Ann Lab Med.
2020 Jan;40(1):21-26. 10.3343/alm.2020.40.1.21.
Adjustment of Modified Carbapenem Inactivation Method Conditions for Rapid Detection of Carbapenemase-Producing Acinetobacter baumannii
- Affiliations
-
- 1Department of Laboratory Medicine and Research Institute of Bacterial Resistance, Yonsei University College of Medicine, Seoul, Korea. microbyun@gmail.com, jhbyun@gnuh.co.kr
- 2Brain Korea 21 PLUS Project for Medical Science, Yonsei University, Seoul, Korea.
- 3Department of Laboratory Medicine, Gyeongsang National University College of Medicine, Gyeongsang National University Hospital, Jinju, Korea.
- 4J. Craig Venter Institute, Rockville, Maryland, USA.
- KMID: 2457489
- DOI: http://doi.org/10.3343/alm.2020.40.1.21
Abstract
- BACKGROUND
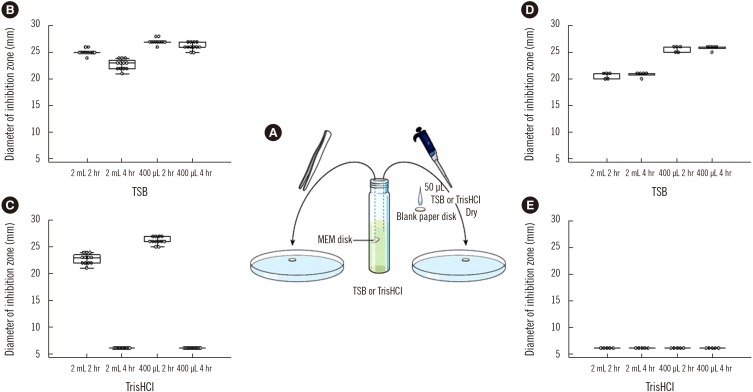
The existing modified carbapenem inactivation methods (mCIMs) recommended by the CLSI for detecting carbapenemase production have not been applicable for Acinetobacter baumannii. We evaluated the influence of matrices used in mCIMs and CIMTris on the stability of the disks for detecting carbapenemase producers and suggested optimal mCIM conditions for detecting carbapenemase-producing A. baumannii.
METHODS
Seventy-three A. baumannii isolates characterized for antimicrobial susceptibility and carbapenemase encoding genes were tested for carbapenemase production using mCIM and CIMTris. The influence of the matrices (Tryptic soy broth [TSB] and Tris-HCl) used in these methods on the stability of the meropenem (MEM) disk was also evaluated. The mCIM conditions were adjusted to enhance screening sensitivity and specificity for detecting carbapenemase-producing A. baumannii.
RESULTS
The matrices had an impact on the stability of the MEM disk after the incubation period (two or four hrs). TSB nutrient broth is an appropriate matrix for mCIM compared with Tris-HCl pH 7.6, which leads to the loss of MEM activity in CIMTris. The sensitivity and the specificity of the optimal mCIM were both 100%.
CONCLUSIONS
We established optimal mCIM conditions for simple, accurate, and reproducible detection of carbapenemase-producing A. baumannii.
MeSH Terms
Figure
Reference
-
1. Dijkshoorn L, Nemec A, Seifert H. An increasing threat in hospitals: multidrug-resistant Acinetobacter baumannii. Nat Rev Microbiol. 2007; 5:939–951. PMID: 18007677.2. Peleg AY, Seifert H, Paterson DL. Acinetobacter baumannii: Emergence of a successful pathogen. Clin Microbiol Rev. 2008; 21:538–582. PMID: 18625687.3. Poirel L, Nordmann P. Carbapenem resistance in Acinetobacter baumannii: mechanisms and epidemiology. Clin Microbiol Infect. 2006; 12:826–836. PMID: 16882287.4. Sunenshine RH, Wright MO, Maragakis LL, Harris AD, Song X, Hebden J, et al. Multidrug-resistant Acinetobacter infection mortality rate and length of hospitalization. Emerg Infect Dis. 2007; 13:97–103. PMID: 17370521.5. Queenan AM, Bush K. Carbapenemases: the versatile β-lactamases. Clin Microbiol Rev. 2007; 20:440–458. PMID: 17630334.6. Pierce VM, Simner PJ, Lonsway DR, Roe-Carpenter DE, Johnson JK, Brasso WB, et al. Modified carbapenem inactivation method for phenotypic detection of carbapenemase production among Enterobacteriaceae. J Clin Microbiol. 2017; 55:2321–2333. PMID: 28381609.7. van der Zwaluw K, de Haan A, Pluister GN, Bootsma HJ, de Neeling AJ, Schouls LM. The carbapenem inactivation method (CIM), a simple and low-cost alternative for the Carba NP test to assess phenotypic carbapenemase activity in Gram-negative rods. PLoS One. 2015; 10:e0123690. PMID: 25798828.8. Walther-Rasmussen J, Høiby N. OXA-type carbapenemases. J Antimicrob Chemother. 2006; 57:373–383. PMID: 16446375.9. Simner PJ, Opene BN, Chambers KK, Naumann ME, Carroll KC, Tamma PD. Carbapenemase detection among carbapenem-resistant glucose-nonfermenting Gram-negative bacilli. J Clin Microbiol. 2017; 55:2858–2864. PMID: 28701421.10. CLSI. Performance standards for antimicrobial susceptibility testing. 26th ed. CLSI supplement M100-S26. Wayne, PA, USA: Clinical and Laboratory Standards Institute;2016.11. CLSI. Performance standards for antimicrobial susceptibility testing. 27th ed. CLSI supplement M100-S27. Wayne, PA, USA: Clinical and Laboratory Standards Institute;2017.12. CLSI. Performance standards for antimicrobial susceptibility testing. 28th ed. CLSI supplement M100-S28. Wayne, PA, USA: Clinical and Laboratory Standards Institute;2018.13. CLSI. Test for carbapenemases in Enterobacteriaceae and Pseudomonas aeruginosa. 28th ed. CLSI supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute;2018.14. Uechi K, Tada T, Shimada K, Kuwahara-Arai K, Arakaki M, Tome T, et al. A modified carbapenem inactivation method, CIMTris, for carbapenemase production in Acinetobacter and Pseudomonas species. J Clin Microbiol. 2017; 55:3405–3410. PMID: 28954898.15. CLSI. Performance standards for antimicrobial susceptibility testing. 20th ed. CLSI supplement M100-S20. Wayne, PA, USA: Clinical and Laboratory Standards Institute;2010.16. Woodford N, Ellington MJ, Coelho JM, Turton JF, Ward ME, Brown S, et al. Multiple PCR for genes encoding prevalent OXA carbapenemases in Acinetobacter spp. Int J Antimicrob Agents. 2006; 27:351–353. PMID: 16564159.17. Bahador A, Raoofian R, Farshadzadeh Z, Beitollahi L, Khaledi A, Rahimi S, et al. The prevalence of ISAba1 and ISAba4 in Acinetobacter baumannii species of different international clone lineages among patients with burning in Tehran, Iran. Jundishapur J Microbiol. 2015; 8:e17167. PMID: 26396712.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Carbapenem Inactivation Method: Accurate Detection and Easy Interpretation of Carbapenemase Production in Enterobacteriaceae and Pseudomonas spp.
- Laboratory Diagnosis of Carbapenemase-producing Enterobacterales
- Carbapenem-Resistant Acinetobacter baumannii
- Evaluation of Six Phenotypic Methods for the Detection of Carbapenemases in Gram-Negative Bacteria With Characterized Resistance Mechanisms
- ISAba15 Inserted into Outer Membrane Protein Gene carO in Acinetobacter baumannii