Cancer Res Treat.
2020 Oct;52(4):1288-1290. 10.4143/crt.2020.278.
EGFR C797S as a Resistance Mechanism of Lazertinib in Non-small Cell Lung Cancer with EGFR T790M Mutation
- Affiliations
-
- 1Division of Hematology-Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 2Samsung Biomedical Research Institute, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- KMID: 2507954
- DOI: http://doi.org/10.4143/crt.2020.278
Abstract
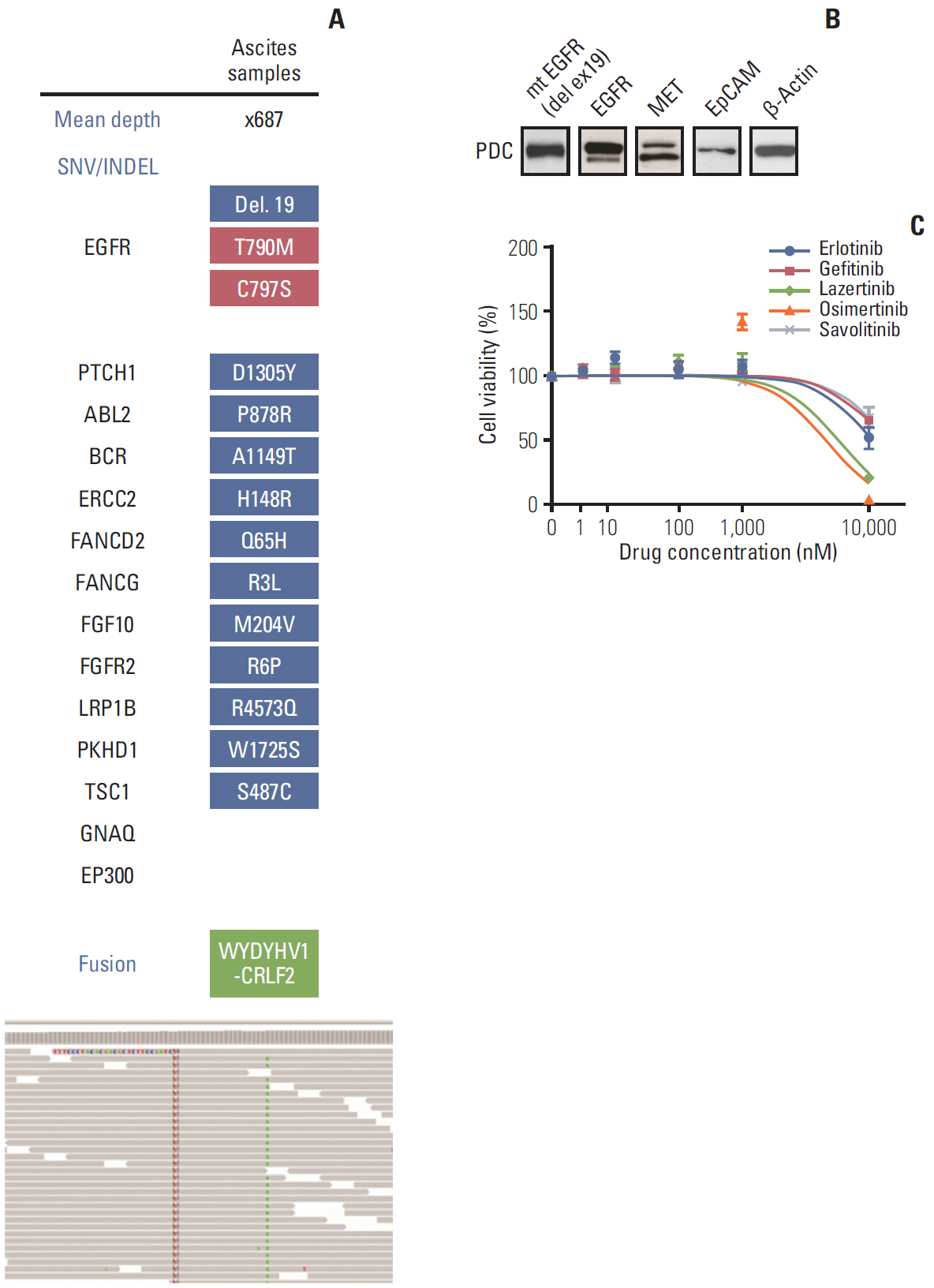
- The non-small cell lung cancer with activating epidermal growth factor receptor (EGFR) mutation eventually acquires resistant to either first or second-generation EGFR tyrosine kinase inhibitor (TKI). As the following option, targeting EGFR T790M with third-generation EGFR TKI is now established as a standard treatment option. In this study, we are reporting the first case of resistance mechanism to the novel third-generation EGFR TKI, lazertinib, which showed promising clinical efficacy in phase 1-2 study. The patients showed resistance to the treatment by acquiring the additional EGFR C797S mutation in cis which is also confirmed from the patient-derived cell lines.
Keyword
Figure
Reference
-
References
1. Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011; 3:75ra26.
Article2. Ahn MJ, Han JY, Lee KH, Kim SW, Kim DW, Lee YG, et al. Lazertinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: results from the dose escalation and dose expansion parts of a first-in-human, open-label, multicentre, phase 1-2 study. Lancet Oncol. 2019; 20:1681–90.
Article3. Shin HT, Choi YL, Yun JW, Kim NK, Kim SY, Jeon HJ, et al. Prevalence and detection of low-allele-fraction variants in clinical cancer samples. Nat Commun. 2017; 8:1377.
Article4. Oxnard GR, Hu Y, Mileham KF, Husain H, Costa DB, Tracy P, et al. Assessment of resistance mechanisms and clinical implications in patients with EGFR T790M-positive lung cancer and acquired resistance to osimertinib. JAMA Oncol. 2018; 4:1527–34.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chronicles of EGFR Tyrosine Kinase Inhibitors: Targeting EGFR C797S Containing Triple Mutations
- Acquired Resistance Mechanism of EGFR Kinase Domain Duplication to EGFR TKIs in Non–Small Cell Lung Cancer
- Lazertinib: on the Way to Its Throne
- Should We Perform Repeated Re-biopsy for the Detection of T790M Mutation?
- Combining Erlotinib with Cytotoxic Chemotherapy May Overcome Resistance Caused by T790M Mutation of EGFR Gene in Non-Small Cell Lung Carcinoma