J Clin Neurol.
2019 Jan;15(1):27-37. 10.3988/jcn.2019.15.1.27.
Qualitative Comparison of Semantic Memory Impairment in Patients with Amnestic Mild Cognitive Impairment Based on β-Amyloid Status
- Affiliations
-
- 1Department of Neurology, University of Ulsan College of Medicine, Gangneung Asan Hospital, Gangneung, Korea.
- 2Department of Neurology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea. jhlee@amc.seoul.kr
- 3Biomedical Research Institute, Pusan National University Hospital, Busan, Korea.
- 4Department of Neurology, Keimyung University College of Medicine, Dongsan Medical Center, Daegu, Korea.
- 5Department of Nuclear Medicine, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea.
- KMID: 2451142
- DOI: http://doi.org/10.3988/jcn.2019.15.1.27
Abstract
- BACKGROUND AND PURPOSE
Semantic memory remains more stable than episodic memory across the lifespan, which makes it potentially useful as a marker for distinguishing pathological aging from normal senescence. To obtain a better understanding of the transitional stage evolving into Alzheimer's dementia (AD), we focused on the amnestic mild cognitive impairment (aMCI) stage stratified based on β-amyloid (Aβ) pathology.
METHODS
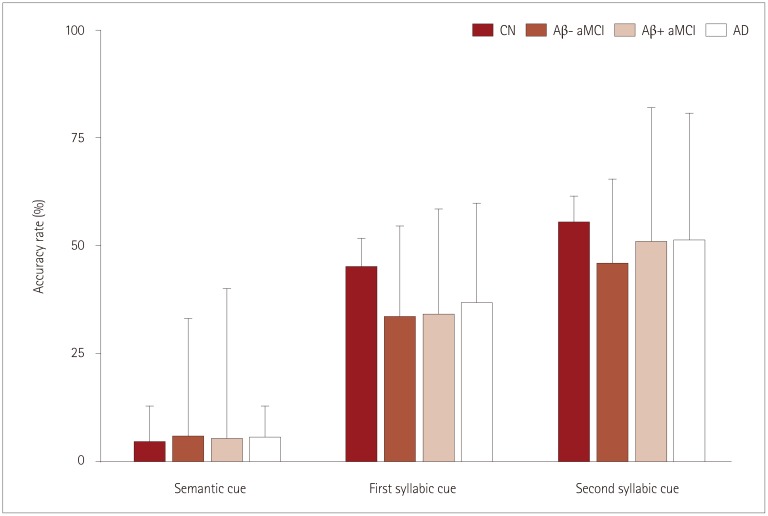
We analyzed the raw data from Korean version of the Boston Naming Test (K-BNT) and the Controlled Oral Word Association Test (COWAT). For K-BNT, the frequencies of six error types and accuracy rates were evaluated. For a qualitative assessment of the COWAT, we computed the number of switching, number of clusters, and mean cluster size.
RESULTS
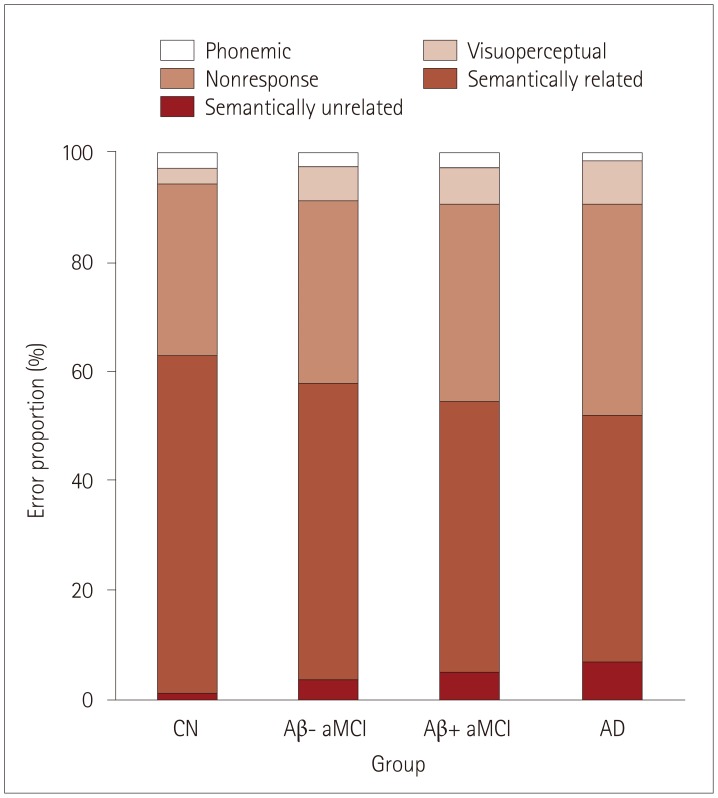
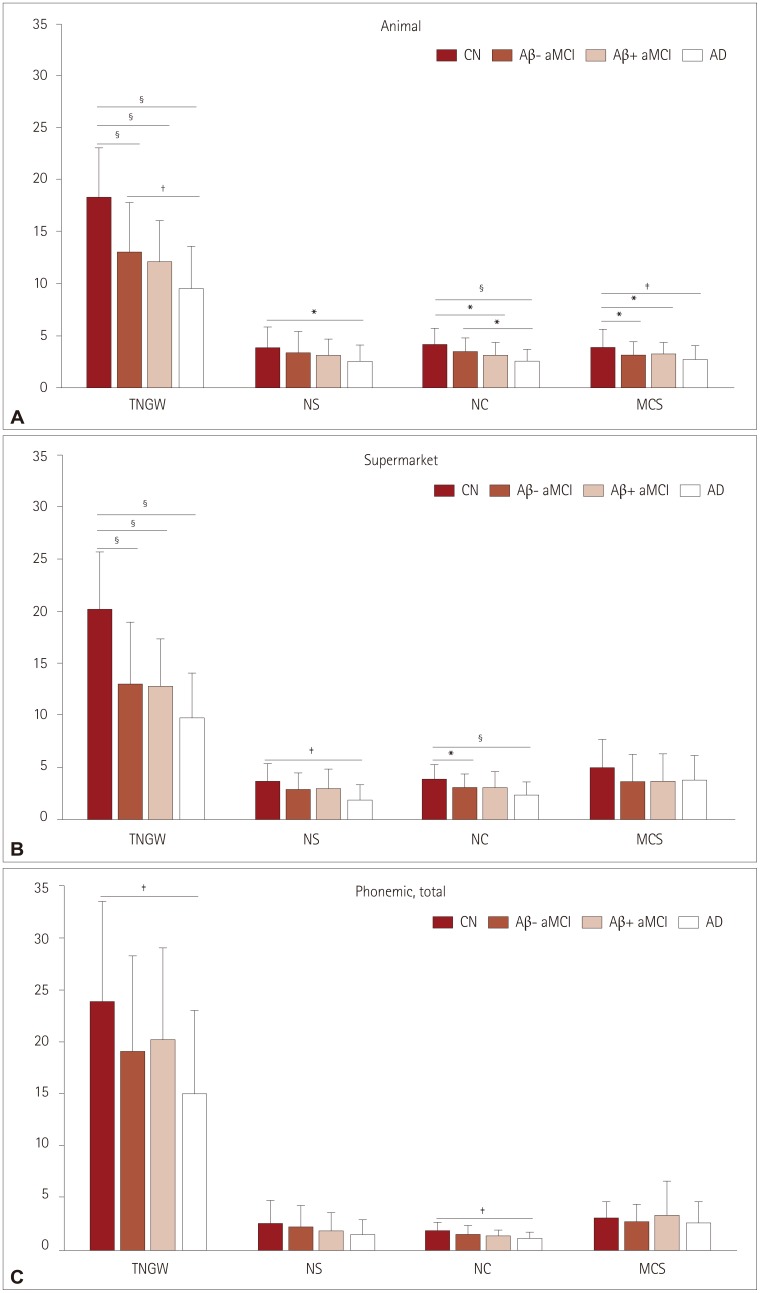
The data from 217 participants were analyzed (53 normal controls, 66 with Aβ− aMCI, 56 with Aβ+ aMCI, and 42 disease controls). There were fewer semantically related errors and more semantically unrelated errors on the K-BNT in Aβ+ aMCI than in Aβ− aMCI, without a gross difference in the z score. We also found that Aβ+ aMCI showed a more prominent deficit in the number of clusters in the semantic fluency task [especially for animal names (living items)] than Aβ− aMCI.
CONCLUSIONS
In spite of similar clinical manifestations, Aβ+ aMCI was more similar to AD than Aβ− aMCI in terms of semantic memory disruption. Semantic memory may serve as an early indicator of brain Aβ pathology. Therefore, semantic memory dysfunction deserves more consideration in clinical practice. Longitudinal research with the follow-up data is needed.
MeSH Terms
Figure
Reference
-
1. Tulving E. Multiple memory systems and consciousness. Hum Neurobiol. 1987; 6:67–80. PMID: 3305441.2. Squire LR, Wixted JT. The cognitive neuroscience of human memory since H.M. Annu Rev Neurosci. 2011; 34:259–288. PMID: 21456960.
Article3. Squire LR, Zola-Morgan S. The medial temporal lobe memory system. Science. 1991; 253:1380–1386. PMID: 1896849.
Article4. Ribeiro F, de Mendonça A, Guerreiro M. Mild cognitive impairment: deficits in cognitive domains other than memory. Dement Geriatr Cogn Disord. 2006; 21:284–290. PMID: 16484806.
Article5. Portet F, Ousset PJ, Visser PJ, Frisoni GB, Nobili F, Scheltens P, et al. Mild cognitive impairment (MCI) in medical practice: a critical review of the concept and new diagnostic procedure. Report of the MCI Working Group of the European Consortium on Alzheimer's disease. J Neurol Neurosurg Psychiatry. 2006; 77:714–718. PMID: 16549412.
Article6. Choi H, Kim JH, Lee CM, Kim JI. Features of semantic language impairment in patients with amnestic mild cognitive impairment. Dement Neurocognitive Disord. 2013; 12:33–40.
Article7. Duong A, Whitehead V, Hanratty K, Chertkow H. The nature of lexico-semantic processing deficits in mild cognitive impairment. Neuropsychologia. 2006; 44:1928–1935. PMID: 16616942.
Article8. Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA. MR signal abnormalities at 1.5 T in Alzheimer's dementia and normal aging. AJR Am J Roentgenol. 1987; 149:351–356. PMID: 3496763.
Article9. Petersen RC, Doody R, Kurz A, Mohs RC, Morris JC, Rabins PV, et al. Current concepts in mild cognitive impairment. Arch Neurol. 2001; 58:1985–1992. PMID: 11735772.
Article10. McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7:263–269. PMID: 21514250.
Article11. Huff FJ, Corkin S, Growdon JH. Semantic impairment and anomia in Alzheimer's disease. Brain Lang. 1986; 28:235–249. PMID: 3730816.
Article12. Adlam AL, Bozeat S, Arnold R, Watson P, Hodges JR. Semantic knowledge in mild cognitive impairment and mild Alzheimer's disease. Cortex. 2006; 42:675–684. PMID: 16909626.
Article13. Rosser A, Hodges JR. Initial letter and semantic category fluency in Alzheimer's disease, Huntington's disease, and progressive supranuclear palsy. J Neurol Neurosurg Psychiatry. 1994; 57:1389–1394. PMID: 7964817.
Article14. Kim H, Kim EY, Na DL. Naming deficits in patients with dementia of the Alzheimer type: error analysis of Korean version-Boston Naming Test. J Korean Neurol Assoc. 1997; 15:1012–1021.15. Yoon JH, Na DL, Chin J, Ahn HJ, Lee BH, Suh MK, et al. Is phonological-lexical representation preserved in moderate stage Alzheimer disease? Evidence from the efficacy of Korean syllabic cues. Alzheimer Dis Assoc Disord. 2010; 24:334–338. PMID: 20811269.
Article16. Troyer AK, Moscovitch M, Winocur G. Clustering and switching as two components of verbal fluency: evidence from younger and older healthy adults. Neuropsychology. 1997; 11:138–146. PMID: 9055277.
Article17. Kim J, Yoon JH, Kim SR, Kim H. Effect of literacy level on cognitive and language tests in Korean illiterate older adults. Geriatr Gerontol Int. 2014; 14:911–917. PMID: 24261573.
Article18. Kim JW, Kim HH. Animal naming performance in Korean elderly: effects of age, education, and gender, and typicality. Int J Contents. 2012; 8:26–33.
Article19. Troyer AK. Normative data for clustering and switching on verbal fluency tasks. J Clin Exp Neuropsychol. 2000; 22:370–378. PMID: 10855044.
Article20. Cardebat D, Demonet JF, Celsis P, Puel M. Living/non-living dissociation in a case of semantic dementia: a SPECT activation study. Neuropsychologia. 1996; 34:1175–1179. PMID: 8951828.
Article21. Garrard P, Lambon Ralph MA, Watson PC, Powis J, Patterson K, Hodges JR. Longitudinal profiles of semantic impairment for living and nonliving concepts in dementia of Alzheimer's type. J Cogn Neurosci. 2001; 13:892–909. PMID: 11595093.
Article22. Lambon Ralph MA, Lowe C, Rogers TT. Neural basis of category-specific semantic deficits for living things: evidence from semantic dementia, HSVE and a neural network model. Brain. 2007; 130:1127–1137. PMID: 17438021.
Article23. Bayles KA, Tomoeda CK, Kaszniak AW, Trosset MW. Alzheimer's disease effects on semantic memory: loss of structure or impaired processing? J Cogn Neurosci. 1991; 3:166–182. PMID: 23972091.
Article24. Hodges JR, Salmon DP, Butters N. Semantic memory impairment in Alzheimer's disease: failure of access or degraded knowledge? Neuropsychologia. 1992; 30:301–314. PMID: 1603295.
Article25. Henry JD, Crawford JR, Phillips LH. Verbal fluency performance in dementia of the Alzheimer's type: a meta-analysis. Neuropsychologia. 2004; 42:1212–1222. PMID: 15178173.
Article26. Lonie JA, Herrmann LL, Tierney KM, Donaghey C, O'Carroll R, Lee A, et al. Lexical and semantic fluency discrepancy scores in aMCI and early Alzheimer's disease. J Neuropsychol. 2009; 3:79–92. PMID: 19338718.
Article27. Warrington EK, Shallice T. Category specific semantic impairments. Brain. 1984; 107:829–854. PMID: 6206910.
Article28. Estévez-González A, García-Sánchez C, Boltes A, Otermín P, Pascual-Sedano B, Gironell A, et al. Semantic knowledge of famous people in mild cognitive impairment and progression to Alzheimer's disease. Dement Geriatr Cogn Disord. 2004; 17:188–195. PMID: 14739543.
Article29. Joubert S, Felician O, Barbeau EJ, Didic M, Poncet M, Ceccaldi M. Patterns of semantic memory impairment in mild cognitive impairment. Behav Neurol. 2008; 19:35–40. PMID: 18413914.
Article30. Binder JR, Desai RH, Graves WW, Conant LL. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex. 2009; 19:2767–2796. PMID: 19329570.
Article31. Barbeau EJ, Didic M, Joubert S, Guedj E, Koric L, Felician O, et al. Extent and neural basis of semantic memory impairment in mild cognitive impairment. J Alzheimers Dis. 2012; 28:823–837. PMID: 22112550.
Article32. Joubert S, Brambati SM, Ansado J, Barbeau EJ, Felician O, Didic M, et al. The cognitive and neural expression of semantic memory impairment in mild cognitive impairment and early Alzheimer's disease. Neuropsychologia. 2010; 48:978–988. PMID: 19954747.
Article33. Hyman BT, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Carrillo MC, et al. National Institute on Aging-Alzheimer's Association guidelines for the neuropathologic assessment of Alzheimer's disease. Alzheimers Dement. 2012; 8:1–13. PMID: 22265587.
Article34. Balthazar ML, Martinelli JE, Cendes F, Damasceno BP. Lexical semantic memory in amnestic mild cognitive impairment and mild Alzheimer's disease. Arq Neuropsiquiatr. 2007; 65:619–622. PMID: 17876402.
Article35. Price SE, Kinsella GJ, Ong B, Storey E, Mullaly E, Phillips M, et al. Semantic verbal fluency strategies in amnestic mild cognitive impairment. Neuropsychology. 2012; 26:490–497. PMID: 22746308.
Article36. Woodard JL, Seidenberg M, Nielson KA, Antuono P, Guidotti L, Durgerian S, et al. Semantic memory activation in amnestic mild cognitive impairment. Brain. 2009; 132:2068–2078. PMID: 19515831.
Article37. Nelson PT, Jicha GA, Schmitt FA, Liu H, Davis DG, Mendiondo MS, et al. Clinicopathologic correlations in a large Alzheimer disease center autopsy cohort: neuritic plaques and neurofibrillary tangles “do count” when staging disease severity. J Neuropathol Exp Neurol. 2007; 66:1136–1146. PMID: 18090922.38. Nelson PT, Alafuzoff I, Bigio EH, Bouras C, Braak H, Cairns NJ, et al. Correlation of Alzheimer disease neuropathologic changes with cognitive status: a review of the literature. J Neuropathol Exp Neurol. 2012; 71:362–381. PMID: 22487856.
Article39. Moreaud O, David D, Charnallet A, Pellat J. Are semantic errors actually semantic?: evidence from Alzheimer's disease. Brain Lang. 2001; 77:176–186. PMID: 11300702.
Article40. Balthazar ML, Cendes F, Damasceno BP. Semantic error patterns on the Boston Naming Test in normal aging, amnestic mild cognitive impairment, and mild Alzheimer's disease: is there semantic disruption? Neuropsychology. 2008; 22:703–709. PMID: 18999343.
Article41. Lin CY, Chen TB, Lin KN, Yeh YC, Chen WT, Wang KS, et al. Confrontation naming errors in Alzheimer's disease. Dement Geriatr Cogn Disord. 2014; 37:86–94. PMID: 24107364.
Article42. Gleichgerrcht E, Fridriksson J, Bonilha L. Neuroanatomical foundations of naming impairments across different neurologic conditions. Neurology. 2015; 85:284–292. PMID: 26115732.
Article43. Rogers TT, Ivanoiu A, Patterson K, Hodges JR. Semantic memory in Alzheimer's disease and the frontotemporal dementias: a longitudinal study of 236 patients. Neuropsychology. 2006; 20:319–335. PMID: 16719625.
Article44. Henry JD, Crawford JR. A meta-analytic review of verbal fluency performance following focal cortical lesions. Neuropsychology. 2004; 18:284–295. PMID: 15099151.
Article45. Baldo JV, Schwartz S, Wilkins D, Dronkers NF. Role of frontal versus temporal cortex in verbal fluency as revealed by voxel-based lesion symptom mapping. J Int Neuropsychol Soc. 2006; 12:896–900. PMID: 17064451.
Article46. Biesbroek JM, van Zandvoort MJ, Kappelle LJ, Velthuis BK, Biessels GJ, Postma A. Shared and distinct anatomical correlates of semantic and phonemic fluency revealed by lesion-symptom mapping in patients with ischemic stroke. Brain Struct Funct. 2016; 221:2123–2134. PMID: 25939335.
Article47. Monsch AU, Bondi MW, Butters N, Salmon DP, Katzman R, Thal LJ. Comparisons of verbal fluency tasks in the detection of dementia of the Alzheimer type. Arch Neurol. 1992; 49:1253–1258. PMID: 1449404.
Article48. Rohrer D, Salmon DP, Wixted JT, Paulsen JS. The disparate effects of Alzheimer's disease and Huntington's disease on semantic memory. Neuropsychology. 1999; 13:381–388. PMID: 10447299.
Article49. Weakley A, Schmitter-Edgecombe M. Analysis of verbal fluency ability in Alzheimer's disease: the role of clustering, switching and semantic proximities. Arch Clin Neuropsychol. 2014; 29:256–268. PMID: 24687588.
Article50. Troyer AK, Moscovitch M, Winocur G, Alexander MP, Stuss D. Clustering and switching on verbal fluency: the effects of focal frontal- and temporal-lobe lesions. Neuropsychologia. 1998; 36:499–504. PMID: 9705059.
Article51. Haugrud N, Crossley M, Vrbancic M. Clustering and switching strategies during verbal fluency performance differentiate Alzheimer's disease and healthy aging. J Int Neuropsychol Soc. 2011; 17:1153–1157. PMID: 22014065.
Article52. Fagundo AB, López S, Romero M, Guarch J, Marcos T, Salamero M. Clustering and switching in semantic fluency: predictors of the development of Alzheimer's disease. Int J Geriatr Psychiatry. 2008; 23:1007–1013. PMID: 18416452.
Article53. Gonnerman LM, Andersen ES, Devlin JT, Kempler D, Seidenberg MS. Double dissociation of semantic categories in Alzheimer's disease. Brain Lang. 1997; 57:254–279. PMID: 9126416.
Article54. Whatmough C, Chertkow H, Murtha S, Templeman D, Babins L, Kelner N. The semantic category effect increases with worsening anomia in Alzheimer's type dementia. Brain Lang. 2003; 84:134–147. PMID: 12537956.
Article55. Brambati SM, Myers D, Wilson A, Rankin KP, Allison SC, Rosen HJ, et al. The anatomy of category-specific object naming in neurodegenerative diseases. J Cogn Neurosci. 2006; 18:1644–1653. PMID: 17014369.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Correlation between Sleep and C-reactive Protein of Patients in Amnestic Mild Cognitive Impairment and Alzheimer’s Dementia
- Comparison of Amyloid Positivity Rate and Accumulation Pattern between Amnestic and Non-Amnestic Type Mild Cognitive Impairment
- Anti-LGI1 Antibody Limbic Encephalitis Presented with Amnestic Mild Cognitive Impairment
- Early Detection Program Development of Amnestic Mild Cognitive Impairment in the Community : Preliminary Study
- The Effect of Clinical Characteristics and Subtypes on Amyloid Positivity in Patients with Amnestic Mild Cognitive Impairment