic effects of ultra-low-molecular-weight heparin against cerebral ischemia/reperfusion injury in rats. Pharmacol Res. 2007. 56:350–355.
Article6. Duarte AI., Santos MS, Oliveira CR, Rego AC. Insulin neuroprotection against oxidative stress in cortical neurons-involvement of uric acid and glutathione antioxidant defenses. Free Radic Biol Med. 2005. 39:876–889.
Article7. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983. 65:55–63.8. Cavaliere F, D'Ambrosi N, Sancesario G, Bernardi G, Volonté C. Hypoglycaemia-induced cell death: features of neuroprotection by the P2 receptor antagonist basilen blue. Neurochem Int. 2001. 38:199–207.
Article9. Wolozin B, Iwasaki K, Vito P, Ganjei JK, Lacanà E, Sunderland T, et al. Participation of presenilin 2 in apoptosis: enhanced basal activity conferred by an Alzheimer mutation. Science. 1996. 274:1710–1713.
Article10. Grynkiewicz G, Poenie M, Tsien RY. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985. 260:3440–3450.
Article11. Sur P, Sribnick EA, Wingrave JM, Nowak MW, Ray SK, Banik NL. Estrogen attenuates oxidative stress-induced apoptosis in C6 glial cells. Brain Res. 2003. 971:178–188.
Article12. Bleich S, Romer K, Wiltfang J, Kornhuber J. Glutamate and the glutamate receptor system: a target for drug action. Int J Geriatr Psychiatry. 2003. 18:Suppl 1. S33–S40.
Article13. Conn PJ. Physiological roles and therapeutic potential of metabotropic glutamate receptors. Ann N Y Acad Sci. 2003. 1003:12–21.
Article14. Spillson AB, Russell JW. Metabotropic glutamate receptor regulation of neuronal cell death. Exp Neurol. 2003. 184:Suppl 1. S97–S105.
Article15. Mori H, Mishina M. Roles of diverse glutamate receptors in brain functions elucidated by subunit-specific and region-specific gene targeting. Life Sci. 2003. 74:329–336.16. Huettner JE. Kainate receptors and synaptic transmission. Prog Neurobiol. 2003. 70:387–407.17. Manev H, Favaron M, Guidotti A, Costa E. Delayed increase of Ca2+ influx elicited by glutamate: role in neuronal death. Mol Pharmacol. 1989. 36:106–112.18. Naarala JT, Loikkanen JJ, Ruotsalainen MH, Savolainen KM. Lead amplifies glutamate-induced oxidative stress. Free Radic Biol Med. 1995. 19:689–693.
Article19. Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, et al. Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron. 1995. 15:961–973.
Article20. Choi DW. Ionic dependence of glutamate neurotoxicity. J Neurosci. 1987. 7:369–379.21. Koh JY, Yang LL, Cotman CW. Beta-amyloid protein increases the vulnerability of cultured cortical neurons to excitotoxic damage. Brain Res. 1990. 533:315–320.22. Smith T, Groom A, Zhu B, Turski L. Autoimmune encephalomyelitis ameliorated by AMPA antagonists. Nat Med. 2000. 6:62–66.
Article23. Turski L, Bressler K, Rettig KJ, Löschmann PA, Wachtel H. Protection of substantia nigra form MPP+ neurotoxicity by N-methyl-D-aspartate antagonists. Nature. 1991. 349:414–418.
Article24. Matsuzaki H, Tamatani M, Mitsuda N, Namikawa K, Kiyama H, Miyake S, et al. Activation of Akt kinase inhibits apoptosis and changes in Bcl-2 and Bax expression induced by nitric oxide in primary hippocampal neurons. J Neurochem. 1999. 73:2037–2046.
Article25. Choi DW, Rothman SM. The role of glutamate neurotoxicity in hypoxic-ischemic neuronal death. Annu Rev Neurosci. 1990. 13:171–182.
Article26. Kristián T, Siesjö BK. Calcium-related damage in ischemia. Life Sci. 1996. 59:357–367.
Article27. Nishizawa Y. Glutamate release and neuronal damage in ischemia. Life Sci. 2001. 69:369–381.
Article28. Vaux DL, Cory S, Adams JM. Bcl-2 gene promotes haemopoietic cell survival and cooperates with c-myc to immortalize pre-B cells. Nature. 1988. 335:440–442.
Article29. Ghosh TK, Eis PS, Mullaney JM, Ebert CL, Gill DL. Competitive, reversible, and potent antagonism of inositol 1,4,5-trisphosphate-activated calcium release by heparin. J Biol Chem. 1988. 263:11075–11079.
Article30. Cullen PJ, Comerford JG, Dawson AP. Heparin inhibits the inositol 1,4,5-trisphosphate-induced Ca2+ release from rat liver microsomes. FEBS Lett. 1988. 228:57–59.
Article31. Berridge MJ. Inositol trisphosphate and calcium signalling. Nature. 1993. 361:315–325.
Article32. Trump BF, Berezesky IK. Calcium-mediated cell injury and cell death. FASEB J. 1995. 9:219–228.
Article33. Pasquet JM, Dachary-Prigent J, Nurden AT. Calcium influx is a determining factor of calpain activation and microparticle formation in platelets. Eur J Biochem. 1996. 239:647–654.
Article34. Merry DE, Korsmeyer SJ. Bcl-2 gene family in the nervous system. Annu Rev Neurosci. 1997. 20:245–267.
Article35. Staunton MJ, Gaffney EF. Apoptosis: basic concepts and potential significance in human cancer. Arch Pathol Lab Med. 1998. 122:310–319.36. Korsmeyer SJ. Bcl-2 initiates a new category of oncogenes: regulators of cell death. Blood. 1992. 80:879–886.
Article37. Haunstetter A, Izumo S. Apoptosis: basic mechanisms and implications for cardiovascular disease. Circ Res. 1998. 82:1111–1129.38. Kajstura J, Cheng W, Reiss K, Clark WA, Sonnenblick EH, Krajewski S, et al. Apoptotic and necrotic myocyte cell deaths are independent contributing variables of infarct size in rats. Lab Invest. 1996. 74:86–107.39. Oltvai ZN, Milliman CL, Korsmeyer SJ. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell. 1993. 74:609–619.
Article40. Huang Q, Singh B, Sharawy M. Immunohistochemical analysis of Bcl-2 and Bax oncoproteins in rabbit craniomandibular joint. Arch Oral Biol. 2004. 49:143–148.
Article41. Eves EM, Xiong W, Bellacosa A, Kennedy SG, Tsichlis PN, Rosner MR, et al. Akt, a target of phosphatidylinositol 3-kinase, inhibits apoptosis in a differentiating neuronal cell line. Mol Cell Biol. 1998. 18:2143–2152.
Article42. Zhong LT, Kane DJ, Bredesen DE. Bcl-2 blocks glutamate toxicity in neural cell lines. Brain Res Mol Brain Res. 1993. 19:353–355.
Article43. Thornberry NA, Lazebnik Y. Caspases: enemies within. Science. 1998. 281:1312–1316.
Article44. Yang JN, Liu CX, Xu H, Pan QC. Caspases promoted DADAG-induced apoptosis in human leukemia HL-60 cells. Acta Pharmacol Sin. 2002. 23:461–466.45. Daniel PT. Dissecting the pathways to death. Leukemia. 2000. 14:2035–2044.46. Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science. 1997. 275:1132–1136.47. Chen J, Nagayama T, Jin K, Stetler RA, Zhu Rl, Graham SH, et al. Induction of caspase-3-like protease may mediate delayed neuronal death in the hippocampus after transient cerebral ischemia. J Neurosci. 1998. 18:4914–4928.48. Annunziato L, Amoroso S, Pannaccione A, Cataldi M, Pignataro G, D'Alessio A, et al. Apoptosis induced in neuronal cells by oxidative stress: role played by caspases and intracellular calcium ions. Toxicol Lett. 2003. 139:125–133.49. Hills FA, Abrahams VM, González-Timón B, Francis J, Cloke B, Hinkson L, et al. Heparin prevents programmed cell death in human trophoblast. Mol Hum Reprod. 2006. 12:237–243.50. Deepa PR, Varalakshmi P. Influence of a low-molecular-weight heparin derivative on the nitric oxide levels and apoptotic DNA damage in adriamycin-induced cardiac and renal toxicity. Toxicology. 2006. 217:176–183.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of standard heparin and low molecular weight heparin on fibrinolytic activity
- Successful Low Molecular Weight Heparin Treatment for the Global Alteration of Cortical Venous Drainage Developed after Intracranial Operation
- Successful Treatment of Aortic Thrombosis after Umbilical Catheterization with Low-Molecular-Weight Heparin
- Warfarin-induced Skin Necrosis After Valve Surgery
- Dabigatran approaching the realm of heparin-induced thrombocytopenia