J Gastric Cancer.
2019 Jun;19(2):225-233. 10.5230/jgc.2019.19.e21.
Influence of Helicobacter pylori Infection on Endoscopic Findings of Gastric Adenocarcinoma of the Fundic Gland Type
- Affiliations
-
- 1Shinjuku Tsurukame Clinic, Digestive Disease Center, Tokyo, Japan. ishibashi@tsurukamekai.jp
- 2Koganei Tsurukame Clinic, Endoscopic Center, Tokyo, Japan.
- 3Department of Gastroenterology and Hepatology, Tokyo Medical and Dental University, Tokyo, Japan.
- 4Department of Pathology, Tokyo Medical and Dental University, Tokyo, Japan.
- KMID: 2449943
- DOI: http://doi.org/10.5230/jgc.2019.19.e21
Abstract
- PURPOSE
Gastric adenocarcinoma of the fundic gland type (chief cell predominant type) (GA-FG-CCP) was first reported as a rare adenocarcinoma found in the normal fundic mucosa. Recent studies have proposed the possibility that GA-FG-CCPs were also generated in the atrophic mucosa after Helicobacter pylori (HP) eradication therapy. However, little is known on the endoscopic findings of GA-FG-CCP generated in the atrophic mucosa due to its extreme rarity.
MATERIALS AND METHODS
A total of 8 patients who underwent endoscopic submucosal resection and were diagnosed with GA-FG-CCP generated in the HP-uninfected mucosa (4 cases, HP-uninfected group) or HP-eradicated atrophic mucosa (4 cases, HP-eradicated group) were retrospectively analyzed, and their endoscopic findings, including magnifying endoscopy with narrow band imaging (M-NBI), and pathological features were compared.
RESULTS
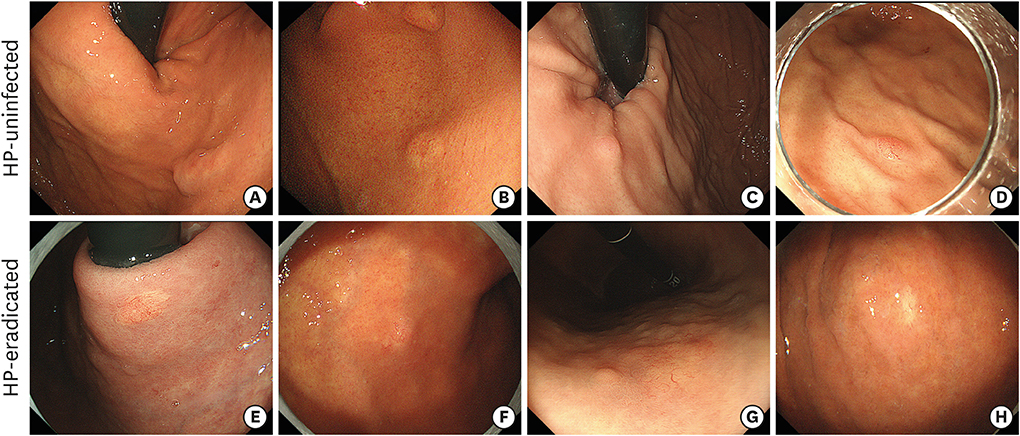
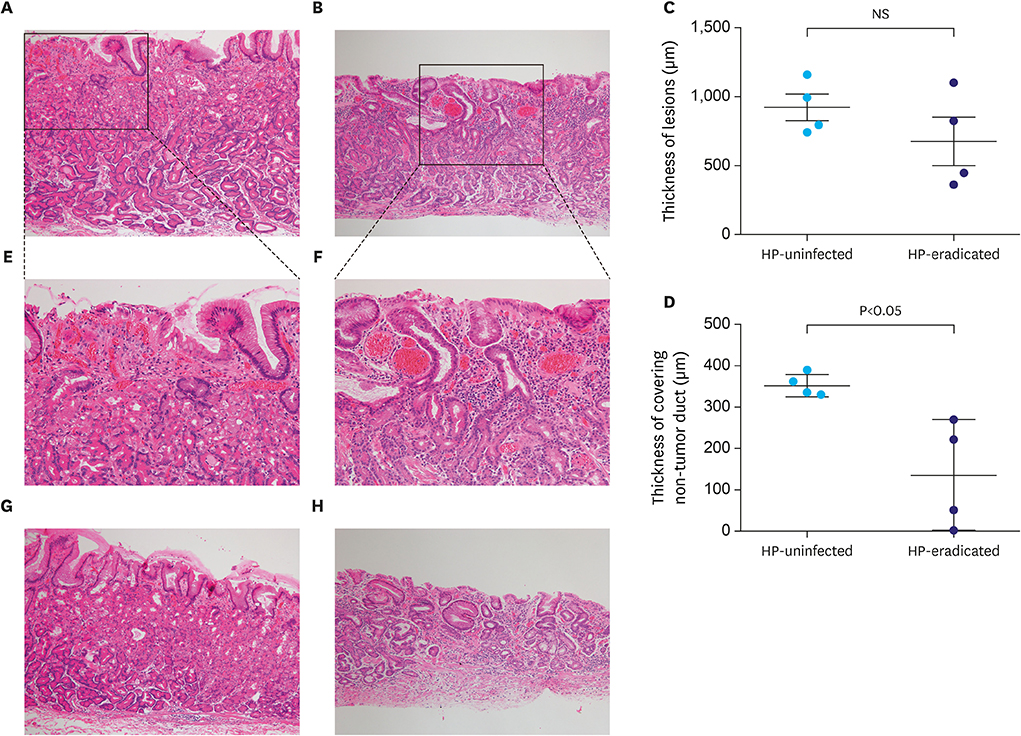
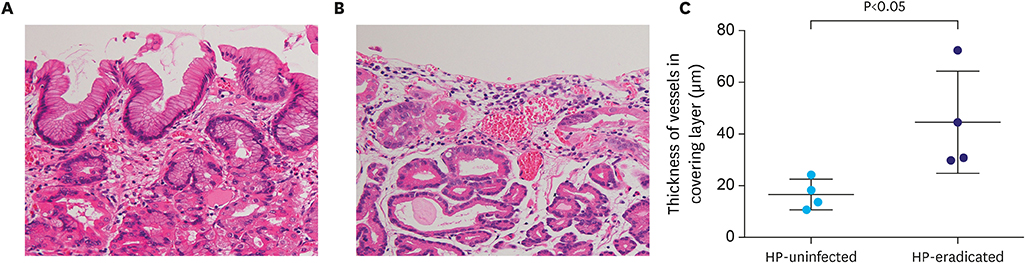
While GA-FG-CCPs in the 2 groups displayed similar macroscopic appearance, M-NBI demonstrated that characteristic microvessels (tapered microvessels like withered branches) were specifically identified in the HP-eradicated group. Pathological investigation revealed that a decreasing number of fundic glands and thinned foveolar epithelium covering tumor ducts were thought to lower the thickness of the covering layer over tumor ducts in the HP-eradicated group. Moreover, dilation of vessels just under the surface of the lesions contributed to the visualization of microvessels by M-NBI.
CONCLUSIONS
The change in background mucosa due to HP infection influenced the thickness of the covering layer over the tumor ducts and M-NBI finding of GA-FG-CCP.
MeSH Terms
Figure
Reference
-
1. Helicobacter and Cancer Collaborative Group. Gastric cancer and Helicobacter pylori: a combined analysis of 12 case control studies nested within prospective cohorts. Gut. 2001; 49:347–353.2. Wang C, Yuan Y, Hunt RH. The association between Helicobacter pylori infection and early gastric cancer: a meta-analysis. Am J Gastroenterol. 2007; 102:1789–1798.3. Nakamura K, Sugano H, Takagi K. Carcinoma of the stomach in incipient phase: its histogenesis and histological appearances. Gan. 1968; 59:251–258.4. Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. an attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965; 64:31–49.5. Moss SF. The clinical evidence linking Helicobacter pylori to gastric cancer. Cell Mol Gastroenterol Hepatol. 2016; 3:183–191.6. Ueyama H, Yao T, Nakashima Y, Hirakawa K, Oshiro Y, Hirahashi M, et al. Gastric adenocarcinoma of fundic gland type (chief cell predominant type): proposal for a new entity of gastric adenocarcinoma. Am J Surg Pathol. 2010; 34:609–619.
Article7. Ueyama H, Matsumoto K, Nagahara A, Hayashi T, Yao T, Watanabe S. Gastric adenocarcinoma of the fundic gland type (chief cell predominant type). Endoscopy. 2014; 46:153–157.
Article8. Japanese Gastric Cancer Association. Japanese Classification of Gastric Carcinoma. 15th ed. Tokyo: Kanehara Shuppan;2017.9. Miyazawa M, Matsuda M, Yano M, Hara Y, Arihara F, Horita Y, et al. Gastric adenocarcinoma of the fundic gland (chief cell-predominant type): a review of endoscopic and clinicopathological features. World J Gastroenterol. 2016; 22:10523–10531.
Article10. Miyazawa M, Matsuda M, Yano M, Hara Y, Arihara F, Horita Y, et al. Gastric adenocarcinoma of fundic gland type: five cases treated with endoscopic resection. World J Gastroenterol. 2015; 21:8208–8214.
Article11. Okumura Y, Takamatsu M, Ohashi M, Yamamoto Y, Yamamoto N, Kawachi H, et al. Gastric adenocarcinoma of fundic gland type with aggressive transformation and lymph node metastasis: a case report. J Gastric Cancer. 2018; 18:409–416.
Article12. Chiba T, Kato K, Masuda T, Ohara S, Iwama N, Shimada T, et al. Clinicopathological features of gastric adenocarcinoma of the fundic gland (chief cell predominant type) by retrospective and prospective analyses of endoscopic findings. Dig Endosc. 2016; 28:722–730.
Article13. Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011; 14:101–112.14. Sano T, Aiko T. New Japanese classifications and treatment guidelines for gastric cancer: revision concepts and major revised points. Gastric Cancer. 2011; 14:97–100.
Article15. Yao K, Iwashita A, Matsui T. A new diagnostic VS classification system produced by magnification endoscopy plus narrow-band imaging in the stomach: microvascular architecture and microsurface structure. In : Niwa H, Tajiri H, Nakajima M, Yasuda K, editors. New Challenges in Gastrointestinal Endoscopy. Tokyo: Springer;2008. p. 169–176.16. Yao K, Anagnostopoulos GK, Ragunath K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy. 2009; 41:462–467.
Article17. Kimura K, Takemoto T. An endoscopic recognition of the atrophic border and its significance in chronic gastritis. Endoscopy. 1969; 1:87–97.
Article18. Nagahama T, Yao K, Maki S, Yasaka M, Takaki Y, Matsui T, et al. Usefulness of magnifying endoscopy with narrow-band imaging for determining the horizontal extent of early gastric cancer when there is an unclear margin by chromoendoscopy (with video). Gastrointest Endosc. 2011; 74:1259–1267.
Article19. Ezoe Y, Muto M, Uedo N, Doyama H, Yao K, Oda I, et al. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology. 2011; 141:2017–2025.e3.
Article20. Muto M, Yao K, Kaise M, Kato M, Uedo N, Yagi K, et al. Magnifying endoscopy simple diagnostic algorithm for early gastric cancer (MESDA-G). Dig Endosc. 2016; 28:379–393.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Long-term Observation of Gastric Adenocarcinoma of Fundic Gland Mucosa Type before and after Helicobacter pylori Eradication: a Case Report
- Gastric Adenocarcinoma of Fundic Gland Type Treated by Endoscopic Submucosal Dissection
- Helicobacter pylori-negative Gastric Cancer
- Clinicopathological and endoscopic features of Helicobacter pylori infection-negative gastric cancer in Japan: a retrospective study
- Gastric Adenocarcinoma of Fundic-gland Type Diagnosed and Treated by Endoscopic Mucosal Resection