Chonnam Med J.
2018 Jan;54(1):17-23. 10.4068/cmj.2018.54.1.17.
Salicylic Acid Reduces OmpF Expression, Rendering Salmonella enterica Serovar Typhimurium More Resistant to Cephalosporin Antibiotics
- Affiliations
-
- 1Department of Microbiology and Immunology, Chonnam National University Medical School, Gwangju, Korea. pyryu@jnu.ac.kr
- 2Molecular Imaging Research Center, Korea Institute of Radiological and Medical Sciences, Seoul, Korea.
- KMID: 2458580
- DOI: http://doi.org/10.4068/cmj.2018.54.1.17
Abstract
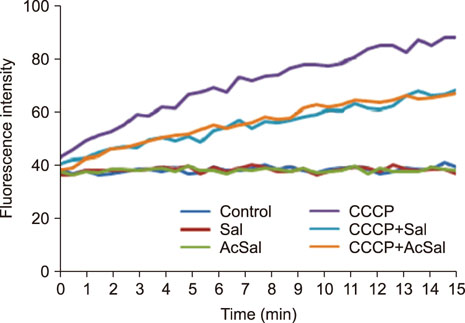
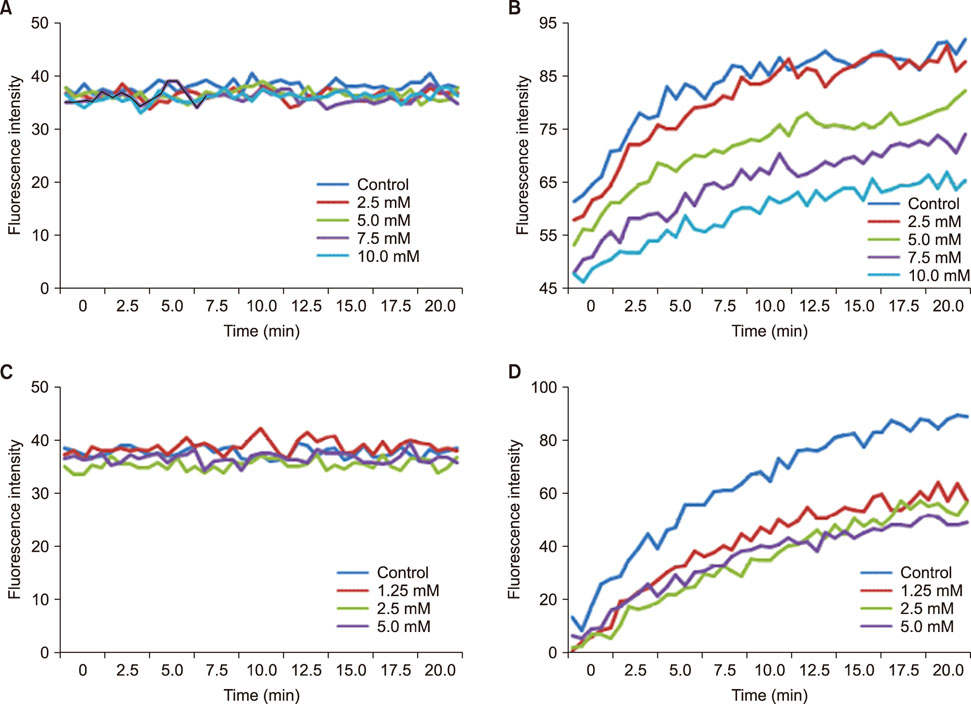
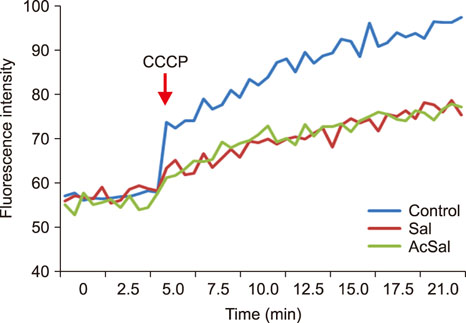
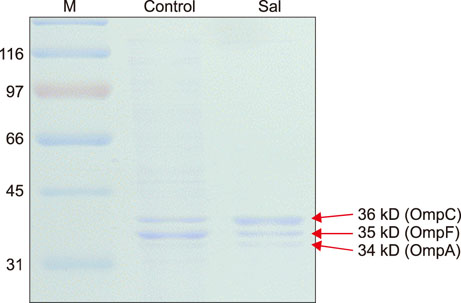
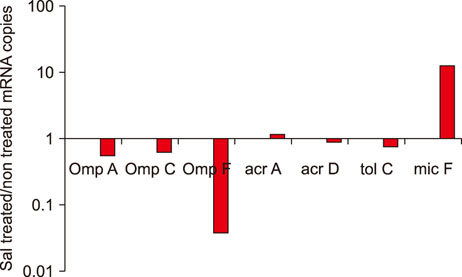
- Salmonella enterica serovar Typhimurium is one of the most important bacterial pathogens causing diarrhea. The resistance of S. typhimurium to antimicrobial agents, which has recently been isolated from patients, is causing serious problems. We investigated the effects of salicylic acid (Sal) and acetyl salicylate (AcSal) on the susceptibility of S. typhimurium to cephalosporin antibiotics, which are known to increase resistance to cephalosporin and quinolone antibiotics. The MIC of cephalosporin antibiotics was higher than that of the media without Sal. The rate of accumulation of ethidium bromide (EtBr) in the bacteria by the outer membrane protein (Omp) was not different from that of the bacteria cultured in the medium containing Sal. However, Carbonyl cyanide-m-chlorophenylhydrazone (CCCP), an inhibitor of bacterial efflux pumps, significantly reduced the rate of accumulation of EtBr in bacteria cultured on Sal containing medium. In the medium containing CCCP, the MIC of the antimicrobial agent tended to decrease as compared with the control. In addition, the MIC of the bacteria treated with CCCP and Sal was higher than that of the antimicrobial agent against the CCCP treated experimental bacteria. These results suggest that Sal decreases the expression of OmpF in the Omp of S. typhimurium and reduces the permeability of cephalosporin antibiotics to bacteria, which may induce tolerance to cephalosporin antibiotics.
Keyword
MeSH Terms
-
Anti-Bacterial Agents
Anti-Infective Agents
Bacteria
Carbonyl Cyanide m-Chlorophenyl Hydrazone
Cephalosporin Resistance
Cephalosporins*
Diarrhea
Ethidium
Humans
Membrane Proteins
Permeability
Salicylic Acid*
Salmonella enterica*
Salmonella typhimurium
Salmonella*
Serogroup*
Anti-Bacterial Agents
Anti-Infective Agents
Carbonyl Cyanide m-Chlorophenyl Hydrazone
Cephalosporins
Ethidium
Membrane Proteins
Salicylic Acid
Figure
Reference
-
1. Cohen SP, McMurry LM, Hooper DC, Wolfson JS, Levy SB. Cross-resistance to fluoroquinolones in multiple-antibiotic- resistant (Mar) Escherichia coli selected by tetracycline or chloramphenicol: decreased drug accumulation associated with membrane changes in addition to OmpF reduction. Antimicrob Agents Chemother. 1989; 33:1318–1325.
Article2. Arlet G, Barrett TJ, Butaye P, Cloeckaert A, Mulvey MR, White DG. Salmonella resistant to extended-spectrum cephalosporins: prevalence and epidemiology. Microbes Infect. 2006; 8:1945–1954.
Article3. Baucheron S, Tyler S, Boyd D, Mulvey MR, Chaslus-Dancla E, Cloeckaert A. AcrAB-TolC directs efflux-mediated multidrug resistance in Salmonella enterica serovar typhimurium DT104. Antimicrob Agents Chemother. 2004; 48:3729–3735.
Article4. Eaves DJ, Ricci V, Piddock LJ. Expression of acrB, acrF, acrD, marA, and soxS in Salmonella enterica serovar Typhimurium: role in multiple antibiotic resistance. Antimicrob Agents Chemother. 2004; 48:1145–1150.
Article5. Medeiros AA, O'Brien TF, Rosenberg EY, Nikaido H. Loss of OmpC porin in a strain of Salmonella typhimurium causes increased resistance to cephalosporins during therapy. J Infect Dis. 1987; 156:751–757.
Article6. Miró E, Vergés C, García I, Mirelis B, Navarro F, Coll P, et al. Resistance to quinolones and beta-lactams in Salmonella enterica due to mutations in topoisomerase-encoding genes, altered cell permeability and expression of an active efflux system. Enferm Infecc Microbiol Clin. 2004; 22:204–211.
Article7. Price CT, Lee IR, Gustafson JE. The effects of salicylate on bacteria. Int J Biochem Cell Biol. 2000; 32:1029–1043.
Article8. Vane JR, Flower RJ, Botting RM. History of aspirin and its mechanism of action. Stroke. 1990; 21:12 Suppl. IV12–IV23.9. Coban AY, Birinci A, Ekinci B, Durupinar B. Effects of acetyl salicylate and ibuprofen on fluoroquinolone MICs on Salmonella enterica serovar typhimurium in vitro. J Chemother. 2004; 16:128–133.
Article10. Riordan JT, Muthaiyan A, Van Voorhies W, Price CT, Graham JE, Wilkinson BJ, et al. Response of Staphylococcus aureus to salicylate challenge. J Bacteriol. 2007; 189:220–227.
Article11. Randall LP, Woodward MJ. Role of the mar locus in virulence of Salmonella enterica serovar Typhimurium DT104 in chickens. J Med Microbiol. 2001; 50:770–779.
Article12. Hartog E, Menashe O, Kler E, Yaron S. Salicylate reduces the antimicrobial activity of ciprofloxacin against extracellular Salmonella enterica serovar Typhimurium, but not against Salmonella in macrophages. J Antimicrob Chemother. 2010; 65:888–896.
Article13. Stalons DR, Thornsberry C. Broth-dilution method for determining the antibiotic susceptibility of anaerobic bacteria. Antimicrob Agents Chemother. 1975; 7:15–21.
Article14. Cockerill FR, Wikler MA, Alder J, Dudley MN, Eliopoulos GM, Ferraro MJ, et al. Performance standards for antimicrobial disk susceptibility tests; approved standard. 11th ed. Wayne, PA: :Clinical and Laboratory Standards Institute;2012.15. Kiehlbauch JA, Hannett GE, Salfinger M, Archinal W, Monserrat C, Carlyn C. Use of the National Committee for Clinical Laboratory Standards guidelines for disk diffusion susceptibility testing in New York state laboratories. J Clin Microbiol. 2000; 38:3341–3348.
Article16. Aoyama H, Sato K, Kato T, Hirai K, Mitsuhashi S. Norfloxacin resistance in a clinical isolate of Escherichia coli. Antimicrob Agents Chemother. 1987; 31:1640–1641.
Article17. Martinez MB, Flickinger M, Higgins L, Krick T, Nelsestuen GL. Reduced outer membrane permeability of Escherichia coli O157:H7: suggested role of modified outer membrane porins and theoretical function in resistance to antimicrobial agents. Biochemistry. 2001; 40:11965–11974.
Article18. Baranova NN, Neyfakh AA. Apparent involvement of a multidrug transporter in the fluoroquinolone resistance of Streptococcus pneumoniae. Antimicrob Agents Chemother. 1997; 41:1396–1398.
Article19. Paixão L, Rodrigues L, Couto I, Martins M, Fernandes P, de Carvalho CC, et al. Fluorometric determination of ethidium bromide efflux kinetics in Escherichia coli. J Biol Eng. 2009; 3:18.
Article20. Cohen SP, McMurry LM, Levy SB. marA locus causes decreased expression of OmpF porin in multiple-antibiotic-resistant (Mar) mutants of Escherichia coli. J Bacteriol. 1988; 170:5416–5422.
Article21. Cohen SP, Levy SB, Foulds J, Rosner JL. Salicylate induction of antibiotic resistance in Escherichia coli: activation of the mar operon and a mar-independent pathway. J Bacteriol. 1993; 175:7856–7862.
Article22. Hutsul JA, Worobec E. Molecular characterization of the Serratia marcescens OmpF porin, and analysis of S. marcescens OmpF and OmpC osmoregulation. Microbiology. 1997; 143:2797–2806.
Article23. Rosner JL, Chai TJ, Foulds J. Regulation of ompF porin expression by salicylate in Escherichia coli. J Bacteriol. 1991; 173:5631–5638.
Article24. Rodrigue DC, Tauxe RV, Rowe B. International increase in Salmonella enteritidis: a new pandemic? Epidemiol Infect. 1990; 105:21–27.
Article25. Chalker RB, Blaser MJ. A review of human salmonellosis: III. Magnitude of Salmonella infection in the United States. Rev Infect Dis. 1988; 10:111–124.
Article26. Lee LA, Puhr ND, Maloney EK, Bean NH, Tauxe RV. Increase in antimicrobial-resistant Salmonella infections in the United States, 1989–1990. J Infect Dis. 1994; 170:128–134.
Article27. Yang SJ, Park KY, Kim SH, No KM, Besser TE, Yoo HS, et al. Antimicrobial resistance in Salmonella enterica serovars Enteritidis and Typhimurium isolated from animals in Korea: comparison of phenotypic and genotypic resistance characterization. Vet Microbiol. 2002; 86:295–301.
Article28. Neu HC. The crisis in antibiotic resistance. Science. 1992; 257:1064–1073.
Article29. Nikaido H. Outer membrane barrier as a mechanism of antimicrobial resistance. Antimicrob Agents Chemother. 1989; 33:1831–1836.
Article30. Osborn MJ, Wu HC. Proteins of the outer membrane of gram-negative bacteria. Annu Rev Microbiol. 1980; 34:369–422.
Article31. Sawai T, Hirano S, Yamaguchi A. Repression of porin synthesis by salicylate in Escherichia coli, Klebsiella pneumoniae and Serratia marcescens. FEMS Microbiol Lett. 1987; 40:233–237.
Article32. Sulavik MC, Dazer M, Miller PF. The Salmonella typhimurium mar locus: molecular and genetic analyses and assessment of its role in virulence. J Bacteriol. 1997; 179:1857–1866.
Article33. Alekshun MN, Levy SB. Alteration of the repressor activity of MarR, the negative regulator of the Escherichia coli marRAB locus, by multiple chemicals in vitro. J Bacteriol. 1999; 181:4669–4672.
Article34. Martin RG, Rosner JL. Binding of purified multiple antibiotic-resistance repressor protein (MarR) to mar operator sequences. Proc Natl Acad Sci U S A. 1995; 92:5456–5460.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Salmonella Serovars from Foodborne and Waterborne Diseases in Korea, 1998-2007: Total Isolates Decreasing Versus Rare Serovars Emerging
- Modulation of Humoral and Cell-Mediated Immunity Against Avian Influenza and Newcastle Disease Vaccines by Oral Administration of Salmonella enterica Serovar Typhimurium Expressing Chicken Interleukin-18
- Characteristics of Epidemic Multidrugresistant Salmonella enterica Serovar Typhimurium DT104 Strains First Isolated in Korea
- Prevalence of Specific Clone of Salmonella enterica Serovar Typhimurium Clones in the Swine Population of Kyungpook Province During 1998 to 2000
- Detection of Antibiotic Resistant Genes in Salmonella enterica Serovar Typhimurium Isolated from Foodborne Patients in Seoul Using Multiplex-PCR