Cancer Res Treat.
2024 Jan;56(1):199-207. 10.4143/crt.2023.490.
GASTric Cancer HER2 Re-Assessment Study 2 (GASTHER2): HER2 Re-assessment for Initially HER2-Negative Advanced Gastric Cancer Patients after Progression on First-Line Treatment
- Affiliations
-
- 1Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 2Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2550336
- DOI: http://doi.org/10.4143/crt.2023.490
Abstract
- Purpose
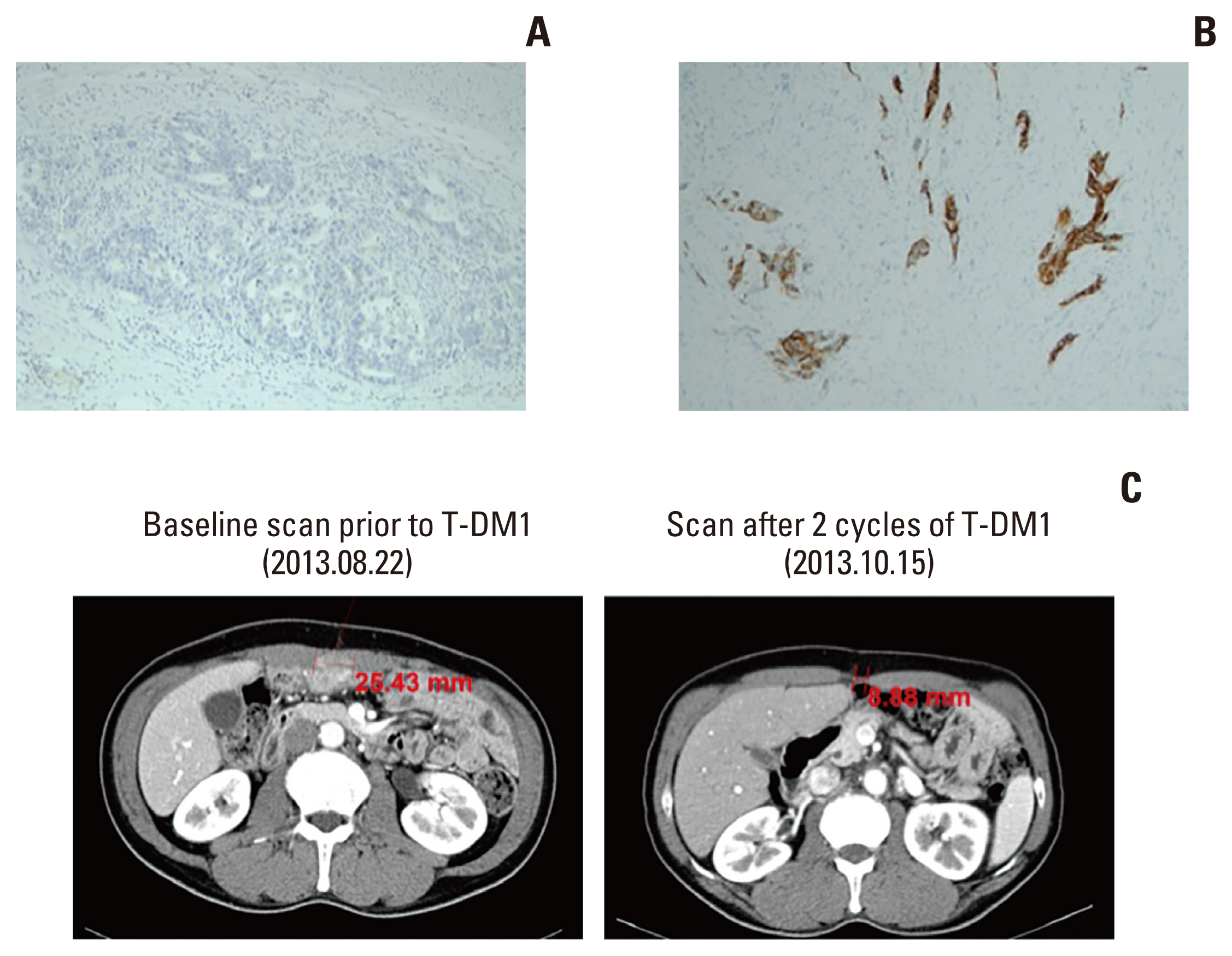
Heterogeneous human epidermal growth factor receptor 2 (HER2) overexpression in gastric cancer may lead to a misdiagnosis of HER2 status. Accurate assessment of HER2 status is essential for optimal treatment as novel HER2-directed agents are being investigated in various clinical settings. We evaluated the usefulness of HER2 re-assessment following progression on first-line treatment in initially HER2-negative advanced gastric cancer (AGC) patients.
Materials and Methods
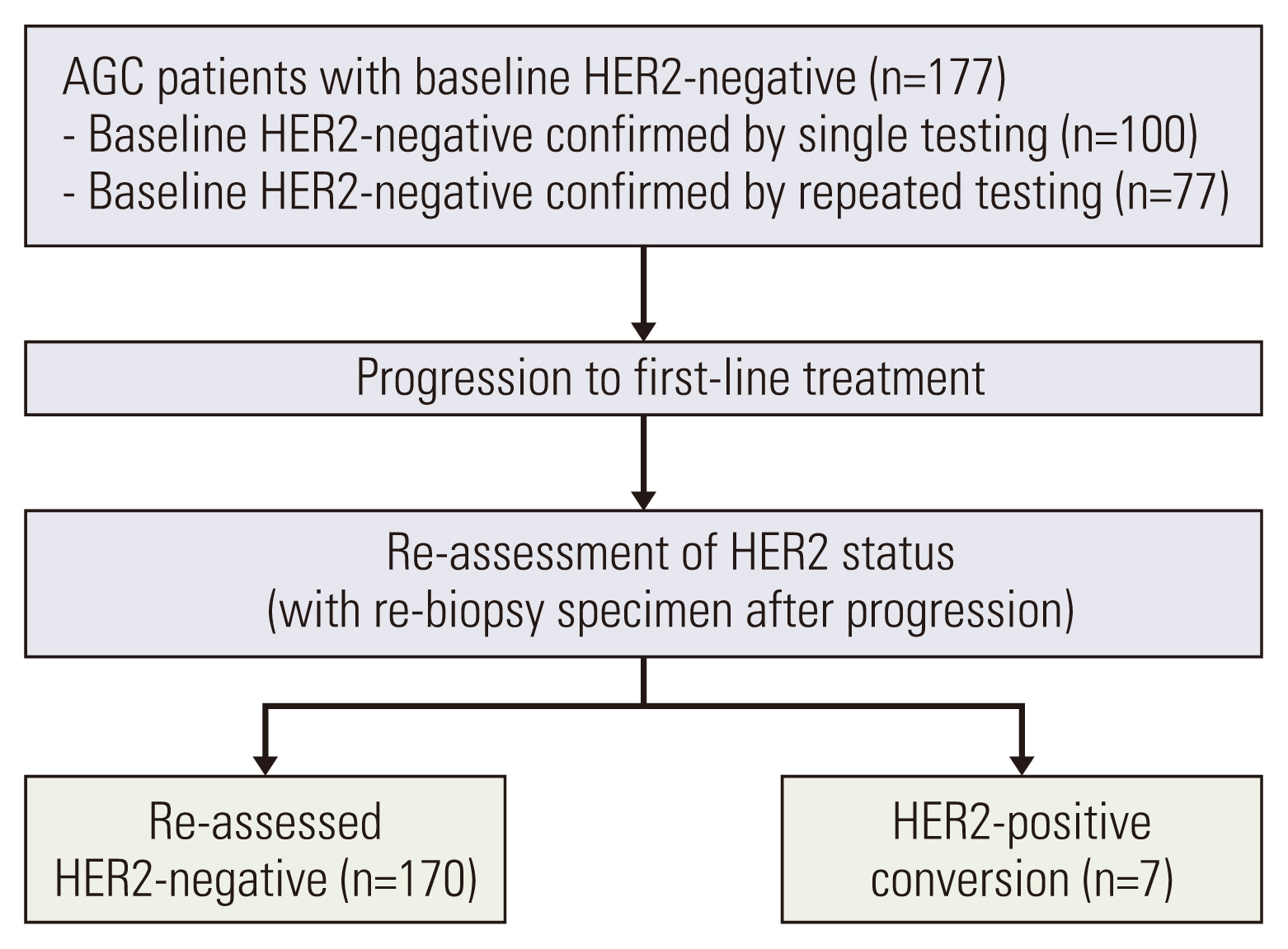
We enrolled 177 patients with baseline HER2-negative AGC and performed HER2 re-assessment after progression on first-line treatment from February 2012 to June 2016 at Asan Medical Center, Seoul, Korea. The re-assessed HER2 status was analyzed with baseline HER2 status and clinical characteristics.
Results
The median age was 54 years (range, 24 to 80 years), and 123 patients (69.5%) were men. Seven patients (4.0%) were HER2-positive on the re-assessment. Patients with baseline HER2 negativity confirmed by a single test (n=100) had a higher HER2-positive re-assessment rate compared to those who had repeated baseline testing (n=77) (5.0% vs. 2.6%). Among the patients with single baseline HER2 testing, the rate was higher in patients with baseline HER2 immunohistochemistry (IHC) 1+ compared to those with IHC 0 (13.4% vs. 3.6%).
Conclusion
Overall, 4.0% of patients with baseline HER2-negative AGC were HER2-positive on re-assessment, and the HER2-positive re-assessment rate was higher among patients who had a single test at baseline. HER2 re assessment may be considered for initially HER2-negative patients to determine their eligibility for HER2-directed therapy, particularly if their HER2 negativity was determined by a single test, especially if they had a single baseline HER2 IHC 1+ test.
Figure
Reference
-
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018; 68:7–30.2. Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020; 396:635–48.3. Nakamura Y, Kawazoe A, Lordick F, Janjigian YY, Shitara K. Biomarker-targeted therapies for advanced-stage gastric and gastro-oesophageal junction cancers: an emerging paradigm. Nat Rev Clin Oncol. 2021; 18:473–87.4. Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010; 376:687–97.5. Gastric cancer (version 2.2022) [Internet]. Plymouth Meeting, PA: National Comprehensive Cancer Network;c2022 [cited 2022 Jun 14]. Available from: https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf .6. Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513:202–9.7. Van Cutsem E, Bang YJ, Feng-Yi F, Xu JM, Lee KW, Jiao SC, et al. HER2 screening data from ToGA: targeting HER2 in gastric and gastroesophageal junction cancer. Gastric Cancer. 2015; 18:476–84.8. Yagi S, Wakatsuki T, Yamamoto N, Chin K, Takahari D, Ogura M, et al. Clinical significance of intratumoral HER2 heterogeneity on trastuzumab efficacy using endoscopic biopsy specimens in patients with advanced HER2 positive gastric cancer. Gastric Cancer. 2019; 22:518–25.9. Tominaga N, Gotoda T, Hara M, Hale MD, Tsuchiya T, Matsubayashi J, et al. Five biopsy specimens from the proximal part of the tumor reliably determine HER2 protein expression status in gastric cancer. Gastric Cancer. 2016; 19:553–60.10. Bartley AN, Washington MK, Colasacco C, Ventura CB, Ismaila N, Benson AB 3rd, et al. HER2 testing and clinical decision making in gastroesophageal adenocarcinoma: guideline from the College of American Pathologists, American Society for Clinical Pathology, and the American Society of Clinical Oncology. J Clin Oncol. 2017; 35:446–64.11. Park SR, Park YS, Ryu MH, Ryoo BY, Woo CG, Jung HY, et al. Extragain of HER2-positive cases through HER2 reassessment in primary and metastatic sites in advanced gastric cancer with initially HER2-negative primary tumours: Results of GASTric cancer HER2 reassessment study 1 (GASTHER1). Eur J Cancer. 2016; 53:42–50.12. R: A Language and Environment for Statistical Computing [Internet]. Vienna: R Foundation for Statistical Computing;c2021 [cited 2022 Jun 14]. Available from: https://www.R-project.org/ .13. Thuss-Patience PC, Shah MA, Ohtsu A, Van Cutsem E, Ajani JA, Castro H, et al. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): an international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol. 2017; 18:640–53.14. Saeki H, Oki E, Kashiwada T, Arigami T, Makiyama A, Iwatsuki M, et al. Re-evaluation of HER2 status in patients with HER2-positive advanced or recurrent gastric cancer refractory to trastuzumab (KSCC1604). Eur J Cancer. 2018; 105:41–9.15. Pietrantonio F, Caporale M, Morano F, Scartozzi M, Gloghini A, De Vita F, et al. HER2 loss in HER2-positive gastric or gastroesophageal cancer after trastuzumab therapy: implication for further clinical research. Int J Cancer. 2016; 139:2859–64.16. Seo S, Ryu MH, Park YS, Ahn JY, Park Y, Park SR, et al. Loss of HER2 positivity after anti-HER2 chemotherapy in HER2-positive gastric cancer patients: results of the GASTric cancer HER2 reassessment study 3 (GASTHER3). Gastric Cancer. 2019; 22:527–35.17. Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014; 15:1224–35.18. Shah MA, Kang YK, Thuss-Patience PC, Ohtsu A, Ajani JA, Van Cutsem E, et al. Biomarker analysis of the GATSBY study of trastuzumab emtansine versus a taxane in previously treated HER2-positive advanced gastric/gastroesophageal junction cancer. Gastric Cancer. 2019; 22:803–16.19. Haffner I, Schierle K, Raimundez E, Geier B, Maier D, Hasenauer J, et al. HER2 expression, test deviations, and their impact on survival in metastatic gastric cancer: results from the prospective multicenter VARIANZ study. J Clin Oncol. 2021; 39:1468–78.20. Wakatsuki T, Yamamoto N, Sano T, Chin K, Kawachi H, Takahari D, et al. Clinical impact of intratumoral HER2 heterogeneity on trastuzumab efficacy in patients with HER2-positive gastric cancer. J Gastroenterol. 2018; 53:1186–95.21. Bang K, Cheon J, Park YS, Kim HD, Ryu MH, Park Y, et al. Association between HER2 heterogeneity and clinical outcomes of HER2-positive gastric cancer patients treated with trastuzumab. Gastric Cancer. 2022; 25:794–803.22. Shitara K, Bang YJ, Iwasa S, Sugimoto N, Ryu MH, Sakai D, et al. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N Engl J Med. 2020; 382:2419–30.23. Ogitani Y, Aida T, Hagihara K, Yamaguchi J, Ishii C, Harada N, et al. DS-8201a, a novel HER2-targeting ADC with a novel DNA topoisomerase I inhibitor, demonstrates a promising antitumor efficacy with differentiation from T-DM1. Clin Cancer Res. 2016; 22:5097–108.24. Ogitani Y, Hagihara K, Oitate M, Naito H, Agatsuma T. Bystander killing effect of DS-8201a, a novel anti-human epidermal growth factor receptor 2 antibody-drug conjugate, in tumors with human epidermal growth factor receptor 2 heterogeneity. Cancer Sci. 2016; 107:1039–46.25. Nishikawa K, Takahashi T, Takaishi H, Miki A, Noshiro H, Yoshikawa T, et al. Phase II study of the effectiveness and safety of trastuzumab and paclitaxel for taxane- and trastuzumab-naive patients with HER2-positive, previously treated, advanced, or recurrent gastric cancer (JFMC45-1102). Int J Cancer. 2017; 140:188–96.26. Catenacci DV, Kang YK, Saeed A, Yamaguchi K, Qin S, Lee KW, et al. FIGHT: a randomized, double-blind, placebo-controlled, phase II study of bemarituzumab (bema) combined with modified FOLFOX6 in 1L FGFR2b+ advanced gastric/gastroesophageal junction adenocarcinoma (GC). J Clin Oncol. 2021; 39(15 Suppl):4010.27. Sahin U, Tureci O, Manikhas G, Lordick F, Rusyn A, Vynnychenko I, et al. FAST: a randomised phase II study of zolbetuximab (IMAB362) plus EOX versus EOX alone for first-line treatment of advanced CLDN18.2-positive gastric and gastro-oesophageal adenocarcinoma. Ann Oncol. 2021; 32:609–19.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnosis and Treatment of HER2-Positive Breast Cancer
- HER2-negative or low expression as an unfavorable prognostic factor in patients with stage I/II uterine carcinosarcoma
- Breakthroughs in the Systemic Treatment of HER2-Positive Advanced/Metastatic Gastric Cancer: From Singlet Chemotherapy to Triple Combination
- HER2 status in breast cancer: changes in guidelines and complicating factors for interpretation
- Prognostic Value of the Evolution of HER2-Low Expression after Neoadjuvant Chemotherapy