Intest Res.
2019 Jul;17(3):340-348. 10.5217/ir.2019.00012.
Ustekinumab is effective in biological refractory Crohn's disease patients–regardless of approval study selection criteria
- Affiliations
-
- 1Department of Internal Medicine I, University Hospital Tübingen, Tübingen, Germany. thomas.klag@med.uni-tuebingen.de
- KMID: 2454742
- DOI: http://doi.org/10.5217/ir.2019.00012
Abstract
- BACKGROUND/AIMS
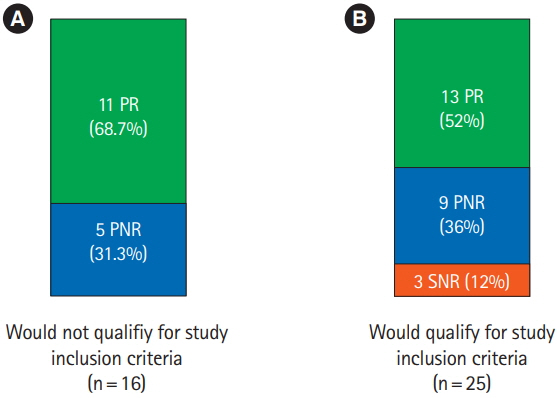
Ustekinumab is effective in active Crohn's disease. In a retrospective study, we assessed the clinical outcome in nonresponders to anti-tumor necrosis factor therapy, and/or conventional therapy and/or the α4β7-integrin inhibitor vedolizumab. As approval study populations do not always reflect the average "real world" patient cohort, we assessed weather patients who would not have qualified for approval studies show similar outcomes.
METHODS
Forty-one patients with mild to severe active Crohn's disease were treated with ustekinumab (intravenous 6 mg per kg/body weight) followed by subcutaneous ustekinumab (90 mg) at week 8. Depending on the clinical response maintenance therapy was chosen every 8 or 12 weeks. Clinical response was defined by Crohn's Disease Activity Index (CDAI) decline, decline of stool frequency or clinical improvement. Inclusion criteria for approval studies were assessed.
RESULTS
The 58.5% (24/41) showed clinical response to ustekinumab. The 58.3% of this group (14/24) achieved clinical remission. Clinical response correlated significantly with drop of stool frequency and improvement of CDAI score. The 39 out of 41 patients had no side effects and we observed no serious infections. About a third of our patients would not have met ustekinumab approval study criteria. However, patients who did not meet study criteria showed clinical improvement numerically in the same range compared to patients who would have qualified for approval studies.
CONCLUSIONS
Ustekinumab is effective, safe and well tolerated in a highly therapy refractory patient cohort. Even though a reasonable number of patients did not meet ustekinumab approval study criteria, approval study results seem to be representative to the overall patient cohort.
Keyword
MeSH Terms
Figure
Reference
-
1. Ananthakrishnan AN. Epidemiology and risk factors for IBD. Nat Rev Gastroenterol Hepatol. 2015; 12:205–217.
Article2. Wehkamp J, Stange EF. Paneth’s disease. J Crohns Colitis. 2010; 4:523–531.
Article3. Ballester Ferré MP, Boscá-Watts MM, Mínguez Pérez M. Crohn’s disease. Med Clin (Barc). 2018; 151:26–33.
Article4. Klag T, Stange EF, Wehkamp J. Defective antibacterial barrier in inflammatory bowel disease. Dig Dis. 2013; 31:310–316.
Article5. Hibi T, Imai Y, Murata Y, Matsushima N, Zheng R, Gasink C. Efficacy and safety of ustekinumab in Japanese patients with moderately to severely active Crohn’s disease: a subpopulation analysis of phase 3 induction and maintenance studies. Intest Res. 2017; 15:475–486.
Article6. Ng SC, Shi HY, Hamidi N, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2018; 390:2769–2778.
Article7. Hart AR, Luben R, Olsen A, et al. Diet in the aetiology of ulcerative colitis: a European prospective cohort study. Digestion. 2008; 77:57–64.
Article8. Ananthakrishnan AN, Cagan A, Gainer VS, et al. Normalization of plasma 25-hydroxy vitamin D is associated with reduced risk of surgery in Crohn’s disease. Inflamm Bowel Dis. 2013; 19:1921–1927.
Article9. Sonnenberg A. Occupational distribution of inflammatory bowel disease among German employees. Gut. 1990; 31:1037–1040.
Article10. Ananthakrishnan AN, Khalili H, Konijeti GG, et al. Sleep duration affects risk for ulcerative colitis: a prospective cohort study. Clin Gastroenterol Hepatol. 2014; 12:1879–1886.
Article11. Klag T, Mazurak N, Fantasia L, et al. High demand for psychotherapy in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2017; 23:1796–1802.
Article12. Amiot A, Peyrin-Biroulet L. Current, new and future biological agents on the horizon for the treatment of inflammatory bowel diseases. Therap Adv Gastroenterol. 2015; 8:66–82.
Article13. Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet. 2002; 359:1541–1549.
Article14. Rutgeerts P, Feagan BG, Lichtenstein GR, et al. Comparison of scheduled and episodic treatment strategies of infliximab in Crohn’s disease. Gastroenterology. 2004; 126:402–413.
Article15. Sandborn WJ, Rutgeerts P, Enns R, et al. Adalimumab induction therapy for Crohn disease previously treated with infliximab: a randomized trial. Ann Intern Med. 2007; 146:829–838.
Article16. Sands BE, Sandborn WJ, Van Assche G, et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease in patients naïve to or who have failed tumor necrosis factor antagonist therapy. Inflamm Bowel Dis. 2017; 23:97–106.
Article17. Frolkis AD, Lipton DS, Fiest KM, et al. Cumulative incidence of second intestinal resection in Crohn’s disease: a systematic review and meta-analysis of population-based studies. Am J Gastroenterol. 2014; 109:1739–1748.
Article18. Gisbert JP, Marín AC, McNicholl AG, Chaparro M. Systematic review with meta-analysis: the efficacy of a second anti-TNF in patients with inflammatory bowel disease whose previous anti-TNF treatment has failed. Aliment Pharmacol Ther. 2015; 41:613–623.
Article19. Feagan BG, Schwartz D, Danese S, et al. Efficacy of vedolizumab in fistulising Crohn’s disease: exploratory analyses of data from GEMINI 2. J Crohns Colitis. 2018; 12:621–626.
Article20. Ford AC, Bernstein CN, Khan KJ, et al. Glucocorticosteroid therapy in inflammatory bowel disease: systematic review and meta-analysis. Am J Gastroenterol. 2011; 106:590–599.
Article21. Estevinho MM, Afonso J, Rosa I, et al. A systematic review and meta-analysis of 6-thioguanine nucleotide levels and clinical remission in inflammatory bowel disease. J Crohns Colitis. 2017; 11:1381–1392.
Article22. Monteleone G, Biancone L, Marasco R, et al. Interleukin 12 is expressed and actively released by Crohn’s disease intestinal lamina propria mononuclear cells. Gastroenterology. 1997; 112:1169–1178.
Article23. Simon EG, Ghosh S, Iacucci M, Moran GW. Ustekinumab for the treatment of Crohn’s disease: can it find its niche? Therap Adv Gastroenterol. 2016; 9:26–36.
Article24. Berrebi D, Besnard M, Fromont-Hankard G, et al. Interleukin-12 expression is focally enhanced in the gastric mucosa of pediatric patients with Crohn’s disease. Am J Pathol. 1998; 152:667–672.25. Barré A, Colombel JF, Ungaro R. Review article: predictors of response to vedolizumab and ustekinumab in inflammatory bowel disease. Aliment Pharmacol Ther. 2018; 47:896–905.
Article26. Sandborn WJ, Gasink C, Gao LL, et al. Ustekinumab induction and maintenance therapy in refractory Crohn’s disease. N Engl J Med. 2012; 367:1519–1528.
Article27. Feagan BG, Sandborn WJ, Gasink C, et al. Ustekinumab as induction and maintenance therapy for Crohn’s disease. N Engl J Med. 2016; 375:1946–1960.28. Ha C, Ullman TA, Siegel CA, Kornbluth A. Patients enrolled in randomized controlled trials do not represent the inflammatory bowel disease patient population. Clin Gastroenterol Hepatol. 2012; 10:1002–1007.
Article29. Koelewijn CL, Schwartz MP, Samsom M, Oldenburg B. C-reactive protein levels during a relapse of Crohn’s disease are associated with the clinical course of the disease. World J Gastroenterol. 2008; 14:85–89.
Article30. Kopylov U, Afif W, Cohen A, et al. Subcutaneous ustekinumab for the treatment of anti-TNF resistant Crohn’s disease: the McGill experience. J Crohns Colitis. 2014; 8:1516–1522.
Article31. Wils P, Bouhnik Y, Michetti P, et al. Long-term efficacy and safety of ustekinumab in 122 refractory Crohn’s disease patients: a multicentre experience. Aliment Pharmacol Ther. 2018; 47:588–595.
Article32. Harris KA, Horst S, Gadani A, et al. Patients with refractory crohn’s disease successfully treated with ustekinumab. Inflamm Bowel Dis. 2016; 22:397–401.
Article33. Khorrami S, Ginard D, Marín-Jiménez I, et al. Ustekinumab for the treatment of refractory Crohn’s disease: the Spanish experience in a large multicentre open-label cohort. Inflamm Bowel Dis. 2016; 22:1662–1669.34. Fagan EA, Dyck RF, Maton PN, et al. Serum levels of C-reactive protein in Crohn’s disease and ulcerative colitis. Eur J Clin Invest. 1982; 12:351–359.
Article35. Filik L, Dagli U, Ulker A. C-reactive protein and monitoring the activity of Crohn’s disease. Adv Ther. 2006; 23:655–662.
Article36. Chamouard P, Richert Z, Meyer N, Rahmi G, Baumann R. Diagnostic value of C-reactive protein for predicting activity level of Crohn’s disease. Clin Gastroenterol Hepatol. 2006; 4:882–887.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Phase 3 Trials of Ustekinumab as Induction and Maintenance Therapy for Crohn's Disease
- Ustekinumab for the treatment of refractory pediatric Crohn’s disease: a single-center experience
- Emerging Therapies: What Are Promising in the Near Future?
- The Efficacy of Ustekinumab in the Remission Induction and Maintenance of Refractory Crohn's Disease
- Treatment of inflammatory bowel diseases: focusing on biologic agents and new therapies