Dement Neurocogn Disord.
2019 Dec;18(4):130-137. 10.12779/dnd.2019.18.4.130.
The Effect of Clinical Characteristics and Subtypes on Amyloid Positivity in Patients with Amnestic Mild Cognitive Impairment
- Affiliations
-
- 1Department of Neurology, Cognitive Disorders and Dementia Center, Dong-A University College of Medicine, Busan, Korea. neuropark@dau.ac.kr
- 2Department of Nuclear Medicine, Dong-A University College of Medicine, Busan, Korea.
- KMID: 2466756
- DOI: http://doi.org/10.12779/dnd.2019.18.4.130
Abstract
- BACKGROUND AND PURPOSE
Mild cognitive impairment (MCI) is a prodromal stage of dementia. Amyloid deposits in positron-emission tomography (PET) imaging of MCI patients imply a higher risk for advancing to dementia, with rates of 10%-15% yearly. The purpose of this study was to investigate the clinical characteristics of subgroups of amnestic MCI (aMCI) that may have a higher impact on amyloid positivity.
METHODS
We recruited 136 aMCI patients. All patients underwent a 20-minute F-18 florbetaben or flutemetamol PET scan. We classified amyloid PET images as positive or negative according to a semi-quantitative method. We evaluated the amyloid positivity of subgroups of aMCI (early vs. late type, single vs. multiple amnestic type, verbal vs. verbal, and visual amnestic type), and compared baseline clinical characteristics including key risk factors, apolipoprotein E4 (apoE4) genotype, and neuropsychological assessments with amyloid positivity in aMCI.
RESULTS
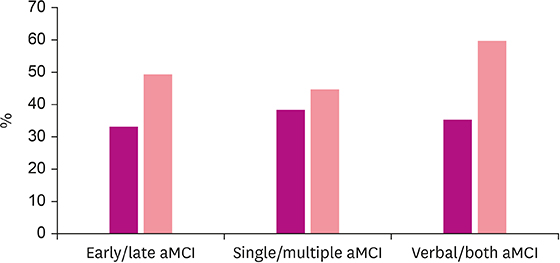
The amyloid positivity in total aMCI was 41%. The positivity rate according to subgroup of aMCI were as follow: Late aMCI (49%) vs. early aMCI (33%) (p=0.13), multiple aMCI (40%) vs. single aMCI (38%) (p=0.51), and verbal and visual aMCI (59%) vs. verbal aMCI (35%) (p=0.01), respectively. The mean age and the frequency of apoE4 allele of the amyloid-positive group was higher than that of the amyloid-negative group in aMCI (p< 0.01).
CONCLUSIONS
We found that the amyloid positivity was related to patterns of clinical subtypes, characteristics, and risk factors in patients with aMCI.
MeSH Terms
Figure
Reference
-
1. Gauthier S, Reisberg B, Zaudig M, Petersen RC, Ritchie K, Broich K, et al. Mild cognitive impairment. Lancet. 2006; 367:1262–1270.
Article2. Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004; 256:183–194.
Article3. Tomadesso C, de La Sayette V, de Flores R, Bourgeat P, Villemagne VL, Egret S, et al. Neuropsychology and neuroimaging profiles of amyloid-positive versus amyloid-negative amnestic mild cognitive impairment patients. Alzheimers Dement (Amst). 2018; 10:269–277.
Article4. Petersen RC, Negash S. Mild cognitive impairment: an overview. CNS Spectr. 2008; 13:45–53.
Article5. Sabri O, Seibyl J, Rowe C, Barthel H. Beta-amyloid imaging with florbetaben. Clin Transl Imaging. 2015; 3:13–26.
Article6. Wolk DA, Price JC, Saxton JA, Snitz BE, James JA, Lopez OL, et al. Amyloid imaging in mild cognitive impairment subtypes. Ann Neurol. 2009; 65:557–568.
Article7. Bennett DA, Schneider JA, Bienias JL, Evans DA, Wilson RS. Mild cognitive impairment is related to Alzheimer disease pathology and cerebral infarctions. Neurology. 2005; 64:834–841.
Article8. Mufson EJ, Chen EY, Cochran EJ, Beckett LA, Bennett DA, Kordower JH. Entorhinal cortex beta-amyloid load in individuals with mild cognitive impairment. Exp Neurol. 1999; 158:469–490.
Article9. Landau SM, Horng A, Fero A, Jagust WJ. Alzheimer's Disease Neuroimaging Initiative. Amyloid negativity in patients with clinically diagnosed Alzheimer disease and MCI. Neurology. 2016; 86:1377–1385.
Article10. de Leon MJ, George AE, Stylopoulos LA, Smith G, Miller DC. Early marker for Alzheimer's disease: the atrophic hippocampus. Lancet. 1989; 2:672–673.
Article11. Jack CR Jr, Petersen RC, Xu YC, O'Brien PC, Smith GE, Ivnik RJ, et al. Prediction of AD with MRI-based hippocampal volume in mild cognitive impairment. Neurology. 1999; 52:1397–1403.
Article12. De Santi S, de Leon MJ, Rusinek H, Convit A, Tarshish CY, Roche A, et al. Hippocampal formation glucose metabolism and volume losses in MCI and AD. Neurobiol Aging. 2001; 22:529–539.
Article13. Chételat G, Desgranges B, de la Sayette V, Viader F, Eustache F, Baron JC. Mild cognitive impairment: can FDG-PET predict who is to rapidly convert to Alzheimer's disease? Neurology. 2003; 60:1374–1377.
Article14. Karas GB, Scheltens P, Rombouts SA, Visser PJ, van Schijndel RA, Fox NC, et al. Global and local gray matter loss in mild cognitive impairment and Alzheimer's disease. Neuroimage. 2004; 23:708–716.
Article15. Korf ES, Wahlund LO, Visser PJ, Scheltens P. Medial temporal lobe atrophy on MRI predicts dementia in patients with mild cognitive impairment. Neurology. 2004; 63:94–100.
Article16. Zhang S, Han D, Tan X, Feng J, Guo Y, Ding Y. Diagnostic accuracy of 18 F-FDG and 11 C-PIB-PET for prediction of short-term conversion to Alzheimer's disease in subjects with mild cognitive impairment. Int J Clin Pract. 2012; 66:185–198.
Article17. Stoub TR, Bulgakova M, Leurgans S, Bennett DA, Fleischman D, Turner DA, et al. MRI predictors of risk of incident Alzheimer disease: a longitudinal study. Neurology. 2005; 64:1520–1524.
Article18. Park JH, Park H, Sohn SW, Kim S, Park KW. Memory performance on the story recall test and prediction of cognitive dysfunction progression in mild cognitive impairment and Alzheimer's dementia. Geriatr Gerontol Int. 2017; 17:1603–1609.
Article19. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997; 15:300–308.20. Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993; 43:2412–2414.21. Fouty HE, Ailes EL, Brown KD, Morgan CL, Sivertsen E, Briceno KY, et al. Content validity of the geriatric depression scale in inpatient health care settings. GSTF J Psychol. 2016; 3:7–9.22. Petersen RC, Thomas RG, Grundman M, Bennett D, Doody R, Ferris S, et al. Vitamin E and donepezil for the treatment of mild cognitive impairment. N Engl J Med. 2005; 352:2379–2388.
Article23. Vega JN, Newhouse PA. Mild cognitive impairment: diagnosis, longitudinal course, and emerging treatments. Curr Psychiatry Rep. 2014; 16:490.
Article24. Molano J, Boeve B, Ferman T, Smith G, Parisi J, Dickson D, et al. Mild cognitive impairment associated with limbic and neocortical Lewy body disease: a clinicopathological study. Brain. 2010; 133:540–556.
Article25. Patterson BW, Elbert DL, Mawuenyega KG, Kasten T, Ovod V, Ma S, et al. Age and amyloid effects on human central nervous system amyloid-beta kinetics. Ann Neurol. 2015; 78:439–453.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Amyloid Positivity Rate and Accumulation Pattern between Amnestic and Non-Amnestic Type Mild Cognitive Impairment
- Correlation between Sleep and C-reactive Protein of Patients in Amnestic Mild Cognitive Impairment and Alzheimer’s Dementia
- Tracking Cognitive Trajectories in Mild Cognitive Impairment Using a Machine Learning Technique of Subtype and Stage Inference
- Anti-LGI1 Antibody Limbic Encephalitis Presented with Amnestic Mild Cognitive Impairment
- Can We Further Divide Amnestic Mild Cognitive Impairment Based on the Pattern of Memory Deficit?: A Preliminary Study