J Breast Cancer.
2019 Dec;22(4):562-578. 10.4048/jbc.2019.22.e56.
Notch1 in Tumor Microvascular Endothelial Cells and Tumoral miR-34a as Prognostic Markers in Locally Advanced Triple-Negative Breast Cancer
- Affiliations
-
- 1Department of Hospital Pathology, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. klee@catholic.ac.kr
- 2Department of Biomedicine & Health Sciences, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 3Division of Medical Oncology, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 4Cancer Research Institute, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 5Department of Radiology, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 6Division of Breast-Thyroid Surgery, Department of Surgery, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2470893
- DOI: http://doi.org/10.4048/jbc.2019.22.e56
Abstract
- PURPOSE
Triple-negative breast cancer (TNBC) is associated with poor prognosis with limited treatment options. Angiogenesis is known to be involved in the progression of TNBC, and targeting this pathway results in modest clinical benefits. In this study, we analyzed the role of tumor microvascular endothelial Notch1 (EC Notch1) and tumoral miR-34a as prognostic markers in patients with TNBC.
METHODS
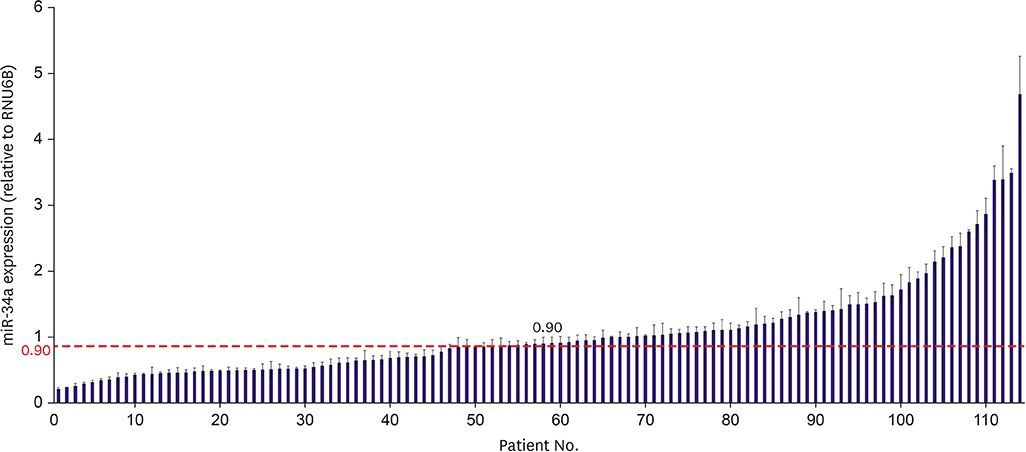
The expression of miR-34a was analyzed using archival tumor tissues from 114 patients with TNBC. Simultaneously, archival tumor tissues were also checked for the expression of CD34 and Notch1 by immunostaining. The ratio of Notch1-microvascular density (MVD) to CD34-MVD was defined as EC Notch1. The association between the expression of miR-34a or EC Notch1 and clinicopathological characteristics was analyzed.
RESULTS
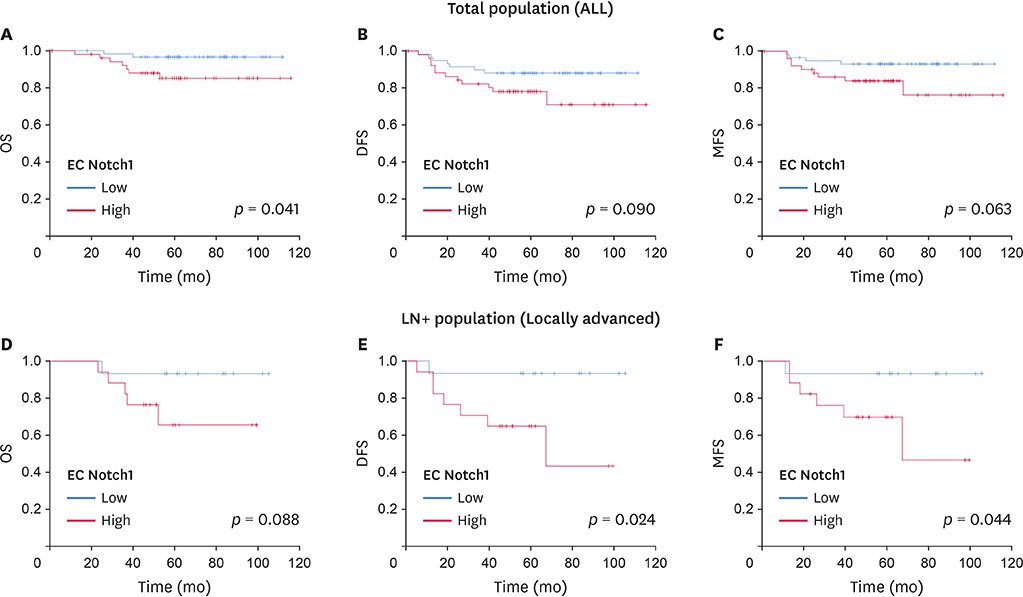
In the overall patient population, patients with low expression of EC Notch1 was associated with better overall survival (OS, p = 0.041) than those with high expression of EC Notch1. In lymph node-positive TNBC patients, high levels of miR-34a and low levels of EC Notch1 correlated significantly with higher survival benefits in terms of OS (p = 0.026), disease-free survival (p = 0.009), and metastasis-free survival (p = 0.038) relative to that in other patients. Decreased expression of EC Notch1 and increased expression of miR-34a also showed a survival benefit in locally advanced TNBC.
CONCLUSION
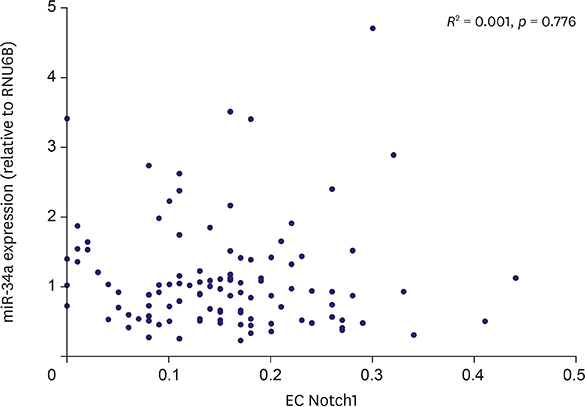
The fact that miR-34a and EC Notch1 are associated with the angiogenesis suggests that angiogenesis may play a role in the development and progression of TNBC.
MeSH Terms
Figure
Reference
-
1. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136:E359–E386.
Article2. Jung KW, Won YJ, Kong HJ, Lee ES. Cancer Statistics in Korea: incidence, mortality, survival, and prevalence in 2016. Cancer Res Treat. 2019; 51:417–430.
Article3. Brufsky A, Valero V, Tiangco B, Dakhil S, Brize A, Rugo HS, et al. Second-line bevacizumab-containing therapy in patients with triple-negative breast cancer: subgroup analysis of the RIBBON-2 trial. Breast Cancer Res Treat. 2012; 133:1067–1075.
Article4. Ribatti D, Nico B, Ruggieri S, Tamma R, Simone G, Mangia A. Angiogenesis and antiangiogenesis in triple-negative breast cancer. Transl Oncol. 2016; 9:453–457.
Article5. Speiser J, Foreman K, Drinka E, Godellas C, Perez C, Salhadar A, et al. Notch-1 and Notch-4 biomarker expression in triple-negative breast cancer. Int J Surg Pathol. 2012; 20:139–145.
Article6. Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007; 356:115–124.
Article7. Lopez-Chavez A, Young T, Fages S, Leon L, Schiller JH, Dowlati A, et al. Bevacizumab maintenance in patients with advanced non-small-cell lung cancer, clinical patterns, and outcomes in the Eastern Cooperative Oncology Group 4599 Study: results of an exploratory analysis. J Thorac Oncol. 2012; 7:1707–1712.
Article8. Gray R, Bhattacharya S, Bowden C, Miller K, Comis RL. Independent review of E2100: a phase III trial of bevacizumab plus paclitaxel versus paclitaxel in women with metastatic breast cancer. J Clin Oncol. 2009; 27:4966–4972.
Article9. Miles DW, Diéras V, Cortés J, Duenne AA, Yi J, O'Shaughnessy J. First-line bevacizumab in combination with chemotherapy for HER2-negative metastatic breast cancer: pooled and subgroup analyses of data from 2447 patients. Ann Oncol. 2013; 24:2773–2780.
Article10. Guo S, Liu M, Gonzalez-Perez RR. Role of Notch and its oncogenic signaling crosstalk in breast cancer. Biochim Biophys Acta. 2011; 1815:197–213.
Article11. Gridley T. Notch signaling in the vasculature. Curr Top Dev Biol. 2010; 92:277–309.
Article12. Dong Y, Zhang T, Li J, Deng H, Song Y, Zhai D, et al. Oridonin inhibits tumor growth and metastasis through anti-angiogenesis by blocking the Notch signaling. PLoS One. 2014; 9:e113830.
Article13. Krebs LT, Xue Y, Norton CR, Shutter JR, Maguire M, Sundberg JP, et al. Notch signaling is essential for vascular morphogenesis in mice. Genes Dev. 2000; 14:1343–1352.
Article14. Ridgway J, Zhang G, Wu Y, Stawicki S, Liang WC, Chanthery Y, et al. Inhibition of Dll4 signalling inhibits tumour growth by deregulating angiogenesis. Nature. 2006; 444:1083–1087.
Article15. Yu S, Sun J, Zhang J, Xu X, Li H, Shan B, et al. Aberrant expression and association of VEGF and Dll4/Notch pathway molecules under hypoxia in patients with lung cancer. Histol Histopathol. 2013; 28:277–284.16. Patel NS, Dobbie MS, Rochester M, Steers G, Poulsom R, Le Monnier K, et al. Up-regulation of endothelial delta-like 4 expression correlates with vessel maturation in bladder cancer. Clin Cancer Res. 2006; 12:4836–4844.
Article17. Bonetti P, Climent M, Panebianco F, Tordonato C, Santoro A, Marzi MJ, et al. Dual role for miR-34a in the control of early progenitor proliferation and commitment in the mammary gland and in breast cancer. Oncogene. 2019; 38:360–374.
Article18. Li Y, Guessous F, Zhang Y, Dipierro C, Kefas B, Johnson E, et al. MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes. Cancer Res. 2009; 69:7569–7576.
Article19. Li Y, Zhang J, Zhang L, Si M, Yin H, Li J. Diallyl trisulfide inhibits proliferation, invasion and angiogenesis of osteosarcoma cells by switching on suppressor microRNAs and inactivating of Notch-1 signaling. Carcinogenesis. 2013; 34:1601–1610.
Article20. Long YJ, Liu XP, Chen SS, Zong DD, Chen Y, Chen P. MiR-34a is involved in CSE-induced apoptosis of human pulmonary microvascular endothelial cells by targeting Notch-1 receptor protein. Respir Res. 2018; 19:21.
Article21. Weidner N, Semple JP, Welch WR, Folkman J. Tumor angiogenesis and metastasis--correlation in invasive breast carcinoma. N Engl J Med. 1991; 324:1–8.
Article22. Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012; 490:61–70.23. Youness RA, Hafez HM, Khallaf E, Assal RA, Abdel Motaal A, Gad MZ. The long noncoding RNA sONE represses triple-negative breast cancer aggressiveness through inducing the expression of miR-34a, miR-15a, miR-16, and let-7a. J Cell Physiol. 2019; 234:20286–20297.
Article24. Yuan X, Zhang M, Wu H, Xu H, Han N, Chu Q, et al. Expression of Notch1 correlates with breast cancer progression and prognosis. PLoS One. 2015; 10:e0131689.
Article25. Qiu M, Peng Q, Jiang I, Carroll C, Han G, Rymer I, et al. Specific inhibition of Notch1 signaling enhances the antitumor efficacy of chemotherapy in triple negative breast cancer through reduction of cancer stem cells. Cancer Lett. 2013; 328:261–270.
Article26. Wieland E, Rodriguez-Vita J, Liebler SS, Mogler C, Moll I, Herberich SE, et al. Endothelial Notch1 activity facilitates metastasis. Cancer Cell. 2017; 31:355–367.
Article27. Zeng Z, Chen X, Zhu D, Luo Z, Yang M. Low expression of circulating microrna-34c is associated with poor prognosis in triple-negative breast cancer. Yonsei Med J. 2017; 58:697–702.
Article28. Imani S, Wei C, Cheng J, Khan MA, Fu S, Yang L, et al. MicroRNA-34a targets epithelial to mesenchymal transition-inducing transcription factors (EMT-TFs) and inhibits breast cancer cell migration and invasion. Oncotarget. 2017; 8:21362–21379.
Article29. Zhang Y, Guan Q, Jin X. MiR-200c serves an important role in H5V endothelial cells in high glucose by targeting Notch1. Mol Med Rep. 2017; 16:2149–2155.
Article30. Sledge GW. Anti-vascular endothelial growth factor therapy in breast cancer: game over? J Clin Oncol. 2015; 33:133–135.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- miR-205 and miR-200c: Predictive Micro RNAs for Lymph Node Metastasis in Triple Negative Breast Cancer
- LncRNA DLG1-AS1 Promotes Cancer Cell Proliferation in Triple Negative Breast Cancer by Downregulating miR-203
- Low Expression of Circulating MicroRNA-34c is Associated with Poor Prognosis in Triple-Negative Breast Cancer
- In vivo validation of metastasis-regulating microRNA-766 in human triple-negative breast cancer cells
- The Changes of the Histologic and Biologic Markers Induced by Neoadjuvant Chemotherapy in Locally Advanced Breast Cancer