Nat Prod Sci.
2019 Sep;25(3):215-221. 10.20307/nps.2019.25.3.215.
Anti-inflammatory Activity of Sambucus Plant Bioactive Compounds against TNF-α and TRAIL as Solution to Overcome Inflammation Associated Diseases: The Insight from Bioinformatics Study
- Affiliations
-
- 1Department of Biology, Faculty of Mathematics and Natural Sciences, Universitas Negeri Malang, Indonesia.
- 2Department of Nutrition, Faculty of Medicine, Halu Oleo University, Indonesia.
- 3Department of Biology, Faculty of Mathematics and Natural Sciences, Brawijaya University, Indonesia. rifa123@ub.ac.id
- KMID: 2459962
- DOI: http://doi.org/10.20307/nps.2019.25.3.215
Abstract
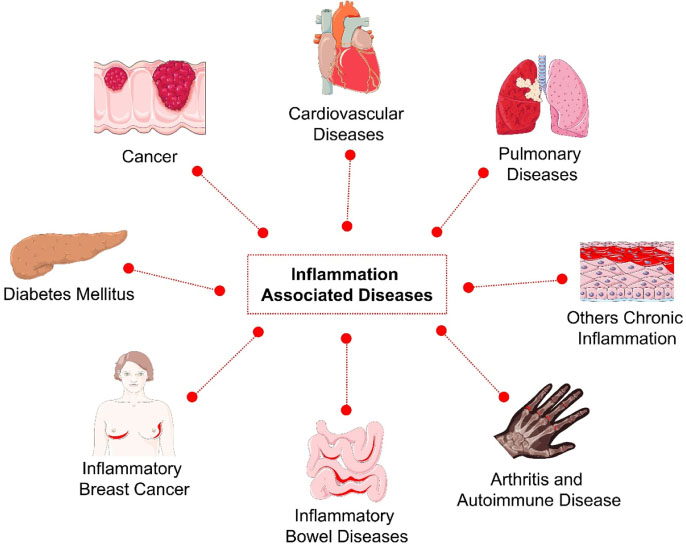
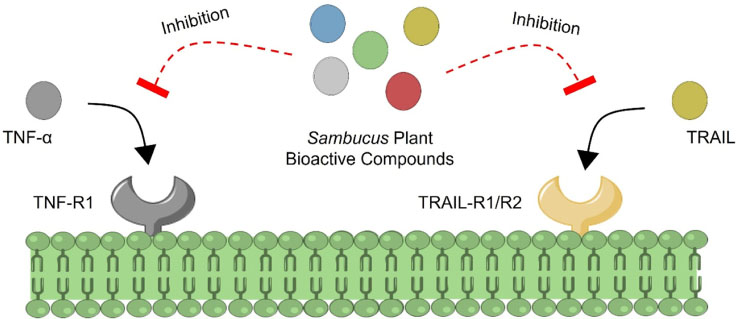
- Inflammation is the crucial biological process of immune system which acts as body's defense and protective response against the injuries or infection. However, the systemic inflammation devotes the adverse effects such as multiple inflammation associated diseases. One of the best ways to treat this entity is by blocking the tumor necrosis factor alpha (TNF-α) and TNF-related apoptosis-inducing ligand (TRAIL) to avoid the proinflammation cytokines production. Thus, this study aims to evaluate the potency of Sambucus bioactive compounds as anti-inflammation through in silico approach. In order to assess that, molecular docking was performed to evaluate the interaction properties between the TNF-α or TRAIL with the ligands. The 2D structure of ligands were retrieved online via PubChem and the 3D protein modeling was done by using SWISS Model. The prediction results of the study showed that caffeic acid (−6.4 kcal/mol) and homovanillic acid (−6.6 kcal/mol) have the greatest binding affinity against the TNF-α and TRAIL respectively. This evidence suggests that caffeic acid and homovanillic acid may potent as anti-inflammatory agent against the inflammation associated diseases. Finally, this study needs further examination and evaluation to validate the potency of Sambucus bioactive compounds.
Keyword
MeSH Terms
Figure
Reference
-
1. Medzhitov R. Cell. 2010; 140:771–776.2. Okin D, Medzhitov R. Curr Biol. 2012; 22:R733–R740.3. Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, Li Y, Wang X, Zhao L. Oncotarget. 2017; 9:7204–7218.4. Nathan C. Nature. 2002; 420:846–852.5. Liu Z, Wang Y, Wang Y, Ning Q, Zhang Y, Gong C, Zhao W, Jing G, Wang Q. Int Immunopharmacol. 2016; 35:210–216.6. Czaja AJ. World J Gastroenterol. 2014; 20:2515–2532.7. Trinchieri G. Annu Rev Immunol. 2012; 30:677–706.
Article8. Hotamisligil GS, Erbay E. Nat Rev Immunol. 2008; 8:923–934.9. Libby P. Am J Clin Nutr. 2006; 83:456S–460S.10. Wyss-Coray T, Mucke L. Neuron. 2002; 35:419–432.11. Arulselvan P, Fard MT, Tan WS, Gothai S, Fakurazi S, Norhaizan ME, Kumar SS. Oxid Med Cell Longev. 2016; 2016:5276130.12. Chen C, Huang H, Wu CH. Methods Mol Biol. 2017; 1558:3–39.13. Zhou Y, Hong Y, Huang H. Kidney Blood Press Res. 2016; 41:901–910.14. Lintermans LL, Stegeman CA, Heeringa P, Abdulahad WH. Front Immunol. 2014; 5:504.15. Schmid-Schönbein GW. Annu Rev Biomed Eng. 2006; 8:93–151.16. Christensen LP, Kaack K, Fretté XC. Eur Food Res Technol. 2008; 227:293–305.17. Veberic R, Jakopic J, Stampar F, Schmitzer V. Food Chemistry. 2009; 114:511–515.18. Özgen M, Scheerens JC, Reese RN, Miller RA. Pharmacogn Mag. 2010; 6:198–203.19. Ho GT, Wangensteen H, Barsett H. Int J Mol Sci. 2017; 18:E584.20. Putra WE, Rifa'i M. Nat Prod Sci. 2019; 25:59–63.21. Stepanchikova AV, Lagunin AA, Filimonov DA, Poroikov VV. Curr Med Chem. 2003; 10:225–233.22. Kim S, Thiessen PA, Bolton EE, Chen J, Fu G, Gindulyte A, Han L, He J, He S, Shoemaker BA, Wang J, Yu B, Zhang J, Bryant SH. Nucleic Acids Res. 2016; 44:D1202–D1213.23. Waterhouse A, Bertoni M, Bienert S, Studer G, Tauriello G, Gumienny R, Heer FT, de Beer TAP, Rempfer C, Bordoli L, Lepore R, Schwede T. Nucleic Acids Res. 2018; 46:W296–W303.24. Putra WE, Wafaretta E, Ardiana O, Januarisasi ID, Soewondo A, Rifa'i M. Bio Research. 2017; 14:201–213.25. Putra WE. FUW Trends Sci Technol J. 2018; 3:682–685.26. Trott O, Olson AJ. J Comput Chem. 2010; 31:455–461.
Article27. Liu CH, Abrams ND, Carrick DM, Chander P, Dwyer J, Hamlet MRJ, Macchiarini F, PrabhuDas M, Shen GL, Tandon P, Vedamony MM. Nat Immunol. 2017; 18:1175–1180.28. DeFronzo RA, Ferrannini E, Groop L, Henry RR, Herman WH, Holst JJ, Hu FB, Kahn CR, Raz I, Shulman GI, Simonson DC, Testa MA, Weiss R. Nat Rev Dis Primers. 2015; 1:15019.29. Fakhoury M, Negrulj R, Mooranian A, Al-Salami H. J Inflamm Res. 2014; 7:113–120.30. Yamauchi H, Woodward WA, Valero V, Alvarez RH, Lucci A, Buchholz TA, Iwamoto T, Krishnamurthy S, Yang W, Reuben JM, Hortobágyi GN, Ueno NT. Oncologist. 2012; 17:891–899.31. Dai X, ZhangJ , Arfuso F, Chinnathambi A, Zayed ME, Alharbi SA, Kumar AP, Ahn KS, Sethi G. Exp Biol Med (Maywood). 2015; 240:760–773.32. Srinivasan S, Kumar R, Koduru S, Chandramouli A, Damodaran C. Apoptosis. 2010; 15:153–161.33. Sellappan S, Akoh CC, Krewer G. J Agric Food Chem. 2002; 50:2432–2438.34. Mattila P, Kumpulainen J. J Agric Food Chem. 2002; 50:3660–3667.35. Chao CY, Mong MC, Chan KC, Yin MC. Mol Nutr Food Res. 2010; 54:388–395.36. Chao PC, Hsu CC, Yin MC. Nutr Metab (Lond). 2009; 6:33.37. Makena PS, Chung KT. Food Chem Toxicol. 2007; 45:1899–1909.38. Prakash D, Suri S, Upadhyay G, Singh BN. Int J Food Sci Nutr. 2007; 58:18–28.39. Jung UJ, Lee MK, Park YB, Jeon SM, Choi MS. J Pharmacol Exp Ther. 2006; 318:476–483.40. Armutcu F, Akyol S, Ustunsoy S, Turan FF. Exp Ther Med. 2015; 9:1582–1588.41. Jung WK, Lee DY, Kim JH, Choi I, Park SG, Seo SK, Lee SW, Lee CM, Park YM, Jeon YJ, Lee CH, Jeon BT, Qian ZJ, Kim SK, Choi IW. Process Biochem. 2008; 43:783–787.42. Serreli G, Deiana M. Antioxidants (Basel). 2018; 7:E170.43. Valko-Rokytovská M, Očenáš P, Salayová A, Kostecká Z. J Chromatogr Sep Tech. 2018; 9:1–8.44. Bonifačić D, Aralica M, Sotošek Tokmadžić V, Rački V, Tuškan-Mohar L, Kučić N. Biomarkers. 2017; 22:790–797.45. Tuck KL, Hayball PJ, Stupans I. J Agric Food Chem. 2002; 50:2404–2409.46. Patil R, Das S, Stanley A, Yadav L, Sudhakar A, Varma AK. PLoS One. 2010; 5:e12029.47. Hladílková J, Callisen TH, Lund M. J Phys Chem B. 2016; 120:3303–3310.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Molecular docking of bioactive compounds derived from Moringa oleifera with p53 protein in the apoptosis pathway of oral squamous cell carcinoma
- Mushrooms: An Important Source of Natural Bioactive Compounds
- Phenolic Constituents from Balanophora laxiflora with their Anti-inflammatory and Cytotoxic Effects
- Human Brain Astrocytes Mediate TRAIL-mediated Apoptosis after Treatment with IFN-gamma
- Antioxidant and Anti-inflammatory Activities of Rumex acetosa