J Korean Neurosurg Soc.
2020 Nov;63(6):707-716. 10.3340/jkns.2019.0255.
Survival Association and Cell Cycle Effects of B7H3 in Neuroblastoma
- Affiliations
-
- 1Department of Neurosurgery, Huashan Hospital, Fudan University, Shanghai, P.R. China
- 2Institutes of Biomedical Sciences, Fudan University, Shanghai, P.R. China
- 3Department of Pediatric Surgery, Children’s Hospital of Fudan University, Shanghai, China
- 4Key Laboratory of Neonatal Disease, Ministry of Health, Shanghai, China
- 5Department of Neurosurgery, University of Minnesota, Minneapolis, MN, USA
- KMID: 2508600
- DOI: http://doi.org/10.3340/jkns.2019.0255
Abstract
Objective
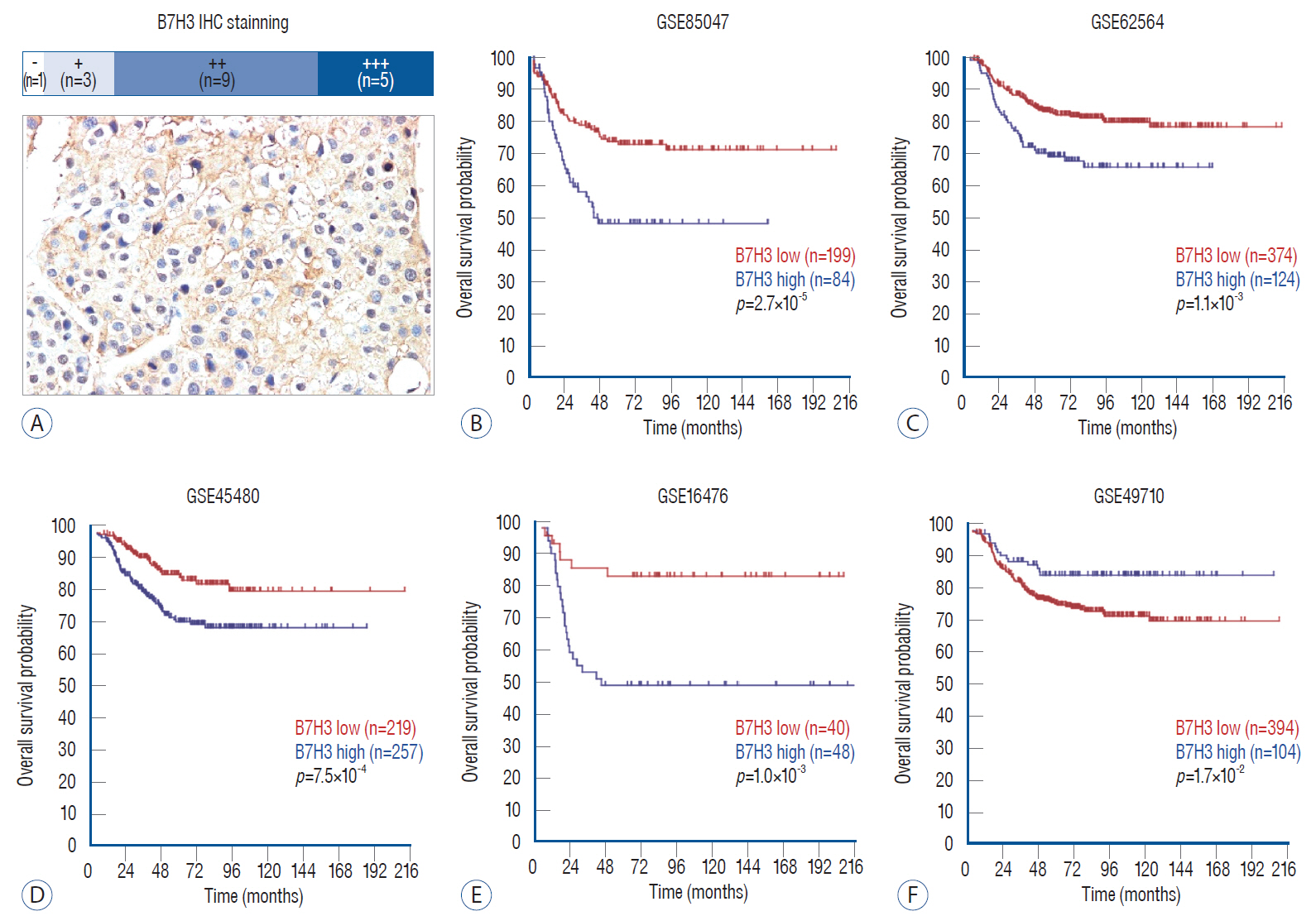
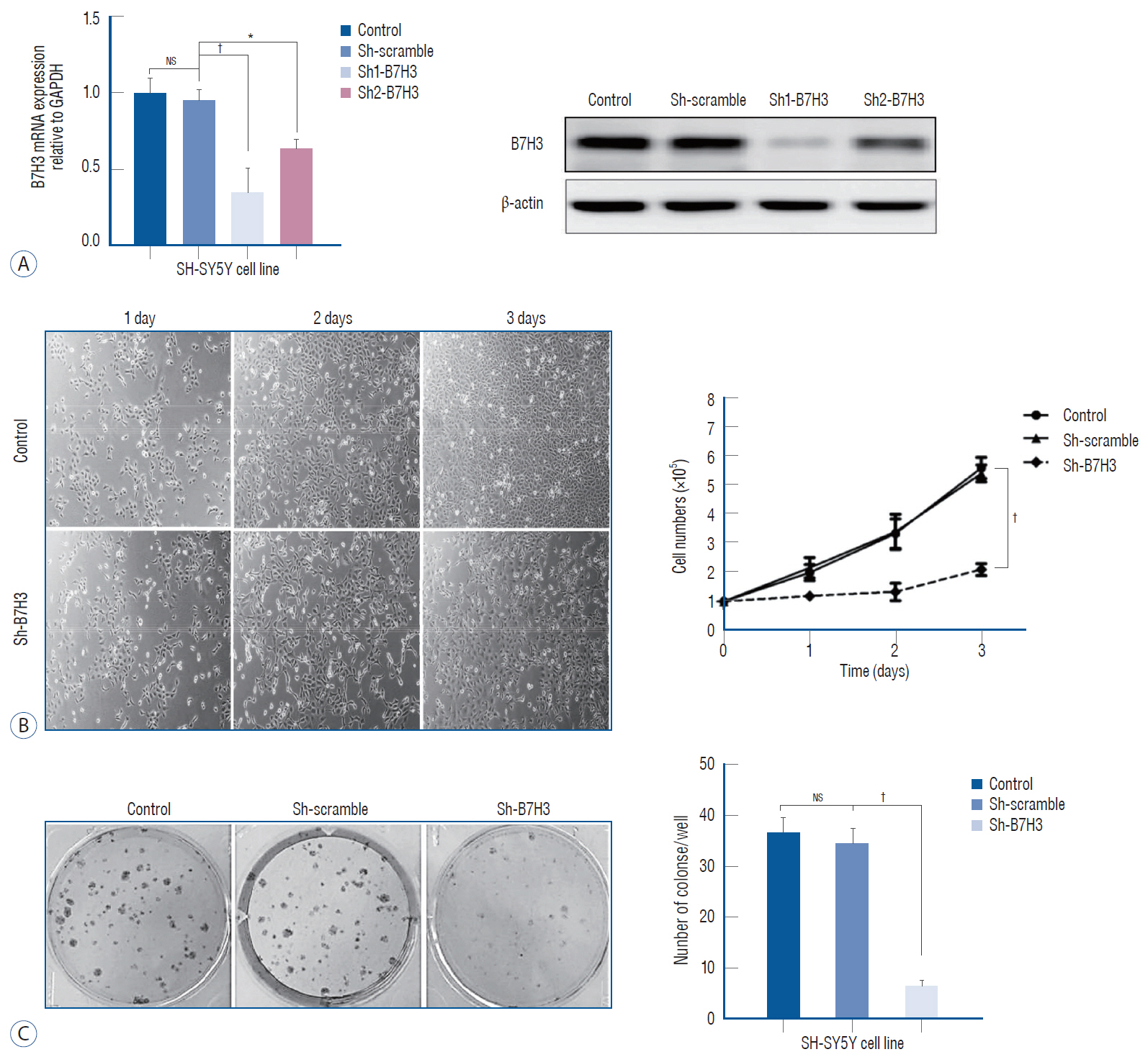
: The function of B7H3, a member of the B7 family of proteins, in neuroblastoma (NB) remains poorly characterized. Here we examine the expression pattern of B7H3 in clinical NB specimens and characterize the phenotype of B7H3 knock-down in NB cell line.
Methods
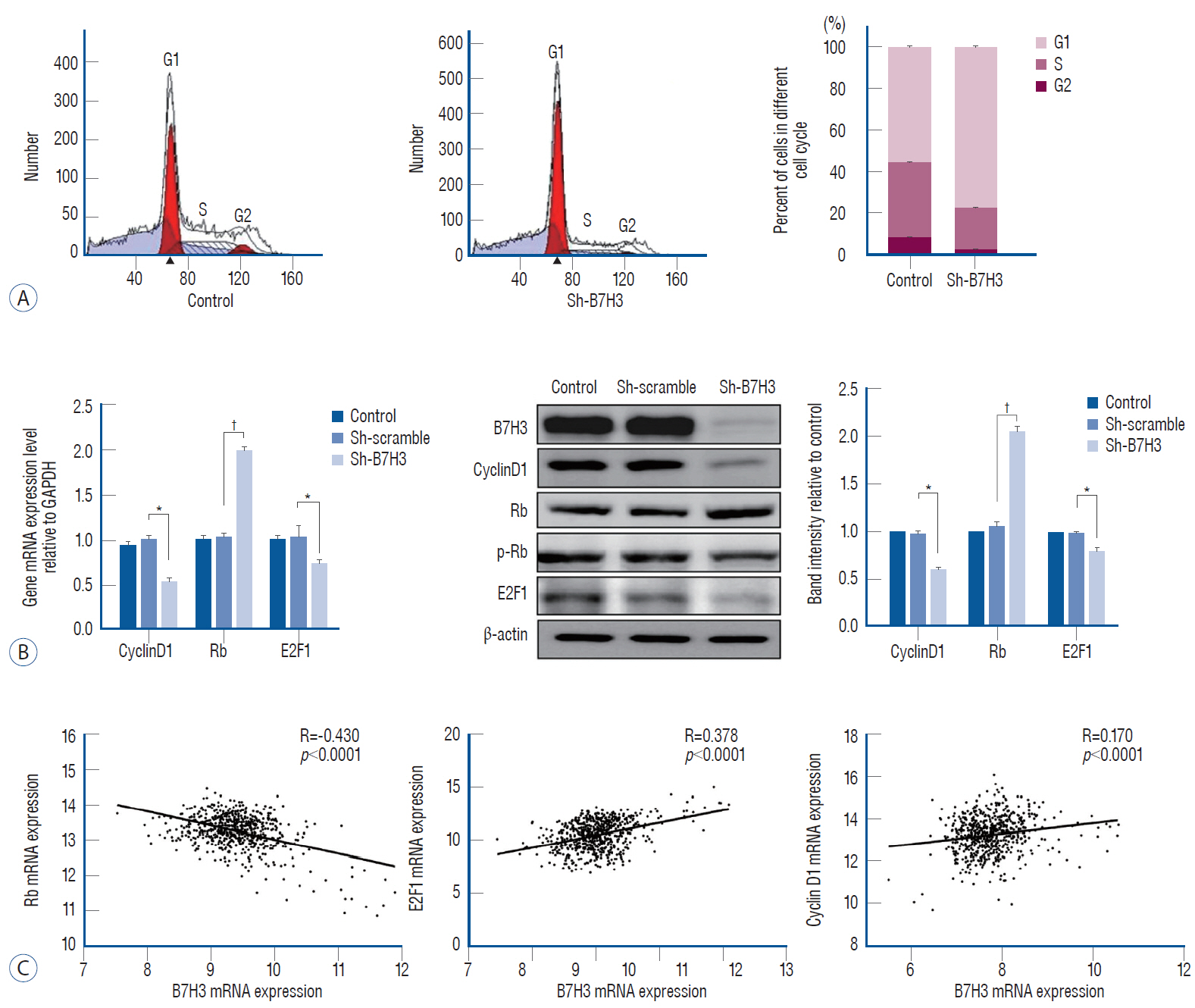
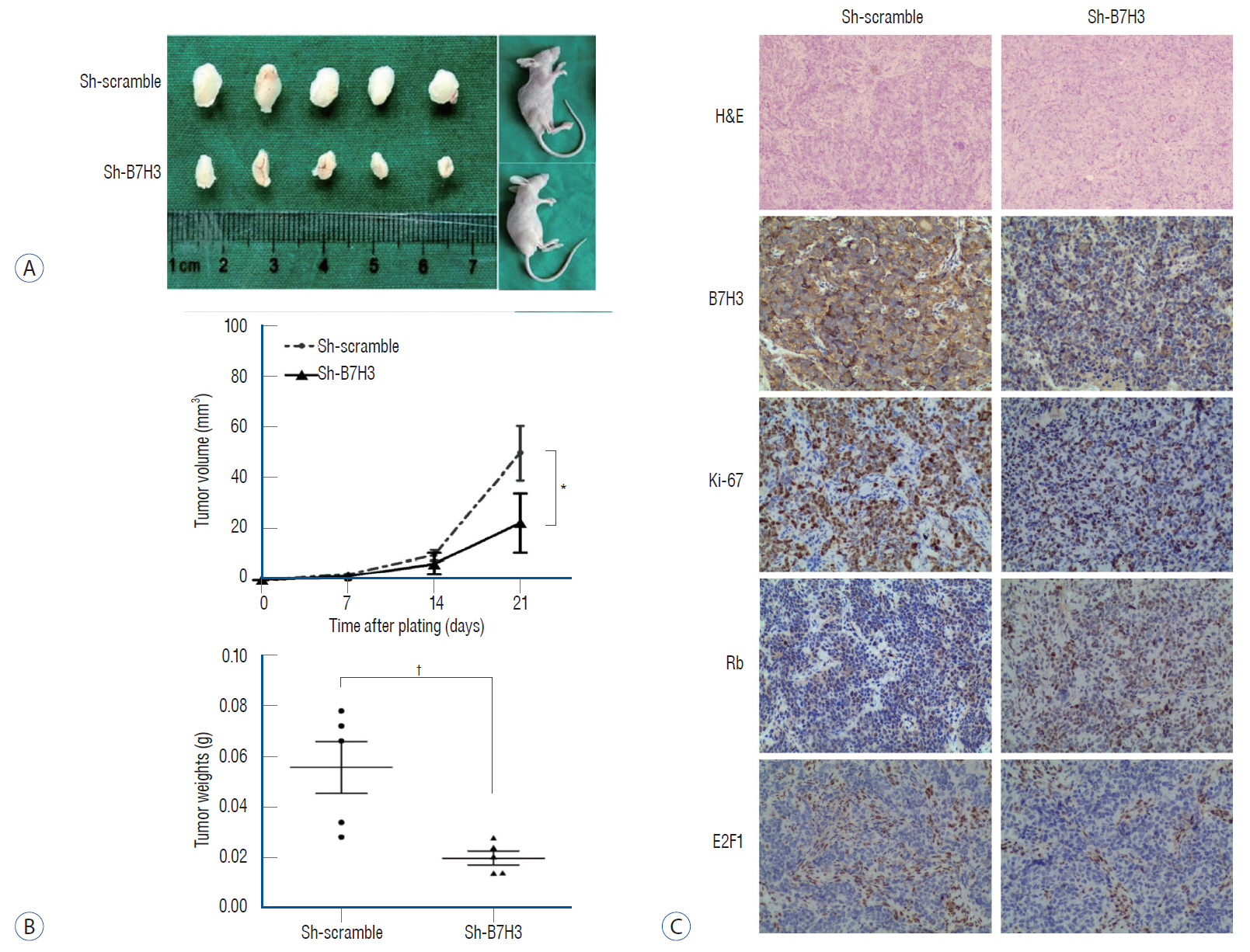
: Immunohistochemical (IHC) staining was carried out to assess the expression of B7H3 in clinical NB specimens. Survival association was analyzed using five Gene Expression Omnibus (GEO) datasets (GSE85047, GSE45480, GSE62564, GSE16476, GSE49710). Clonogenic survival and flow cytometry were performed after B7H3 knockdown to assess the cellular proliferation and cell survival in vitro. Impact of B7H3 silencing on NB growth was examined in vivo using the SH-SY5Y xenograft model.
Results
: On IHC staining, B7H3 was widely expressed in clinical NB specimens. Analysis of the transcriptional profiles of five GEO datasets clinically annotated NB specimens revealed that decreased B7H3 expression was associated with improved overall survival. B7H3 knockdown suppressed the proliferation of the SH-SY5Y NB model in vitro and in vivo. Cell cycle analysis revealed that B7H3 silencing induced G1/S arrest. This arrest was associated with the suppression of E2F1 expression and induction of Rb expression.
Conclusion
: Our results demonstrate that B7H3 expression correlate with clinical survival in NB patients. Preliminary studies suggest that B7H3 may mediate the G1/S transition.
Keyword
Figure
Reference
-
References
1. Bertoli C, Skotheim JM, de Bruin RA. Control of cell cycle transcription during G1 and S phases. Nat Rev Mol Cell Biol. 14:518–528. 2013.
Article2. Castriconi R, Dondero A, Augugliaro R, Cantoni C, Carnemolla B, Sementa AR, et al. Identification of 4Ig-B7-H3 as a neuroblastoma-associated molecule that exerts a protective role from an NK cell-mediated lysis. Proc Natl Acad Sci U S A. 101:12640–12645. 2004.
Article3. Dong P, Xiong Y, Yue J, Hanley SJB, Watari H. B7H3 as a promoter of metastasis and promising therapeutic target. Front Oncol. 8:264. 2018.
Article4. Du H, Hirabayashi K, Ahn S, Kren NP, Montgomery SA, Wang X, et al. Antitumor responses in the absence of toxicity in solid tumors by targeting B7-H3 via chimeric antigen receptor T cells. Cancer Cell. 35:221–237.e8. 2019.
Article5. Eleveld TF, Oldridge DA, Bernard V, Koster J, Colmet Daage L, Diskin SJ, et al. Relapsed neuroblastomas show frequent RAS-MAPK pathway mutations. Nat Genet. 47:864–871. 2015.
Article6. Jeon H, Vigdorovich V, Garrett-Thomson SC, Janakiram M, Ramagopal UA, Abadi YM, et al. Structure and cancer immunotherapy of the B7 family member B7x. Cell Rep. 9:1089–1098. 2014.
Article7. John RR, Malathi N, Ravindran C, Anandan S. Mini review: multifaceted role played by cyclin D1 in tumor behavior. Indian J Dent Res. 28:187–192. 2017.
Article8. Lim S, Liu H, Madeira da Silva L, Arora R, Liu Z, Phillips JB, et al. Immunoregulatory protein B7-H3 reprograms glucose metabolism in cancer cells by ROS-mediated stabilization of HIF1α. Cancer Res. 76:2231–2242. 2016.
Article9. Liu H, Tekle C, Chen YW, Kristian A, Zhao Y, Zhou M, et al. B7-H3 silencing increases paclitaxel sensitivity by abrogating Jak2/Stat3 phosphorylation. Mol Cancer Ther. 10:960–971. 2011.
Article10. Luo D, Xiao H, Dong J, Li Y, Feng G, Cui M, et al. B7-H3 regulates lipid metabolism of lung cancer through SREBP1-mediated expression of FASN. Biochem Biophys Res Commun. 482:1246–1251. 2017.
Article11. Maris JM. Recent advances in neuroblastoma. N Engl J Med. 362:2202–2211. 2010.
Article12. McNair C, Xu K, Mandigo AC, Benelli M, Leiby B, Rodrigues D, et al. Differential impact of RB status on E2F1 reprogramming in human cancer. J Clin Invest. 128:341–358. 2018.
Article13. Otto T, Sicinski P. Cell cycle proteins as promising targets in cancer therapy. Nat Rev Cancer. 17:93–115. 2017.
Article14. Peifer M, Hertwig F, Roels F, Dreidax D, Gartlgruber M, Menon R, et al. Telomerase activation by genomic rearrangements in high-risk neuroblastoma. Nature. 526:700–704. 2015.
Article15. Seaman S, Zhu Z, Saha S, Zhang XM, Yang MY, Hilton MB, et al. Eradication of tumors through simultaneous ablation of CD276/B7-H3-positive tumor cells and tumor vasculature. Cancer Cell. 31:501–515. e8. 2017.
Article16. Tan WQ, Chen G, Ye M, Jia B. Artemether regulates chemosensitivity to doxorubicin via regulation of B7-H3 in human neuroblastoma cells. Med Sci Monit. 23:4252–4259. 2017.
Article17. Tekle C, Nygren MK, Chen YW, Dybsjord I, Nesland JM, Maelandsmo GM, et al. B7-H3 contributes to the metastatic capacity of melanoma cells by modulation of known metastasis-associated genes. Int J Cancer. 130:2282–2290. 2012.
Article18. Wu J, Wang F, Liu X, Zhang T, Liu F, Ge X, et al. Correlation of IDH1 and B7H3 expression with prognosis of CRC patients. Eur J Surg Oncol. 44:1254–1260. 2018.19. Yuan H, Wei X, Zhang G, Li C, Zhang X, Hou J. B7-H3 over expression in prostate cancer promotes tumor cell progression. J Urol. 186:1093–1099. 2011.
Article20. Zhang J, Wang J, Marzese DM, Wang X, Yang Z, Li C, et al. B7H3 regulates differentiation and serves as a potential biomarker and theranostic target for human glioblastoma. Lab Invest. 99:1117–1129. 2019.
Article21. Zhang T, Jiang B, Zou ST, Liu F, Hua D. Overexpression of B7-H3 augments anti-apoptosis of colorectal cancer cells by Jak2-STAT3. World J Gastroenterol. 21:1804–1813. 2015.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Palliative effect of (131)I-MIBG in relapsed neuroblastoma after autologous peripheral blood stem cell transplantation
- Genistein Induces G2/M Cell Cycle Arrest and Apoptosis in Rat Neuroblastoma B35 Cells; Involvement of p21(waf1/cip1), Bax and Bcl-2
- Effect on Cell Cycle Progression by N-Myc Knockdown in SK-N-BE(2) Neuroblastoma Cell Line and Cytotoxicity with STI-571 Compound
- Antitumor Effects of Arsenic Trioxide on Neuroblastoma
- Antiproliferative and Cytotoxic Effects of Resveratrol in Mitochondria-Mediated Apoptosis in Rat B103 Neuroblastoma Cells