J Breast Cancer.
2020 Feb;23(1):93-99. 10.4048/jbc.2020.23.e2.
Malignant transformation in a Breast Adenomyoepithelioma Caused by Amplification of c-MYC: A Common pathway to Cancer in a Rare Entity
- Affiliations
-
- 1Arkadi M. Rywlin Department of Pathology and Laboratory Medicine, Mount Sinai Medical Center, Miami Beach, FL, USA. christopher.febres@msmc.com
- 2Section of Surgical Oncology, Mount Sinai Medical Center, Miami Beach, FL, USA.
- 3Department of Pathology, Herbert Wertheim College of Medicine, Florida International University, Miami, FL, USA.
- KMID: 2470885
- DOI: http://doi.org/10.4048/jbc.2020.23.e2
Abstract
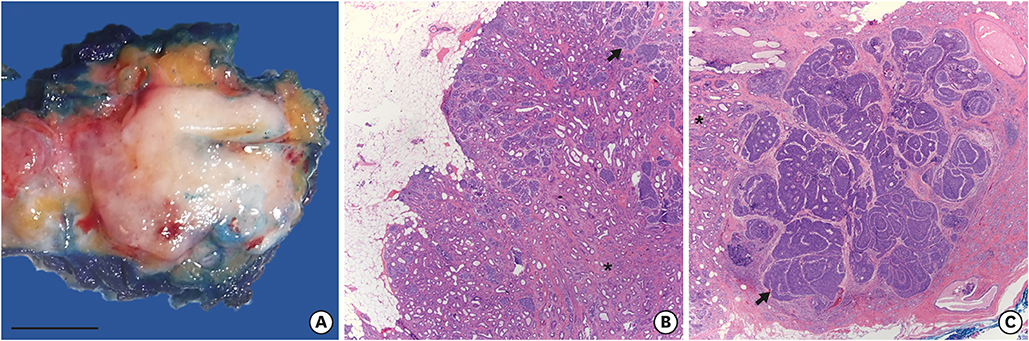
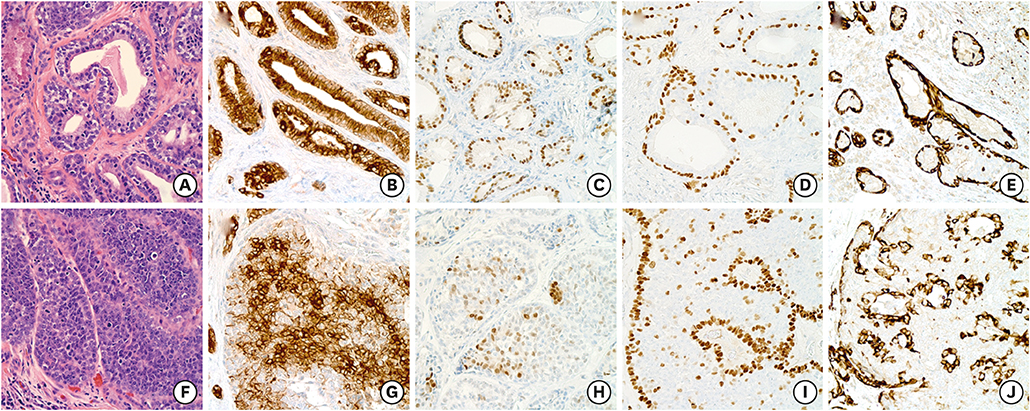
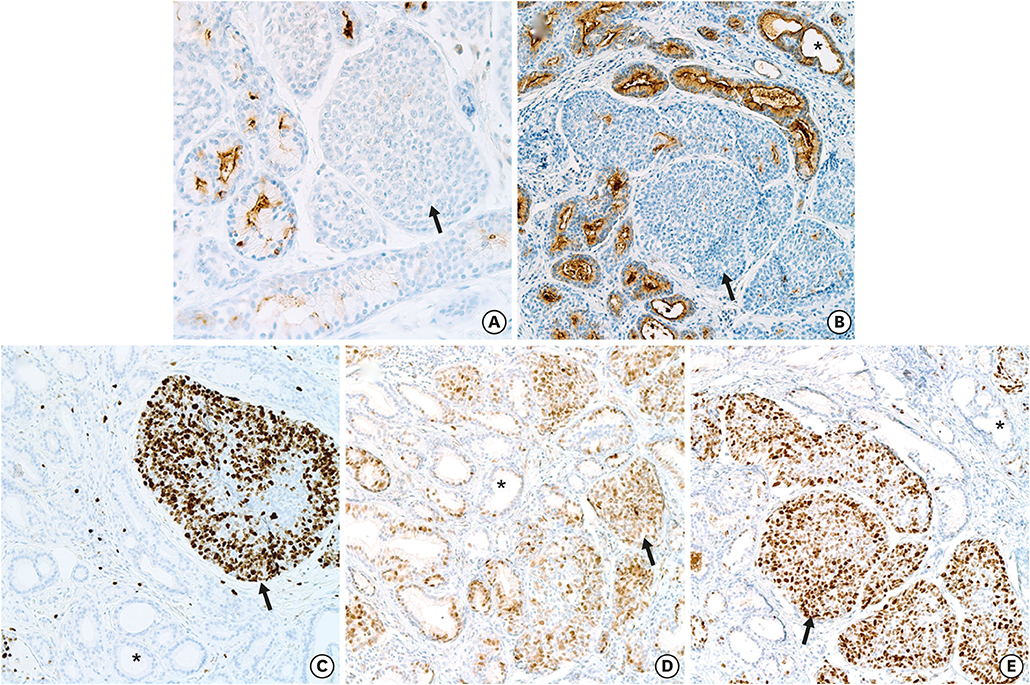
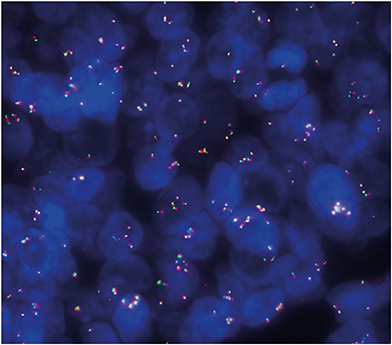
- Breast adenomyoepitheliomas are composed of a biphasic proliferation of myoepithelial cells around small epithelial-lined spaces. Due to the rarity of adenomyoepitheliomas, the molecular data describing them are limited. Adenomyoepitheliomas are considered to be benign or have low malignant potential, and be prone to local recurrence. Malignant transformation has been associated with homozygous deletion of CDKN2A or somatic mutations in TERT, but remains unexplained in many cases. Here, we describe a case of carcinomatous transformation of both epithelial and myoepithelial cells in an estrogen receptor-negative adenomyoepithelioma caused by amplification of MYC. Break-apart fluorescence in situ hybridization revealed an increase in the MYC gene copy number (3-4 copies/cell in 37%, > 4 copies/cell in 40%). Deregulation of MYC is responsible for uncontrolled proliferation and cellular immortalization in basal-like breast cancers. Our case demonstrates that genomic instability events associated with gene amplification may be involved in the carcinogenesis of malignant adenomyoepitheliomas.
Keyword
MeSH Terms
Figure
Reference
-
1. Rasbridge SA, Millis RR. Adenomyoepithelioma of the breast with malignant features. Virchows Arch. 1998; 432:123–130.
Article2. McLaren BK, Smith J, Schuyler PA, Dupont WD, Page DL. Adenomyoepithelioma: clinical, histologic, and immunohistologic evaluation of a series of related lesions. Am J Surg Pathol. 2005; 29:1294–1299.3. Lubin D, Toorens E, Zhang PJ, Jaffer S, Baraban E, Bleiweiss IJ, et al. Adenomyoepitheliomas of the breast frequently harbor recurrent hotspot mutations in PIK3-AKT pathway-related genes and a subset show genetic similarity to salivary gland epithelial-myoepithelial carcinoma. Am J Surg Pathol. 2019; 43:1005–1013.
Article4. Geyer FC, Li A, Papanastasiou AD, Smith A, Selenica P, Burke KA, et al. Recurrent hotspot mutations in HRAS Q61 and PI3K-AKT pathway genes as drivers of breast adenomyoepitheliomas. Nat Commun. 2018; 9:1816.
Article5. Baum JE, Sung KJ, Tran H, Song W, Ginter PS. Mammary epithelial-myoepithelial carcinoma: report of a case with HRAS and PIK3CA mutations by next-generation sequencing. Int J Surg Pathol. 2019; 27:441–445.
Article6. Robanus-Maandag EC, Bosch CA, Kristel PM, Hart AA, Faneyte IF, Nederlof PM, et al. Association of c-MYC amplification with progression from the in situ to the invasive stage in c-MYC-amplified breast carcinomas. J Pathol. 2003; 201:75–82.
Article7. Xu J, Chen Y, Olopade OI. MYC and breast cancer. Genes Cancer. 2010; 1:629–640.8. Seifert G. Are adenomyoepithelioma of the breast and epithelial-myoepithelial carcinoma of the salivary glands identical tumours? Virchows Arch. 1998; 433:285–288.
Article9. Moritz AW, Wiedenhoefer JF, Profit AP, Jagirdar J. Breast adenomyoepithelioma and adenomyoepithelioma with carcinoma (malignant adenomyoepithelioma) with associated breast malignancies: a case series emphasizing histologic, radiologic, and clinical correlation. Breast. 2016; 29:132–139.
Article10. Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ. Adenomyoepithelioma and adenomyoepithelioma with carcinoma. WHO Classification of Tumours of the Breast. 4th ed. Lyon: IARC Press;2012. p. 120–123.11. Grushko TA, Dignam JJ, Das S, Blackwood AM, Perou CM, Ridderstråle KK, et al. MYC is amplified in BRCA1-associated breast cancers. Clin Cancer Res. 2004; 10:499–507.
Article12. Bodvarsdóttir SK, Steinarsdóttir M, Hilmarsdóttir H, Jónasson JG, Eyfjörd JE. MYC amplification and TERT expression in breast tumor progression. Cancer Genet Cytogenet. 2007; 176:93–99.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Malignant Adenomyoepithelioma of the Breast
- Report of Benign and Malignant Adenomyoepithelioma of the Breast
- The Breast Adenomyoepithelioma
- Mammographic & Sonographic findings of Adenomyoepithelioma of the Breast: Case Report
- Malignant Adenomyoepithelioma of the Breast Presenting as a Large Mass that Grew Slowly without Metastasis