J Korean Assoc Oral Maxillofac Surg.
2019 Apr;45(2):97-107. 10.5125/jkaoms.2019.45.2.97.
Are critical size bone notch defects possible in the rabbit mandible?
- Affiliations
-
- 1Department of Craniomaxillofacial Regenerative Medicine, Dental and Trauma Research Detachment, Fort Sam Houston, TX, USA.
- 2Department of Biomedical Engineering, University of Texas at San Antonio, San Antonio, TX, USA. teja.guda@utsa.edu
- KMID: 2467153
- DOI: http://doi.org/10.5125/jkaoms.2019.45.2.97
Abstract
OBJECTIVES
Small animal maxillofacial models, such as non-segmental critical size defects (CSDs) in the rabbit mandible, need to be standardized for use as preclinical models of bone regeneration to mimic clinical conditions such as maxillofacial trauma. The objective of this study is the establishment of a mechanically competent CSD model in the rabbit mandible to allow standardized evaluation of bone regeneration therapies.
MATERIALS AND METHODS
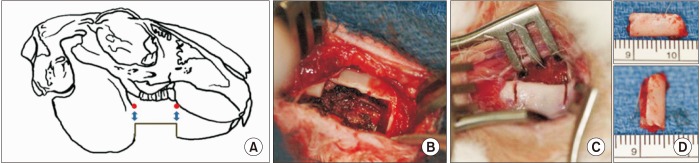
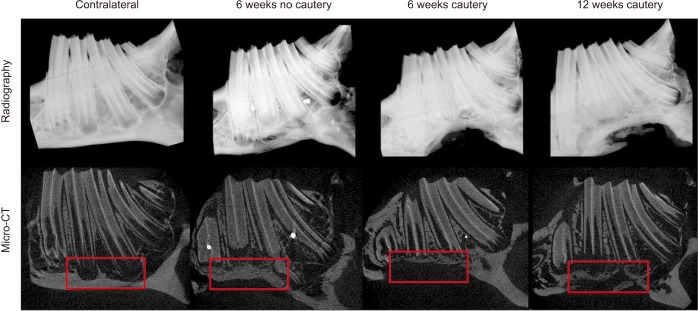
Three sizes of bony defect were generated in the mandibular body of rabbit hemi-mandibles: 12 mm×5 mm, 12 mm×8 mm, and 15 mm×10 mm. The hemi-mandibles were tested to failure in 3-point flexure. The 12 mm×5 mm defect was then chosen for the defect size created in the mandibles of 26 rabbits with or without cautery of the defect margins and bone regeneration was assessed after 6 and 12 weeks. Regenerated bone density and volume were evaluated using radiography, micro-computed tomography, and histology.
RESULTS
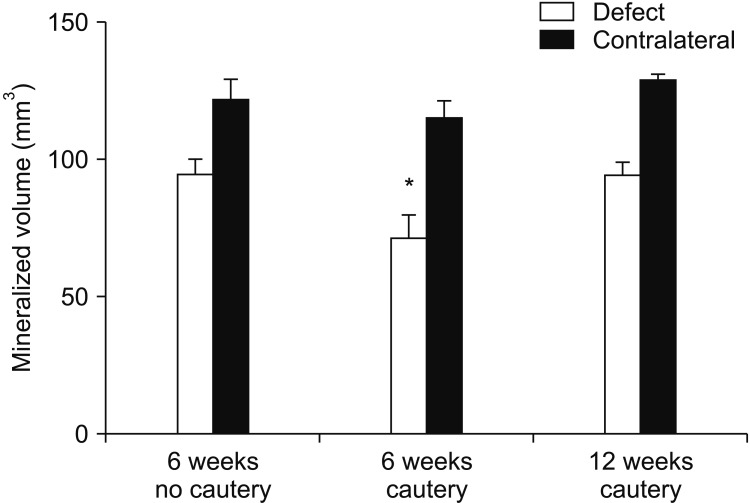
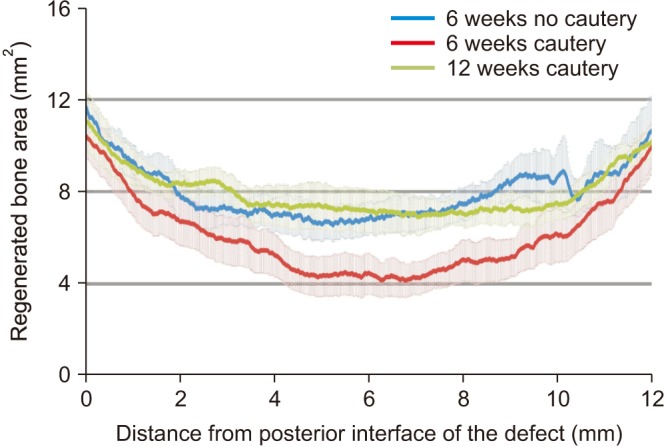
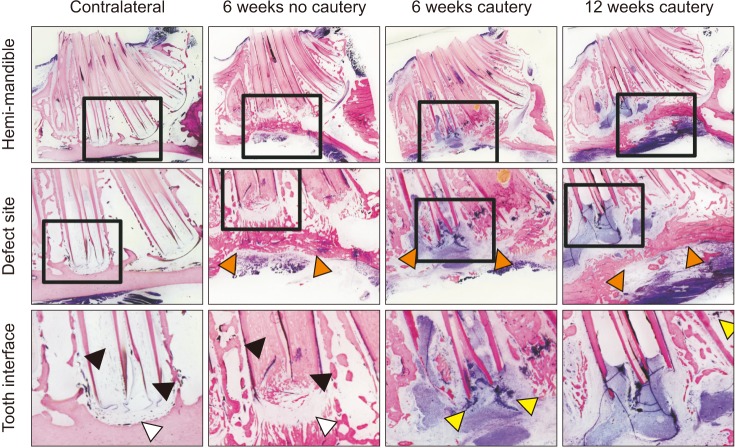
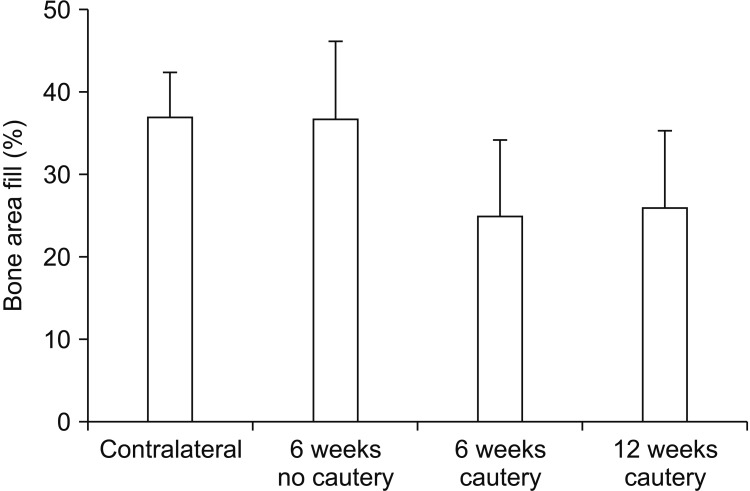
Flexural strength of the 12 mm×5 mm defect was similar to its contralateral; whereas the 12 mm×8 mm and 15 mm×10 mm groups carried significantly less load than their respective contralaterals (P<0.05). This demonstrated that the 12 mm×5 mm defect did not significantly compromise mandibular mechanical integrity. Significantly less (P<0.05) bone was regenerated at 6 weeks in cauterized defect margins compared to controls without cautery. After 12 weeks, the bone volume of the group with cautery increased to that of the control without cautery after 6 weeks.
CONCLUSION
An empty defect size of 12 mm×5 mm in the rabbit mandibular model maintains sufficient mechanical stability to not require additional stabilization. However, this defect size allows for bone regeneration across the defect. Cautery of the defect only delays regeneration by 6 weeks suggesting that the performance of bone graft materials in mandibular defects of this size should be considered with caution.
Keyword
MeSH Terms
Figure
Reference
-
1. Glendor U. Aetiology and risk factors related to traumatic dental injuries--a review of the literature. Dent Traumatol. 2009; 25:19–31. PMID: 19208007.2. Zachar MR, Labella C, Kittle CP, Baer PB, Hale RG, Chan RK. Characterization of mandibular fractures incurred from battle injuries in Iraq and Afghanistan from 2001–2010. J Oral Maxillofac Surg. 2013; 71:734–742. PMID: 23351482.
Article3. Schmitz JP, Hollinger JO. The critical size defect as an experimental model for craniomandibulofacial nonunions. Clin Orthop Relat Res. 1986; (205):299–308. PMID: 3084153.
Article4. Cooper GM, Mooney MP, Gosain AK, Campbell PG, Losee JE, Huard J. Testing the critical size in calvarial bone defects: revisiting the concept of a critical-size defect. Plast Reconstr Surg. 2010; 125:1685–1692. PMID: 20517092.
Article5. Vajgel A, Mardas N, Farias BC, Petrie A, Cimões R, Donos N. A systematic review on the critical size defect model. Clin Oral Implants Res. 2014; 25:879–893. PMID: 23742162.
Article6. Guda T, Darr A, Silliman DT, Magno MH, Wenke JC, Kohn J, et al. Methods to analyze bone regenerative response to different rhBMP-2 doses in rabbit craniofacial defects. Tissue Eng Part C Methods. 2014; 20:749–760. PMID: 24422668.
Article7. Dumas JE, BrownBaer PB, Prieto EM, Guda T, Hale RG, Wenke JC, et al. Injectable reactive biocomposites for bone healing in critical-size rabbit calvarial defects. Biomed Mater. 2012; 7:024112. PMID: 22456057.
Article8. Choi BH, Im CJ, Huh JY, Suh JJ, Lee SH. Effect of platelet-rich plasma on bone regeneration in autogenous bone graft. Int J Oral Maxillofac Surg. 2004; 33:56–59. PMID: 14690660.
Article9. Carlisle PL, Guda T, Silliman DT, Lien W, Hale RG, Brown Baer PR. Investigation of a pre-clinical mandibular bone notch defect model in miniature pigs: clinical computed tomography, micro-computed tomography, and histological evaluation. J Korean Assoc Oral Maxillofac Surg. 2016; 42:20–30. PMID: 26904491.
Article10. Carstens MH, Chin M, Li XJ. In situ osteogenesis: regeneration of 10-cm mandibular defect in porcine model using recombinant human bone morphogenetic protein-2 (rhBMP-2) and Helistat absorbable collagen sponge. J Craniofac Surg. 2005; 16:1033–1042. PMID: 16327551.
Article11. Herford AS, Boyne PJ. Reconstruction of mandibular continuity defects with bone morphogenetic protein-2 (rhBMP-2). J Oral Maxillofac Surg. 2008; 66:616–624. PMID: 18355584.
Article12. Chim H, Gosain AK. Biomaterials in craniofacial surgery: experimental studies and clinical application. J Craniofac Surg. 2009; 20:29–33. PMID: 19164984.13. Misch CE, Qu Z, Bidez MW. Mechanical properties of trabecular bone in the human mandible: implications for dental implant treatment planning and surgical placement. J Oral Maxillofac Surg. 1999; 57:700–706. discussion 706-8. PMID: 10368096.
Article14. Hollinger JO, Kleinschmidt JC. The critical size defect as an experimental model to test bone repair materials. J Craniofac Surg. 1990; 1:60–68. PMID: 1965154.
Article15. Zwetyenga N, Catros S, Emparanza A, Deminiere C, Siberchicot F, Fricain JC. Mandibular reconstruction using induced membranes with autologous cancellous bone graft and HA-betaTCP: animal model study and preliminary results in patients. Int J Oral Maxillofac Surg. 2009; 38:1289–1297. PMID: 19699612.16. Busuttil Naudi K, Ayoub A, McMahon J, Di Silvio L, Lappin D, Hunter KD, et al. Mandibular reconstruction in the rabbit using beta-tricalcium phosphate (β-TCP) scaffolding and recombinant bone morphogenetic protein 7 (rhBMP-7): histological, radiographic and mechanical evaluations. J Craniomaxillofac Surg. 2012; 40:e461–e469. PMID: 22507295.17. Liu HY, Liu X, Zhang LP, Ai HJ, Cui FZ. Improvement on the performance of bone regeneration of calcium sulfate hemihydrate by adding mineralized collagen. Tissue Eng Part A. 2010; 16:2075–2084. PMID: 20136401.
Article18. Young S, Bashoura AG, Borden T, Baggett LS, Jansen JA, Wong M, et al. Development and characterization of a rabbit alveolar bone nonhealing defect model. J Biomed Mater Res A. 2008; 86:182–194. PMID: 17969052.
Article19. Eleftheriadis E, Leventis MD, Tosios KI, Faratzis G, Titsinidis S, Eleftheriadi I, et al. Osteogenic activity of β-tricalcium phosphate in a hydroxyl sulphate matrix and demineralized bone matrix: a histological study in rabbit mandible. J Oral Sci. 2010; 52:377–384. PMID: 20881329.
Article20. Kim KN, Yang JE, Jang JW, Sasikala B, Beng W, Kim IK. Morphometric analysis on bone formation effect of β-TCP and rhBMP-2 in rabbit mandible. J Korean Assoc Oral Maxillofac Surg. 2010; 36:161–171.
Article21. Nguyen C, Young S, Kretlow JD, Mikos AG, Wong M. Surface characteristics of biomaterials used for space maintenance in a mandibular defect: a pilot animal study. J Oral Maxillofac Surg. 2011; 69:11–18. PMID: 21055856.
Article22. He H, Yan W, Chen G, Lu Z. Acceleration of de novo bone formation with a novel bioabsorbable film: a histomorphometric study in vivo. J Oral Pathol Med. 2008; 37:378–382. PMID: 18355176.
Article23. Shah SR, Young S, Goldman JL, Jansen JA, Wong ME, Mikos AG. A composite critical-size rabbit mandibular defect for evaluation of craniofacial tissue regeneration. Nat Protoc. 2016; 11:1989–2009. PMID: 27658014.
Article24. Lu M, Rabie AB. Microarchitecture of rabbit mandibular defects grafted with intramembranous or endochondral bone shown by micro-computed tomography. Br J Oral Maxillofac Surg. 2003; 41:385–391. PMID: 14614867.
Article25. Miloro M, Haralson DJ, Desa V. Bone healing in a rabbit mandibular defect using platelet-rich plasma. J Oral Maxillofac Surg. 2010; 68:1225–1230. PMID: 20227807.
Article26. Zhang JC, Lu HY, Lv GY, Mo AC, Yan YG, Huang C. The repair of critical-size defects with porous hydroxyapatite/polyamide nanocomposite: an experimental study in rabbit mandibles. Int J Oral Maxillofac Surg. 2010; 39:469–477. PMID: 20194003.
Article27. Guo J, Meng Z, Chen G, Xie D, Chen Y, Wang H, et al. Restoration of critical-size defects in the rabbit mandible using porous nanohydroxyapatite-polyamide scaffolds. Tissue Eng Part A. 2012; 18:1239–1252. PMID: 22320360.
Article28. Ye L, Zeng X, Li H, Wang Z. Fabrication and biocompatibility of porously bioactive scaffold of nonstoichiometric apatite and poly(ε-caprolactone) nanocomposite. J Appl Polym Sci. 2010; 116:762–770.
Article29. Li J, Li Y, Ma S, Gao Y, Zuo Y, Hu J. Enhancement of bone formation by BMP-7 transduced MSCs on biomimetic nano-hydroxyapatite/polyamide composite scaffolds in repair of mandibular defects. J Biomed Mater Res A. 2010; 95:973–981. PMID: 20845497.
Article30. Zhang H, Ye XJ, Li JS. Preparation and biocompatibility evaluation of apatite/wollastonite-derived porous bioactive glass ceramic scaffolds. Biomed Mater. 2009; 4:045007. PMID: 19605959.
Article31. Asikainen AJ, Noponen J, Lindqvist C, Pelto M, Kellomäki M, Juuti H, et al. Tyrosine-derived polycarbonate membrane in treating mandibular bone defects. An experimental study. J R Soc Interface. 2006; 3:629–635. PMID: 16971331.
Article32. Ren J, Ren T, Zhao P, Huang Y, Pan K. Repair of mandibular defects using MSCs-seeded biodegradable polyester porous scaffolds. J Biomater Sci Polym Ed. 2007; 18:505–517. PMID: 17550655.
Article33. Bulut T, Durmuş E, Mihmanlı A, Dolanmaz D, Kalaycı A, Sağlam H. Distracted mandible does not reach the same strength as normal mandible in rabbits. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012; 114(5 Suppl):S140–S145. PMID: 23063390.
Article34. Erdogan O, Esen E, Ustün Y, Kürkçü M, Akova T, Gönlüşen G, et al. Effects of low-intensity pulsed ultrasound on healing of mandibular fractures: an experimental study in rabbits. J Oral Maxillofac Surg. 2006; 64:180–188. PMID: 16413888.35. Wang H, Li Y, Zuo Y, Li J, Ma S, Cheng L. Biocompatibility and osteogenesis of biomimetic nano-hydroxyapatite/polyamide composite scaffolds for bone tissue engineering. Biomaterials. 2007; 28:3338–3348. PMID: 17481726.
Article36. Meyer U, Joos U, Wiesmann HP. Biological and biophysical principles in extracorporal bone tissue engineering. Part III. Int J Oral Maxillofac Surg. 2004; 33:635–641. PMID: 15337175.37. Zoumalan RA, Hirsch DL, Levine JP, Saadeh PB. Plating in microvascular reconstruction of the mandible: can fixation be too rigid? J Craniofac Surg. 2009; 20:1451–1454. PMID: 19816277.38. Guda T, Walker JA, Singleton BM, Hernandez JW, Son JS, Kim SG, et al. Guided bone regeneration in long-bone defects with a structural hydroxyapatite graft and collagen membrane. Tissue Eng Part A. 2013; 19:1879–1888. PMID: 22844877.
Article39. Jiang X, Gittens SA, Chang Q, Zhang X, Chen C, Zhang Z. The use of tissue-engineered bone with human bone morphogenetic protein-4-modified bone-marrow stromal cells in repairing mandibular defects in rabbits. Int J Oral Maxillofac Surg. 2006; 35:1133–1139. PMID: 17023144.
Article40. Lu M, Rabie AB. The effect of demineralized intramembranous bone matrix and basic fibroblast growth factor on the healing of allogeneic intramembranous bone grafts in the rabbit. Arch Oral Biol. 2002; 47:831–841. PMID: 12450514.
Article41. Yoshimoto R, Murata M, Akazawa T, Arisue M. Effects of functionally graded hydroxyapatite for large mandibular defects in adult rabbits. J Hard Tissue Biol. 2010; 19:33–42.
Article42. Hassanein AH, Couto RA, Nedder A, Zielins ER, Greene AK. Critical-size defect ossification: effect of leporid age in a cranioplasty model. J Craniofac Surg. 2011; 22:2341–2343. PMID: 22134272.43. Huang GT, Gronthos S, Shi S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res. 2009; 88:792–806. PMID: 19767575.44. Kim SH, Kim KH, Seo BM, Koo KT, Kim TI, Seol YJ, et al. Alveolar bone regeneration by transplantation of periodontal ligament stem cells and bone marrow stem cells in a canine peri-implant defect model: a pilot study. J Periodontol. 2009; 80:1815–1823. PMID: 19905951.
Article45. Kokubu T, Hak DJ, Hazelwood SJ, Reddi AH. Development of an atrophic nonunion model and comparison to a closed healing fracture in rat femur. J Orthop Res. 2003; 21:503–510. PMID: 12706024.
Article46. Mills LA, Simpson AH. In vivo models of bone repair. J Bone Joint Surg Br. 2012; 94:865–874. PMID: 22733938.47. Regan JD, Witherspoon DE, Foyle D. Surgical repair of root and tooth perforations. Endodontic Topics. 2005; 11:152–178.
Article48. Andreasen JO, Borum MK, Andreasen FM. Replantation of 400 avulsed permanent incisors. 3. Factors related to root growth. Endod Dent Traumatol. 1995; 11:69–75. PMID: 7641621.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Investigation of a pre-clinical mandibular bone notch defect model in miniature pigs: clinical computed tomography, micro-computed tomography, and histological evaluation
- Spontaneous healing capacity of rabbit cranial defects of various sizes
- THE EFFECT OF BIOACTIVE GLASS AND A RESORBABLE MEMBRANE ON BONE REGENERATION OF THE MANDIBULAR BONE DEFECTS IN RABBIT
- THE EFFECT OF APPLICATION OF n-PTFE ON MANDIBULAR BONE DEFECTS OF RABBITS
- A Clinical and Anatomical Study on the Mandible for Inferior Alveolar Nerve Conductive Anesthesia in Korean