Int J Thyroidol.
2018 Nov;11(2):82-87. 10.11106/ijt.2018.11.2.82.
Skin-Related Toxicity of Tyrosine Kinase Inhibitor in Thyroid Cancer
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea. ldj6026@catholic.ac.kr
- KMID: 2448976
- DOI: http://doi.org/10.11106/ijt.2018.11.2.82
Abstract
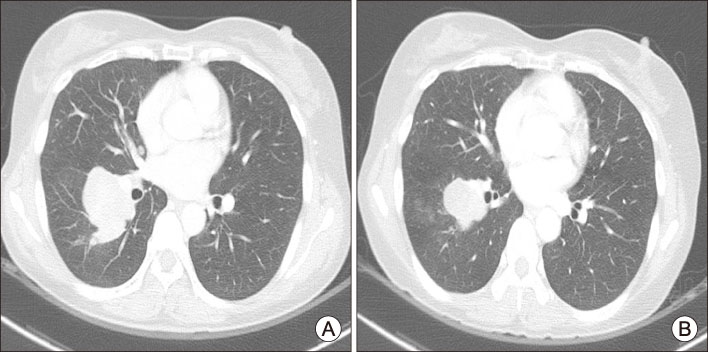
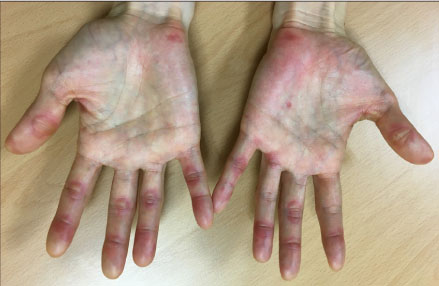
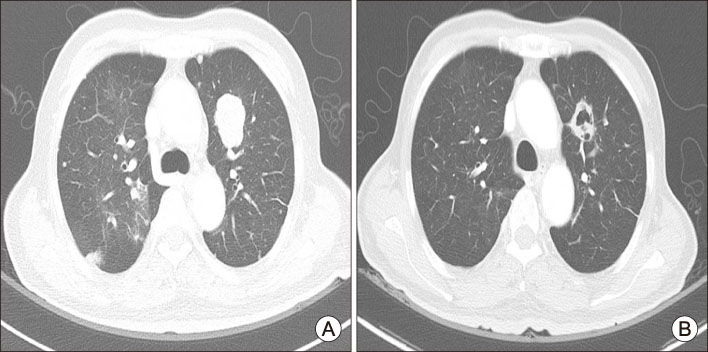
- Skin-related toxicity is one of the most important adverse events from multi-target tyrosine kinase inhibitor (MTKI) to treat radioiodine refractory thyroid cancer. As hand foot skin reaction can limit quality of life and therapeutic effectiveness, it is essential to cope with a variety of severity of skin-related toxicity induced by MTKI. Herein, we will discuss two representative cases of skin-related toxicities which were managed by discontinuation/reduction of therapeutic doses of MTKI and were treated by proper medication in thyroid cancer patients with distant metastasis.
MeSH Terms
Figure
Reference
-
1. Brose MS, Nutting CM, Jarzab B, Elisei R, Siena S, Bastholt L, et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: a randomised, double-blind, phase 3 trial. Lancet. 2014; 384(9940):319–328.
Article2. Schlumberger M, Tahara M, Wirth LJ, Robinson B, Brose MS, Elisei R, et al. Lenvatinib versus placebo in radioiodinerefractory thyroid cancer. N Engl J Med. 2015; 372(7):621–630.
Article3. Chanprapaph K, Rutnin S, Vachiramon V. Multikinase inhibitor-induced hand-foot skin reaction: A review of clinical presentation, pathogenesis, and management. Am J Clin Dermatol. 2016; 17(4):387–402.
Article4. Resteghini C, Cavalieri S, Galbiati D, Granata R, Alfieri S, Bergamini C, et al. Management of tyrosine kinase inhibitors (TKI) side effects in differentiated and medullary thyroid cancer patients. Best Pract Res Clin Endocrinol Metab. 2017; 31(3):349–361.
Article5. Jean GW, Mani RM, Jaffry A, Khan SA. Toxic effects of sorafenib in patients with differentiated thyroid carcinoma compared with other cancers. JAMA Oncol. 2016; 2(4):529–534.
Article6. Erber R, Thurnher A, Katsen AD, Groth G, Kerger H, Hammes HP, et al. Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. FASEB J. 2004; 18(2):338–340.7. Jain L, Gardner ER, Figg WD, Chernick MS, Kong HH. Lack of association between excretion of sorafenib in sweat and hand-foot skin reaction. Pharmacotherapy. 2010; 30(1):52–56.
Article8. Brose MS, Cabanillas ME, Cohen EE, Wirth LJ, Riehl T, Yue H, et al. Vemurafenib in patients with BRAF(V600E)-positive metastatic or unresectable papillary thyroid cancer refractory to radioactive iodine: a non-randomised, multicentre, open-label, phase 2 trial. Lancet Oncol. 2016; 17(9):1272–1282.
Article9. Brose MS, Frenette CT, Keefe SM, Stein SM. Management of sorafenib-related adverse events: a clinician's perspective. Semin Oncol. 2014; 41:Suppl 2. S1–S16.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Management of Severe Fatigue Induced by Tyrosine Kinase Inhibitor in Radioiodine Refractory Thyroid Cancer
- Management of Bleeding Induced by Tyrosine Kinase Inhibitor in Radioiodine Refractory Thyroid Cancer
- A Case Report of Severe Hypocalcemia and Hypothyroidism after Tyrosine Kinase Inhibitor Treatment
- Effect of Afatinib for Lung Cancer on Papillary Thyroid Carcinoma
- Sunitinib-induced hypothyroidism