Int J Stem Cells.
2020 Jul;13(2):202-211. 10.15283/ijsc20033.
Overexpression of p21 Has Inhibitory Effect on Human Hematopoiesis by Blocking Generation of CD43+ Cells via Cell-Cycle Regulation
- Affiliations
-
- 1Research Center for Stem Cell Therapies, Institute of Blood Transfusion, Chinese Academy of Medical Sciences & Peking Union Medical College (CAMS & PUMC), Chengdu, China
- 2State Key Laboratory of Biotherapy, Sichuan University, Chengdu, China
- 3State Key Laboratory of Experimental Hematology, CAMS & PUMC, Tianjin, China
- KMID: 2504333
- DOI: http://doi.org/10.15283/ijsc20033
Abstract
- Background and Objectives
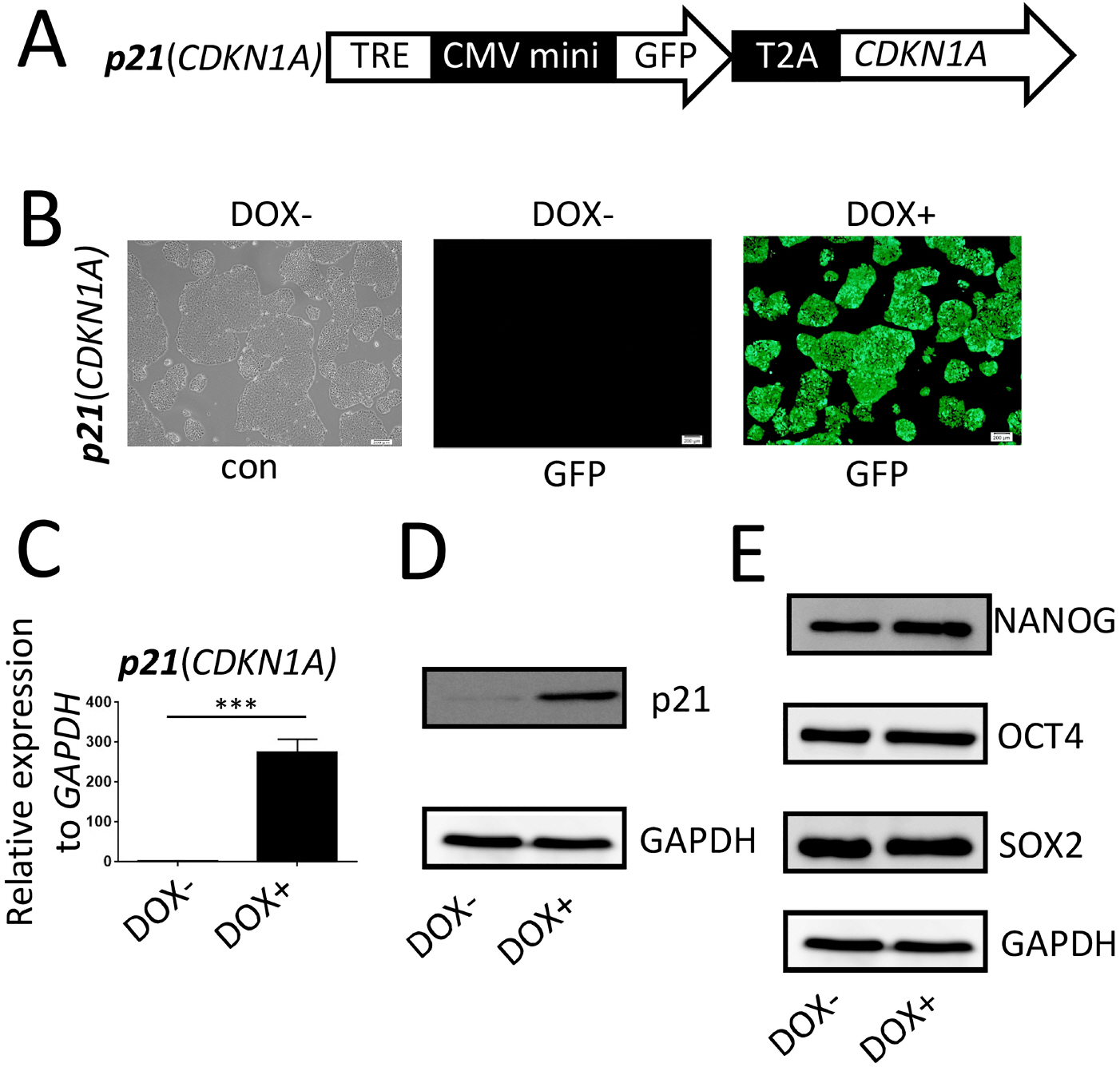
p21, an important member of the Cip/Kip family, is involved in inhibitory effects of RUNX1b overexpression during the early stage of human hematopoiesis.
Methods and Results
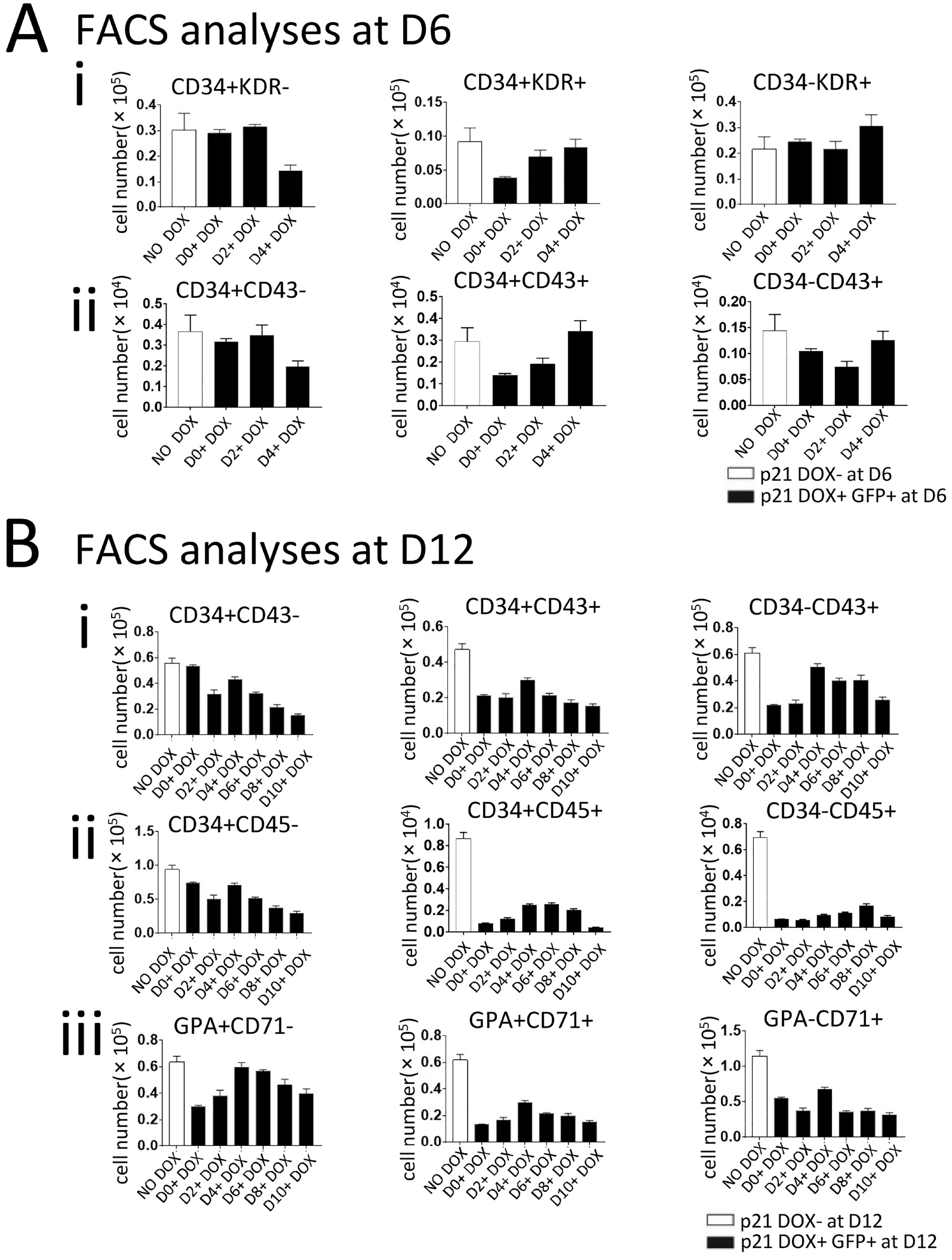
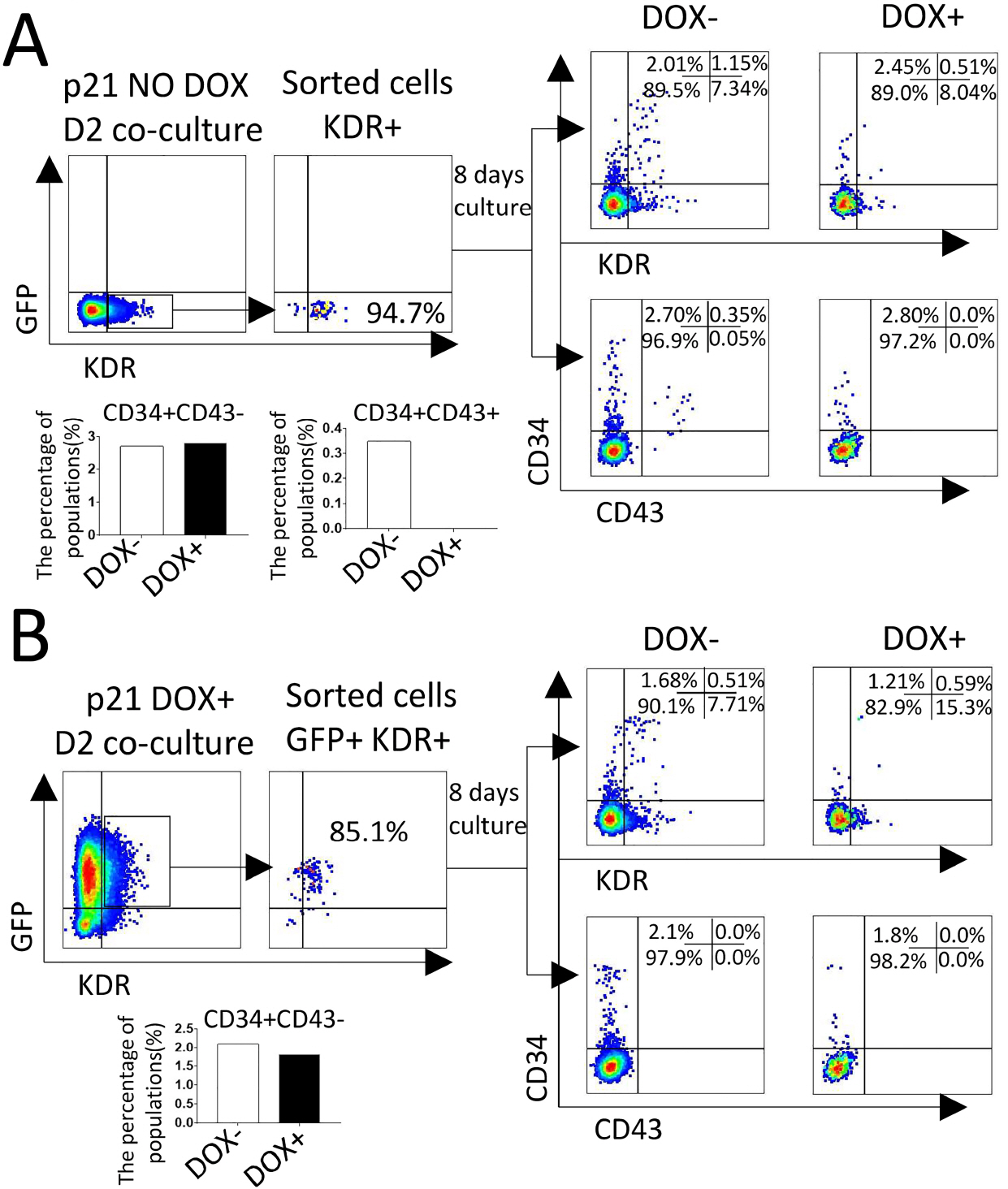
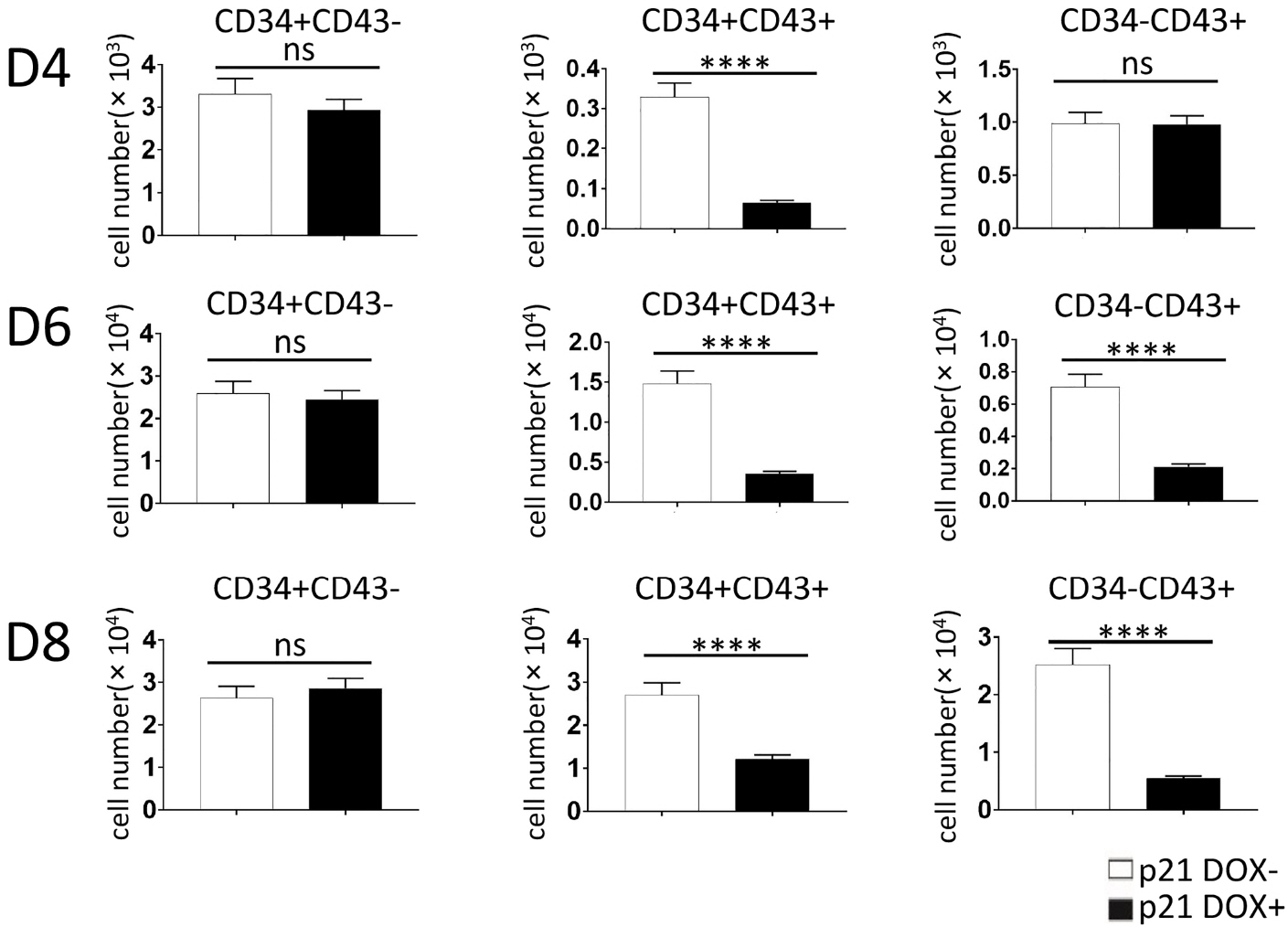
We established a human embryonic stem cell (hESC) line with inducible expression of p21 (p21/hESCs). Overexpression of p21 did not influence either mesoderm induction or emergence of CD34+ cells, but it significantly decreased the production of CD43+ cells and changed the expression profile of hematopoiesis-related factors, leading to the negative effects of p21 on hematopoiesis.
Conclusions
In RUNX1b/hESC co-cultures when RUNX1b was induced from D0, perturbation of the cell cycle caused by upregulation of p21 probably prevented the appearance of CD43+ cells, but not CD34+ cells. The mechanisms via which CD34+ cells are blocked by RUNX1b overexpression remain to be elucidated.
Keyword
Figure
Reference
-
References
1. Palis J, Yoder MC. 2001; Yolk-sac hematopoiesis: the first blood cells of mouse and man. Exp Hematol. 29:927–936. DOI: 10.1016/S0301-472X(01)00669-5.
Article2. Medvinsky A, Dzierzak E. 1996; Definitive hematopoiesis is autonomously initiated by the AGM region. Cell. 86:897–906. DOI: 10.1016/S0092-8674(00)80165-8.
Article3. Rhodes KE, Gekas C, Wang Y, Lux CT, Francis CS, Chan DN, Conway S, Orkin SH, Yoder MC, Mikkola HK. 2008; The emergence of hematopoietic stem cells is initiated in the placental vasculature in the absence of circulation. Cell Stem Cell. 2:252–263. DOI: 10.1016/j.stem.2008.01.001. PMID: 18371450. PMCID: PMC2888040.
Article4. Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. 1998; Embryonic stem cell lines derived from human blastocysts. Science. 282:1145–1147. DOI: 10.1126/science.282.5391.1145. PMID: 9804556.
Article5. Ma F, Wang D, Hanada S, Ebihara Y, Kawasaki H, Zaike Y, Heike T, Nakahata T, Tsuji K. 2007; Novel method for efficient production of multipotential hematopoietic progenitors from human embryonic stem cells. Int J Hematol. 85:371–379. DOI: 10.1532/IJH97.06203. PMID: 17562610.
Article6. Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. 2007; Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 131:861–872. DOI: 10.1016/j.cell.2007.11.019. PMID: 18035408.
Article7. Ma F, Ebihara Y, Umeda K, Sakai H, Hanada S, Zhang H, Zaike Y, Tsuchida E, Nakahata T, Nakauchi H, Tsuji K. 2008; Generation of functional erythrocytes from human embryonic stem cell-derived definitive hematopoiesis. Proc Natl Acad Sci U S A. 105:13087–13092. DOI: 10.1073/pnas.0802220105. PMID: 18755895. PMCID: PMC2526552.
Article8. Palis J. 2016; Hematopoietic stem cell-independent hematopoiesis: emergence of erythroid, megakaryocyte, and myeloid potential in the mammalian embryo. FEBS Lett. 590:3965–3974. DOI: 10.1002/1873-3468.12459. PMID: 27790707.
Article9. Han X, Liu J. 2017; Cell cycle-independent roles of p19INK4d in human terminal erythropoiesis. Chin J Cancer. 36:22. DOI: 10.1186/s40880-017-0189-4. PMID: 28228163. PMCID: PMC5322582.
Article10. Chen B, Teng J, Liu H, Pan X, Zhou Y, Huang S, Lai M, Bian G, Mao B, Sun W, Zhou Q, Yang S, Nakahata T, Ma F. 2017; Inducible overexpression of RUNX1b/c in human embryonic stem cells blocks early hematopoiesis from mesoderm. J Mol Cell Biol. 9:262–273. DOI: 10.1093/jmcb/mjx032. PMID: 28992293.
Article11. Xiong Y, Hannon GJ, Zhang H, Casso D, Kobayashi R, Beach D. 1993; p21 is a universal inhibitor of cyclin kinases. Nature. 366:701–704. DOI: 10.1038/366701a0. PMID: 8259214.
Article12. Ouellet S, Vigneault F, Lessard M, Leclerc S, Drouin R, Guérin SL. 2006; Transcriptional regulation of the cyclin-dependent kinase inhibitor 1A (p21) gene by NFI in proliferating human cells. Nucleic Acids Res. 34:6472–6487. DOI: 10.1093/nar/gkl861. PMID: 17130157. PMCID: PMC1702497.
Article13. Stier S, Cheng T, Forkert R, Lutz C, Dombkowski DM, Zhang JL, Scadden DT. 2003; Ex vivo targeting of p21Cip1/Waf1 permits relative expansion of human hematopoietic stem cells. Blood. 102:1260–1266. DOI: 10.1182/blood-2002-10-3053. PMID: 12702511.
Article14. Matsumura I, Ishikawa J, Nakajima K, Oritani K, Tomiyama Y, Miyagawa J, Kato T, Miyazaki H, Matsuzawa Y, Kanakura Y. 1997; Thrombopoietin-induced differentiation of a human megakaryoblastic leukemia cell line, CMK, involves transcriptional activation of p21(WAF1/Cip1) by STAT5. Mol Cell Biol. 17:2933–2943. DOI: 10.1128/MCB.17.5.2933. PMID: 9111365. PMCID: PMC232145.
Article15. Eilken HM, Nishikawa S, Schroeder T. 2009; Continuous single-cell imaging of blood generation from haemogenic endothelium. Nature. 457:896–900. DOI: 10.1038/nature07760. PMID: 19212410.
Article16. Mao B, Huang S, Lu X, Sun W, Zhou Y, Pan X, Yu J, Lai M, Chen B, Zhou Q, Mao S, Bian G, Zhou J, Nakahata T, Ma F. 2016; Early development of definitive erythroblasts from human pluripotent stem cells defined by expression of glycophorin A/CD235a, CD34, and CD36. Stem Cell Reports. 7:869–883. DOI: 10.1016/j.stemcr.2016.09.002. PMID: 27720903. PMCID: PMC5106477.
Article17. Chang J, Sun W, Zeng J, Xue Y, Zhang Y, Pan X, Zhou Y, Lai M, Bian G, Zhou Q, Liu J, Chen B, Guo F, Ma F. 2019; Establishment of an in vitro system based on AGM-S3 co-culture for screening traditional herbal medicines that stimulate hematopoiesis. J Ethnopharmacol. 240:111938. DOI: 10.1016/j.jep.2019.111938. PMID: 31077780.
Article18. Zhou Y, Zhang Y, Chen B, Dong Y, Zhang Y, Mao B, Pan X, Lai M, Chen Y, Bian G, Zhou Q, Nakahata T, Zhou J, Wu M, Ma F. 2019; Overexpression of GATA2 enhances development and maintenance of human embryonic stem cell-derived hematopoietic stem cell-like progenitors. Stem Cell Reports. 13:31–47. DOI: 10.1016/j.stemcr.2019.05.007. PMID: 31178416. PMCID: PMC6626852.
Article19. Palpant NJ, Wang Y, Hadland B, Zaunbrecher RJ, Redd M, Jones D, Pabon L, Jain R, Epstein J, Ruzzo WL, Zheng Y, Bernstein I, Margolin A, Murry CE. 2017; Chromatin and transcriptional analysis of mesoderm progenitor cells identifies HOPX as a regulator of primitive hematopoiesis. Cell Rep. 20:1597–1608. DOI: 10.1016/j.celrep.2017.07.067. PMID: 28813672. PMCID: PMC5576510.
Article20. Yuan X, Braunstein EM, Ye Z, Liu CF, Chen G, Zou J, Cheng L, Brodsky RA. 2013; Generation of glycosylphosphatidylinositol anchor protein-deficient blood cells from human induced pluripotent stem cells. Stem Cells Transl Med. 2:819–829. DOI: 10.5966/sctm.2013-0069. PMID: 24113066. PMCID: PMC3808197.
Article21. Vodyanik MA, Thomson JA, Slukvin II. 2006; Leukosialin (CD43) defines hematopoietic progenitors in human embryonic stem cell differentiation cultures. Blood. 108:2095–2105. DOI: 10.1182/blood-2006-02-003327. PMID: 16757688. PMCID: PMC1895535.
Article22. Gartel AL, Radhakrishnan SK. 2005; Lost in transcription: p21 repression, mechanisms, and consequences. Cancer Res. 65:3980–3985. DOI: 10.1158/0008-5472.CAN-04-3995. PMID: 15899785.
Article23. Cheng T, Rodrigues N, Shen H, Yang Y, Dombkowski D, Sykes M, Scadden DT. 2000; Hematopoietic stem cell quiescence maintained by p21cip1/waf1. Science. 287:1804–1808. DOI: 10.1126/science.287.5459.1804. PMID: 10710306.
Article24. Waga S, Hannon GJ, Beach D, Stillman B. 1994; The p21 inhibitor of cyclin-dependent kinases controls DNA replication by interaction with PCNA. Nature. 369:574–578. DOI: 10.1038/369574a0. PMID: 7911228.
Article25. Sun W, Teng J, Zeng J, Xue Y, Chang J, Zhang Y, Pan X, Zhou Y, Lai M, Bian G, Zhou Q, Liu J, Chen B, Ma F. 2019; The piggyBac-based double-inducible binary vector system: a novel universal platform for studying gene functions and interactions. Plasmid. 105:102420. DOI: 10.1016/j.plasmid.2019.102420. PMID: 31265838.
Article26. Xiao Y, Wang J, Song H, Zou P, Zhou D, Liu L. 2013; CD34+ cells from patients with myelodysplastic syndrome present different p21 dependent premature senescence. Leuk Res. 37:333–340. DOI: 10.1016/j.leukres.2012.11.006. PMID: 23219618.
Article27. Albanese P, Chagraoui J, Charon M, Cocault L, Dusanter-Fourt I, Romeo PH, Uzan G. 2002; Forced expression of p21 in GPIIb-p21 transgenic mice induces abnormalities in the proliferation of erythroid and megakaryocyte progenitors and primitive hematopoietic cells. Exp Hematol. 30:1263–1272. DOI: 10.1016/S0301-472X(02)00933-5.
Article28. Fortunel NO, Hatzfeld A, Hatzfeld JA. 2000; Transforming growth factor-beta: pleiotropic role in the regulation of hematopoiesis. Blood. 96:2022–2036. DOI: 10.1182/blood.V96.6.2022. PMID: 10979943.
Article29. Cheng T, Shen H, Rodrigues N, Stier S, Scadden DT. 2001; Transforming growth factor beta 1 mediates cell-cycle arrest of primitive hematopoietic cells independent of p21(Cip1/Waf1) or p27(Kip1). Blood. 98:3643–3649. DOI: 10.1182/blood.V98.13.3643. PMID: 11739168.
Article30. Ducos K, Panterne B, Fortunel N, Hatzfeld A, Monier MN, Hatzfeld J. 2000; p21(cip1) mRNA is controlled by endogenous transforming growth factor-beta1 in quiescent human hematopoietic stem/progenitor cells. J Cell Physiol. 184:80–85. DOI: 10.1002/(SICI)1097-4652(200007)184:1<80::AID-JCP8>3.0.CO;2-Q. PMID: 10825236.
Article31. Cordenonsi M, Dupont S, Maretto S, Insinga A, Imbriano C, Piccolo S. 2003; Links between tumor suppressors: p53 is required for TGF-beta gene responses by cooperating with Smads. Cell. 113:301–314. DOI: 10.1016/S0092-8674(03)00308-8. PMID: 12732139.32. Dutto I, Tillhon M, Cazzalini O, Stivala LA, Prosperi E. 2015; Biology of the cell cycle inhibitor p21(CDKN1A): molecular mechanisms and relevance in chemical toxicology. Arch Toxicol. 89:155–178. DOI: 10.1007/s00204-014-1430-4. PMID: 25514883.
Article33. Warbrick E, Lane DP, Glover DM, Cox LS. 1995; A small peptide inhibitor of DNA replication defines the site of interaction between the cyclin-dependent kinase inhibitor p21WAF1 and proliferating cell nuclear antigen. Curr Biol. 5:275–282. DOI: 10.1016/S0960-9822(95)00058-3.
Article34. Chen J, Saha P, Kornbluth S, Dynlacht BD, Dutta A. 1996; Cyclin-binding motifs are essential for the function of p21CIP1. Mol Cell Biol. 16:4673–4682. DOI: 10.1128/MCB.16.9.4673. PMID: 8756624. PMCID: PMC231467.
Article35. Oku T, Ikeda S, Sasaki H, Fukuda K, Morioka H, Ohtsuka E, Yoshikawa H, Tsurimoto T. 1998; Functional sites of human PCNA which interact with p21 (Cip1/Waf1), DNA polymerase delta and replication factor C. Genes Cells. 3:357–369. DOI: 10.1046/j.1365-2443.1998.00199.x. PMID: 9734782.
Article36. Gottifredi V, McKinney K, Poyurovsky MV, Prives C. 2004; Decreased p21 levels are required for efficient restart of DNA synthesis after S phase block. J Biol Chem. 279:5802–5810. DOI: 10.1074/jbc.M310373200. PMID: 14597617.
Article37. Cazzalini O, Perucca P, Riva F, Stivala LA, Bianchi L, Vannini V, Ducommun B, Prosperi E. 2003; p21CDKN1A does not interfere with loading of PCNA at DNA replication sites, but inhibits subsequent binding of DNA polymerase delta at the G1/S phase transition. Cell Cycle. 2:596–603. DOI: 10.4161/cc.2.6.502. PMID: 14504476.38. Smits VA, Klompmaker R, Vallenius T, Rijksen G, Mäkela TP, Medema RH. 2000; p21 inhibits Thr161 phosphorylation of Cdc2 to enforce the G2 DNA damage checkpoint. J Biol Chem. 275:30638–30643. DOI: 10.1074/jbc.M005437200. PMID: 10913154.
Article39. Charrier-Savournin FB, Château MT, Gire V, Sedivy J, Piette J, Dulic V. 2004; p21-Mediated nuclear retention of cyclin B1-Cdk1 in response to genotoxic stress. Mol Biol Cell. 15:3965–3976. DOI: 10.1091/mbc.e03-12-0871. PMID: 15181148. PMCID: PMC515331.
Article40. Gillis LD, Leidal AM, Hill R, Lee PW. 2009; p21Cip1/WAF1 mediates cyclin B1 degradation in response to DNA damage. Cell Cycle. 8:253–256. DOI: 10.4161/cc.8.2.7550. PMID: 19158493.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Ginseng on the Expression of the Onco-suppressor Gene p21in Human Breast-Cancer Cell Lines
- p53 gene transfer does not enhance E2F-1-mediated apoptosis in human colon cancer cells
- The Effect of Transfer of p21/WAF1 Gene on Kidney and Bladder Cancer Cell Lines
- Apoptotic effect of IP6 was not enhanced by co-treatment with myo-inositol in prostate carcinoma PC3 cells
- Effects of p27 Overexpression on Head and Neck Squamous Cell Carcinoma Cell Lines