Korean J Physiol Pharmacol.
2021 May;25(3):177-187. 10.4196/kjpp.2021.25.3.177.
Morin alleviates fructose-induced metabolic syndrome in rats via ameliorating oxidative stress, inflammatory and fibrotic markers
- Affiliations
-
- 1Departments of Pharmacology and Toxicology, Faculty of Pharmacy, Minia University, El-Minia 61111, Egypt
- 2Departments of Biochemistry, Faculty of Pharmacy, Minia University, El-Minia 61111, Egypt
- KMID: 2514943
- DOI: http://doi.org/10.4196/kjpp.2021.25.3.177
Abstract
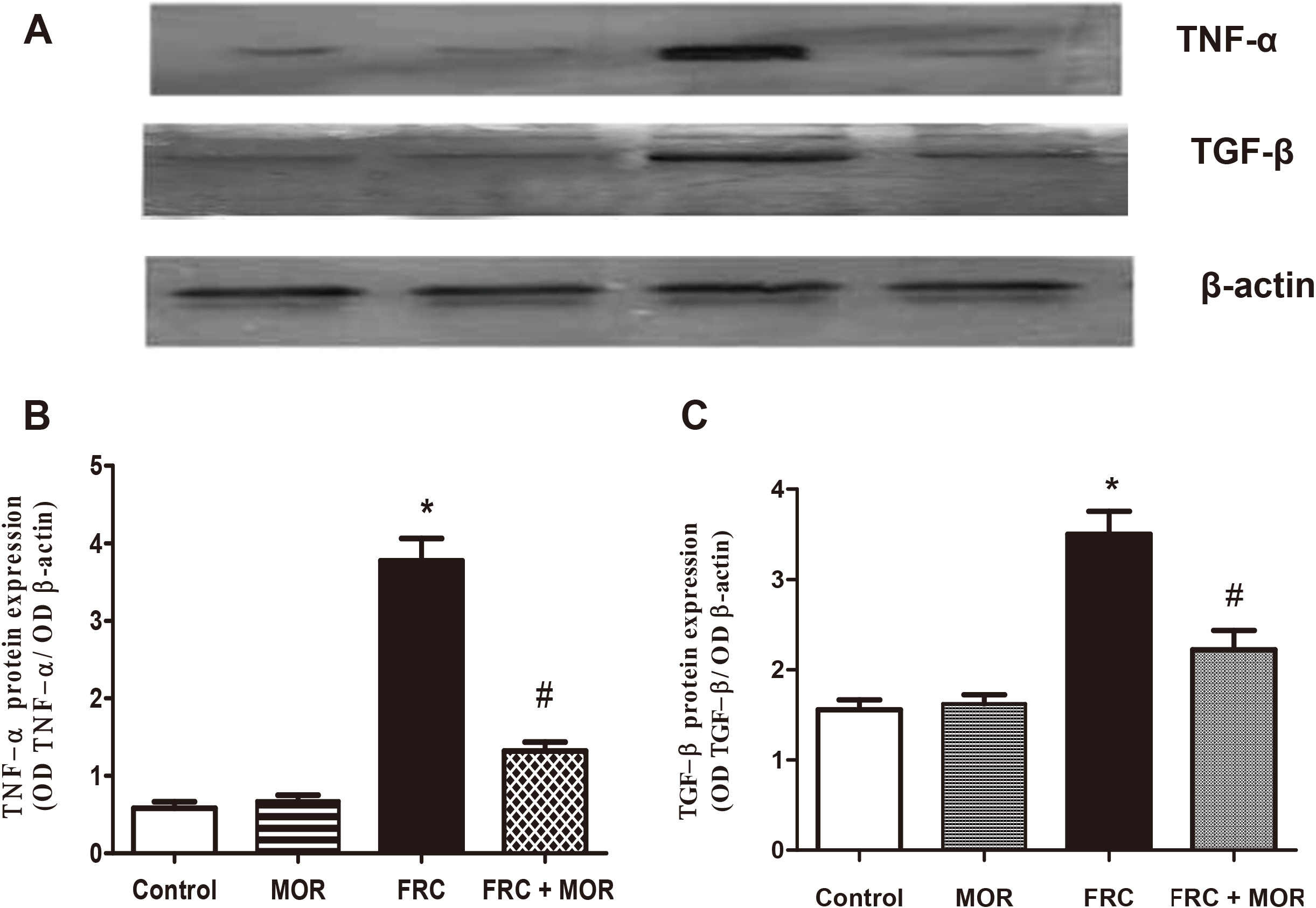
- Metabolic syndrome (MBS) is a widespread disease that has strongly related to unhealthy diet and low physical activity, which initiate more serious conditions such as obesity, cardiovascular diseases and type 2 diabetes mellitus. This study aimed to examine the therapeutic effects of morin, as one of the flavonoids constituents, which widely exists in many herbs and fruits, against some metabolic and hepatic manifestations observed in MBS rats and the feasible related mechanisms. MBS was induced in rats by high fructose diet feeding for 12 weeks. Morin (30 mg/ kg) was administered orally to both normal and MBS rats for 4 weeks. Liver tissues were used for determination of liver index, hepatic expression of glucose transporter 2 (GLUT2) as well as both inflammatory and fibrotic markers. The fat/muscle ratio, metabolic parameters, systolic blood pressure, and oxidative stress markers were also determined. Our data confirmed that the administration of morin in fructose diet rats significantly reduced the elevated systolic blood pressure. The altered levels of metabolic parameters such as blood glucose, serum insulin, serum lipid profile, and oxidative stress markers were also reversed approximately to the normal values. In addition, morin treatment decreased liver index, serum liver enzyme activities, and fat/muscle ratio. Furthermore, morin relatively up-regulated GLUT2 expression, however, down-regulated NF-kB, TNF-β, and TGF-β expressions in the hepatic tissues. Here, we revealed that morin has an exquisite effect against metabolic disorders in the experimental model through, at least in part, antioxidant, anti-inflammatory, and anti-fibrotic mechanisms.
Keyword
Figure
Reference
-
1. Bray GA, Nielsen SJ, Popkin BM. 2004; Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. Am J Clin Nutr. 79:537–543. DOI: 10.1093/ajcn/79.4.537. PMID: 15051594.
Article2. Sánchez-Lozada LG, Tapia E, Jiménez A, Bautista P, Cristóbal M, Nepomuceno T, Soto V, Avila-Casado C, Nakagawa T, Johnson RJ, Herrera-Acosta J, Franco M. 2007; Fructose-induced metabolic syndrome is associated with glomerular hypertension and renal microvascular damage in rats. Am J Physiol Renal Physiol. 292:F423–F429. DOI: 10.1152/ajprenal.00124.2006. PMID: 16940562.
Article3. Kelishadi R, Mansourian M, Heidari-Beni M. 2014; Association of fructose consumption and components of metabolic syndrome in human studies: a systematic review and meta-analysis. Nutrition. 30:503–510. DOI: 10.1016/j.nut.2013.08.014. PMID: 24698343.
Article4. Sabir AA, Bilbis LS, Saidu Y, Jimoh A, Iwuala SO, Isezuo SA, Kaoje AU, Abubakar SA. 2016; Oxidative stress among subjects with metabolic syndrome in Sokoto, North-Western Nigeria. Niger J Clin Pract. 19:128–132. DOI: 10.4103/1119-3077.173705. PMID: 26755231.
Article5. Lorenzo C, Okoloise M, Williams K, Stern MP, Haffner SM. 2003; The metabolic syndrome as predictor of type 2 diabetes: the San Antonio heart study. Diabetes Care. 26:3153–3159. DOI: 10.2337/diacare.26.11.3153. PMID: 14578254.
Article6. Maki KC. 2004; Dietary factors in the prevention of diabetes mellitus and coronary artery disease associated with the metabolic syndrome. Am J Cardiol. 93(11A):12C–17C. DOI: 10.1016/j.amjcard.2004.02.001. PMID: 15178512.
Article7. Sivaramakrishnan V, Devaraj SN. 2010; Morin fosters apoptosis in experimental hepatocellular carcinogenesis model. Chem Biol Interact. 183:284–292. DOI: 10.1016/j.cbi.2009.11.011. PMID: 19931519.
Article8. Zhang R, Kang KA, Kang SS, Park JW, Hyun JW. 2011; Morin (2',3, 4',5,7-pentahydroxyflavone) protected cells against γ-radiation-induced oxidative stress. Basic Clin Pharmacol Toxicol. 108:63–72. DOI: 10.1111/j.1742-7843.2010.00629.x. PMID: 20807219.9. Al Numair KS, Chandramohan G, Alsaif MA, Baskar AA. 2012; Protective effect of morin on cardiac mitochondrial function during isoproterenol-induced myocardial infarction in male Wistar rats. Redox Rep. 17:14–21. DOI: 10.1179/1351000211Y.0000000019. PMID: 22340511. PMCID: PMC6837627.
Article10. Yan X, Qi M, Li P, Zhan Y, Shao H. 2017; Apigenin in cancer therapy: anti-cancer effects and mechanisms of action. Cell Biosci. 7:50. DOI: 10.1186/s13578-017-0179-x. PMID: 29034071. PMCID: PMC5629766.
Article11. Marunaka Y, Marunaka R, Sun H, Yamamoto T, Kanamura N, Inui T, Taruno A. 2017; Actions of quercetin, a polyphenol, on blood pressure. Molecules. 22:209. DOI: 10.3390/molecules22020209. PMID: 28146071. PMCID: PMC6155806.
Article12. Choudhury A, Chakraborty I, Banerjee TS, Vana DR, Adapa D. 2017; Efficacy of morin as a potential therapeutic phytocomponent: insights into the mechanism of action. Int J Med Res Health Sci. 6:175–194.13. Lotito SB, Frei B. 2006; Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: cause, consequence, or epiphenomenon? Free Radic Biol Med. 41:1727–1746. DOI: 10.1016/j.freeradbiomed.2006.04.033. PMID: 17157175.
Article14. Cho YM, Onodera H, Ueda M, Imai T, Hirose M. 2006; A 13-week subchronic toxicity study of dietary administered morin in F344 rats. Food Chem Toxicol. 44:891–897. DOI: 10.1016/j.fct.2005.12.002. PMID: 16442199.
Article15. Caselli A, Cirri P, Santi A, Paoli P. 2016; Morin: a promising natural drug. Curr Med Chem. 23:774–791. DOI: 10.2174/0929867323666160106150821. PMID: 26018232.
Article16. Al-Numair KS, Chandramohan G, Alsaif MA, Veeramani C, El Newehy AS. 2014; Morin, a flavonoid, on lipid peroxidation and antioxidant status in experimental myocardial ischemic rats. Afr J Tradit Complement Altern Med. 11:14–20. DOI: 10.4314/ajtcam.v11i3.3. PMID: 25371558. PMCID: PMC4202414.
Article17. Rehman MU, Rashid SM, Rasool S, Shakeel S, Ahmad B, Ahmad SB, Madkhali H, Ganaie MA, Majid S, Bhat SA. 2019; Zingerone (4-(4-hydroxy-3-methylphenyl)butan-2-one) ameliorates renal function via controlling oxidative burst and inflammation in experimental diabetic nephropathy. Arch Physiol Biochem. 125:201–209. DOI: 10.1080/13813455.2018.1448422. PMID: 29537332.
Article18. Heeba GH, Mahmoud ME. 2014; Therapeutic potential of morin against liver fibrosis in rats: modulation of oxidative stress, cytokine production and nuclear factor kappa B. Environ Toxicol Pharmacol. 37:662–671. DOI: 10.1016/j.etap.2014.01.026. PMID: 24583409.
Article19. Tveden-Nyborg P, Bergmann TK, Lykkesfeldt J. 2018; Basic & Clinical Pharmacology & Toxicology policy for experimental and clinical studies. Basic Clin Pharmacol Toxicol. 123:233–235. DOI: 10.1111/bcpt.13059. PMID: 29931751.20. Botezelli JD, Cambri LT, Ghezzi AC, Dalia RA, Voltarelli FA, de Mello MA. 2012; Fructose-rich diet leads to reduced aerobic capacity and to liver injury in rats. Lipids Health Dis. 11:78. DOI: 10.1186/1476-511X-11-78. PMID: 22713601. PMCID: PMC3473252.
Article21. Ola MS, Aleisa AM, Al-Rejaie SS, Abuohashish HM, Parmar MY, Alhomida AS, Ahmed MM. 2014; Flavonoid, morin inhibits oxidative stress, inflammation and enhances neurotrophic support in the brain of streptozotocin-induced diabetic rats. Neurol Sci. 35:1003–1008. DOI: 10.1007/s10072-014-1628-5. PMID: 24413816.
Article22. Naderali EK, Williams G. 2003; Prolonged endothelial-dependent and -independent arterial dysfunction induced in the rat by short-term feeding with a high-fat, high-sucrose diet. Atherosclerosis. 166:253–259. DOI: 10.1016/S0021-9150(02)00367-2. PMID: 12535737.
Article23. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. 1985; Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 28:412–419. DOI: 10.1007/BF00280883. PMID: 3899825.24. Friedewald WT, Levy RI, Fredrickson DS. 1972; Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 18:499–502. DOI: 10.1093/clinchem/18.6.499. PMID: 4337382.
Article25. Ohkawa H, Ohishi N, Yagi K. 1979; Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 95:351–358. DOI: 10.1016/0003-2697(79)90738-3. PMID: 36810.
Article26. Sastry KV, Moudgal RP, Mohan J, Tyagi JS, Rao GS. 2002; Spectrophotometric determination of serum nitrite and nitrate by copper-cadmium alloy. Anal Biochem. 306:79–82. DOI: 10.1006/abio.2002.5676. PMID: 12069417.
Article27. Beutler E, Duron O, Kelly BM. 1963; Improved method for the determination of blood glutathione. J Lab Clin Med. 61:882–888. PMID: 13967893.28. Johnson RJ, Segal MS, Sautin Y, Nakagawa T, Feig DI, Kang DH, Gersch MS, Benner S, Sánchez-Lozada LG. 2007; Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am J Clin Nutr. 86:899–906. DOI: 10.1093/ajcn/86.4.899. PMID: 17921363.29. Ma Y, Ge A, Zhu W, Liu YN, Ji NF, Zha WJ, Zhang JX, Zeng XN, Huang M. 2016; Morin attenuates ovalbumin-induced airway inflammation by modulating oxidative stress-responsive MAPK signaling. Oxid Med Cell Longev. 2016:5843672. DOI: 10.1155/2016/5843672. PMID: 26783416. PMCID: PMC4691473.
Article30. Abuohashish HM, Al-Rejaie SS, Al-Hosaini KA, Parmar MY, Ahmed MM. 2013; Alleviating effects of morin against experimentally-induced diabetic osteopenia. Diabetol Metab Syndr. 5:5. DOI: 10.1186/1758-5996-5-5. PMID: 23384060. PMCID: PMC3582624.
Article31. Carrier A. 2017; Metabolic syndrome and oxidative stress: a complex relationship. Antioxid Redox Signal. 26:429–431. DOI: 10.1089/ars.2016.6929. PMID: 27796142.
Article32. McCracken E, Monaghan M, eenivasan S Sr. 2018; Pathophysiology of the metabolic syndrome. Clin Dermatol. 36:14–20. DOI: 10.1016/j.clindermatol.2017.09.004. PMID: 29241747.
Article33. Wu TW, Zeng LH, Wu J, Fung KP. 1993; Morin hydrate is a plant-derived and antioxidant-based hepatoprotector. Life Sci. 53:PL213–PL218. DOI: 10.1016/0024-3205(93)90266-6. PMID: 8366767.
Article34. Morales J, Günther G, Zanocco AL, Lemp E. 2012; Singlet oxygen reactions with flavonoids. A theoretical-experimental study. PLoS One. 7:e40548. DOI: 10.1371/journal.pone.0040548. PMID: 22802966. PMCID: PMC3393665.35. Degenhardt K, Mathew R, Beaudoin B, Bray K, Anderson D, Chen G, Mukherjee C, Shi Y, Gélinas C, Fan Y, Nelson DA, Jin S, White E. 2006; Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell. 10:51–64. DOI: 10.1016/j.ccr.2006.06.001. PMID: 16843265. PMCID: PMC2857533.
Article36. Liang C, E X, Jung JU. 2008; Downregulation of autophagy by herpesvirus Bcl-2 homologs. Autophagy. 4:268–272. DOI: 10.4161/auto.5210. PMID: 17993780.
Article37. Lim J, Ali ZM, Sanders RA, Snyder AC, Eells JT, Henshel DS, Watkins JB 3rd. 2009; Effects of low-level light therapy on hepatic antioxidant defense in acute and chronic diabetic rats. J Biochem Mol Toxicol. 23:1–8. DOI: 10.1002/jbt.20257. PMID: 19202557.
Article38. MadanKumar P, NaveenKumar P, Devaraj H, NiranjaliDevaraj S. 2015; Morin, a dietary flavonoid, exhibits anti-fibrotic effect and induces apoptosis of activated hepatic stellate cells by suppressing canonical NF-κB signaling. Biochimie. 110:107–118. DOI: 10.1016/j.biochi.2015.01.002. PMID: 25577997.
Article39. Uchiyama S, Shimizu T, Shirasawa T. 2006; CuZn-SOD deficiency causes ApoB degradation and induces hepatic lipid accumulation by impaired lipoprotein secretion in mice. J Biol Chem. 281:31713–31719. DOI: 10.1016/S0021-9258(19)84085-1. PMID: 16921198.
Article40. K V A, Madhana RM, Kasala ER, Samudrala PK, Lahkar M, Gogoi R. 2016; Morin hydrate mitigates cisplatin-induced renal and hepatic injury by impeding oxidative/nitrosative stress and inflammation in mice. J Biochem Mol Toxicol. 30:571–579. DOI: 10.1002/jbt.21817. PMID: 27111570.
Article41. Kang DG, Moon MK, Sohn EJ, Lee DH, Lee HS. 2004; Effects of morin on blood pressure and metabolic changes in fructose-induced hypertensive rats. Biol Pharm Bull. 27:1779–1783. DOI: 10.1248/bpb.27.1779. PMID: 15516722.
Article42. Vanitha P, Uma C, Suganya N, Bhakkiyalakshmi E, Suriyanarayanan S, Gunasekaran P, Sivasubramanian S, Ramkumar KM. 2014; Modulatory effects of morin on hyperglycemia by attenuating the hepatic key enzymes of carbohydrate metabolism and β-cell function in streptozotocin-induced diabetic rats. Environ Toxicol Pharmacol. 37:326–335. DOI: 10.1016/j.etap.2013.11.017. PMID: 24384280.
Article43. Paoli P, Cirri P, Caselli A, Ranaldi F, Bruschi G, Santi A, Camici G. 2013; The insulin-mimetic effect of Morin: a promising molecule in diabetes treatment. Biochim Biophys Acta. 1830:3102–3111. DOI: 10.1016/j.bbagen.2013.01.017. PMID: 23352912.
Article44. Ghaffari MA, Mojab S. 2007; Influence of flavonols as in vitro on low density lipoprotein glycation. Iran Biomed J. 11:185–191. PMID: 18051779.45. Naowaboot J, Wannasiri S, Pannangpetch P. 2016; Morin attenuates hepatic insulin resistance in high-fat-diet-induced obese mice. J Physiol Biochem. 72:269–280. DOI: 10.1007/s13105-016-0477-5. PMID: 26976296.
Article46. Zhang X, Han X, Zhang P, Zhou T, Chen Y, Jin J, Ma X. 2019; Morin attenuates oxidized low-density lipoprotein-mediated injury by inducing autophagy via activating AMPK signalling in HUVECs. Clin Exp Pharmacol Physiol. 46:1053–1060. DOI: 10.1111/1440-1681.13160. PMID: 31407376.47. Khitan Z, Kim DH. 2013; Fructose: a key factor in the development of metabolic syndrome and hypertension. J Nutr Metab. 2013:682673. DOI: 10.1155/2013/682673. PMID: 23762544. PMCID: PMC3677638.
Article48. Masai M, Fujioka Y, Fujiwara M, Morimoto S, Miyoshi A, Suzuki H, Iwasaki T. 2001; Activation of Na+/H+ exchanger is associated with hyperinsulinemia in borderline hypertensive rats. Eur J Clin Invest. 31:193–200. DOI: 10.1046/j.1365-2362.2001.00008.x. PMID: 11264645.
Article49. Prahalathan P, Kumar S, Raja B. 2012; Morin attenuates blood pressure and oxidative stress in deoxycorticosterone acetate-salt hypertensive rats: a biochemical and histopathological evaluation. Metabolism. 61:1087–1099. DOI: 10.1016/j.metabol.2011.12.012. PMID: 22386933.
Article50. Prahalathan P, Saravanakumar M, Raja B. 2012; The flavonoid morin restores blood pressure and lipid metabolism in DOCA-salt hypertensive rats. Redox Rep. 17:167–175. DOI: 10.1179/1351000212Y.0000000015. PMID: 22781105. PMCID: PMC6837552.
Article51. Merwid-Ląd A, Trocha M, Chlebda-Sieragowska E, Sozański T, Szandruk M, Magdalan J, Ksiądzyna D, Pieśniewska M, Fereniec-Gołębiewska L, Kwiatkowska J, Szeląg A. 2014; The impact of morin, a natural flavonoid, on cyclophosphamide-induced changes in the oxidative stress parameters in rat livers. Adv Clin Exp Med. 23:505–509. DOI: 10.17219/acem/37213. PMID: 25166433.52. Hajiaghaalipour F, Khalilpourfarshbafi M, Arya A. 2015; Modulation of glucose transporter protein by dietary flavonoids in type 2 diabetes mellitus. Int J Biol Sci. 11:508–524. DOI: 10.7150/ijbs.11241. PMID: 25892959. PMCID: PMC4400383.
Article53. Cordero-Herrera I, Martín MA, Bravo L, Goya L, Ramos S. 2013; Cocoa flavonoids improve insulin signalling and modulate glucose production via AKT and AMPK in HepG2 cells. Mol Nutr Food Res. 57:974–985. DOI: 10.1002/mnfr.201200500. PMID: 23456781.
Article54. Cordero-Herrera I, Martín MÁ, Goya L, Ramos S. 2014; Cocoa flavonoids attenuate high glucose-induced insulin signalling blockade and modulate glucose uptake and production in human HepG2 cells. Food Chem Toxicol. 64:10–19. DOI: 10.1016/j.fct.2013.11.014. PMID: 24262486.
Article55. Arab HH, Al-Shorbagy MY, Abdallah DM, Nassar NN. 2014; Telmisartan attenuates colon inflammation, oxidative perturbations and apoptosis in a rat model of experimental inflammatory bowel disease. PLoS One. 9:e97193. DOI: 10.1371/journal.pone.0097193. PMID: 24831514. PMCID: PMC4022743.
Article56. Lee HS, Jung KH, Park IS, Kwon SW, Lee DH, Hong SS. 2009; Protective effect of morin on dimethylnitrosamine-induced hepatic fibrosis in rats. Dig Dis Sci. 54:782–788. DOI: 10.1007/s10620-008-0404-x. PMID: 18629640.
Article57. Lee HS, Jung KH, Hong SW, Park IS, Lee C, Han HK, Lee DH, Hong SS. 2008; Morin protects acute liver damage by carbon tetrachloride (CCl(4)) in rat. Arch Pharm Res. 31:1160–1165. DOI: 10.1007/s12272-001-1283-5. PMID: 18806959.
Article58. Kim JM, Lee EK, Park G, Kim MK, Yokozawa T, Yu BP, Chung HY. 2010; Morin modulates the oxidative stress-induced NF-kappaB pathway through its anti-oxidant activity. Free Radic Res. 44:454–461. DOI: 10.3109/10715761003610737. PMID: 20187708.59. Gupta SC, Tyagi AK, Deshmukh-Taskar P, Hinojosa M, Prasad S, Aggarwal BB. 2014; Downregulation of tumor necrosis factor and other proinflammatory biomarkers by polyphenols. Arch Biochem Biophys. 559:91–99. DOI: 10.1016/j.abb.2014.06.006. PMID: 24946050.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Morin ameliorates myocardial injury in diabetic rats via modulation of inflammatory pathways
- Activation of the renin-angiotensin system in high fructose-induced metabolic syndrome
- Recent update on pathogenesis of nonalcoholic fatty liver disease
- Protective effect of β-sitosterol against high-fructose diet-induced oxidative stress, and hepatorenal derangements in growing female sprague-dawley rats
- The Effects of Hydro-Alcoholic Extract of Fenugreek Seeds on the Lipid Profile and Oxidative Stress in Fructose-Fed Rats