J Bone Metab.
2019 May;26(2):65-74. 10.11005/jbm.2019.26.2.65.
Sarcopenia Associated with Chronic Obstructive Pulmonary Disease
- Affiliations
-
- 1Department of Rehabilitation Medicine, Biomedical Research Institute, Pusan National University Hospital, Pusan National University School of Medicine, Busan, Korea. drshinmj@gmail.com
- 2Department of Internal Medicine, Pusan National University School of Medicine, Busan, Korea.
- KMID: 2448953
- DOI: http://doi.org/10.11005/jbm.2019.26.2.65
Abstract
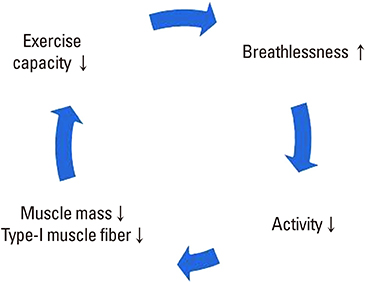
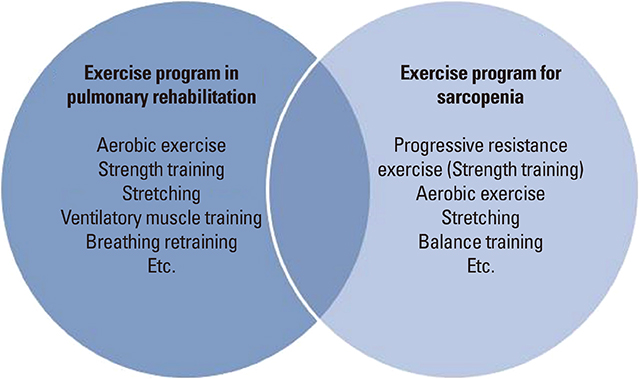
- Sarcopenia is frequently associated with chronic diseases such as chronic obstructive pulmonary disease (COPD) and cancer. COPD, which is characterized by an irreversible airflow limitation, exacerbates respiratory distress as the disease progresses. The prevalence of sarcopenia in stable COPD was reported to be 15% to 25% in previous foreign studies and 25% in a Korean study. As the amount of activity decreases, muscle mass decreases and eventually oxygen cannot be used effectively, resulting in a vicious cycle of deterioration of exercise capacity. Deconditioning due to decreased activity is a major cause of limb muscle dysfunction in patients with COPD. In these patients, the factors that decrease muscle strength and endurance include chronic inflammation, oxidative stress, inactivity, hypoxemia, hormone abnormality, deficits of nutrients such as protein and vitamin D, and the use of systemic corticosteroid. Therefore, treatment and management should either inhibit this process or should be directed toward supplementing the deficiency, such as with exercise, nutritional support, and medications and supplements. The relationship between sarcopenia and COPD is increasingly being reported, with some overlap in clinical features and treatments. We are fascinated to be able to diagnose 2 diseases through similar physical performance tests and to improve both diseases using the same treatment such as exercise. Therefore, this review summarizes the clinical relevance and integrative management of the 2 diseases.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Aldosterone Inhibits
In Vitro Myogenesis by Increasing Intracellular Oxidative Stress via Mineralocorticoid Receptor
Jin Young Lee, Da Ae Kim, Eunah Choi, Yun Sun Lee, So Jeong Park, Beom-Jun Kim
Endocrinol Metab. 2021;36(4):865-874. doi: 10.3803/EnM.2021.1108.
Reference
-
1. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010; 39:412–423.
Article2. Rosenberg IH. Sarcopenia: origins and clinical relevance. J Nutr. 1997; 127:990s–991s.
Article3. Roubenoff R. Sarcopenia: effects on body composition and function. J Gerontol A Biol Sci Med Sci. 2003; 58:1012–1017.
Article4. Fielding RA, Vellas B, Evans WJ, et al. International working group on sarcopenia. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. J Am Med Dir Assoc. 2011; 12:249–256.
Article5. Kwon HJ, Ha YC, Park HM. Prevalence of sarcopenia in the Korean woman based on the Korean National Health and Nutritional Examination Surveys. J Bone Metab. 2016; 23:23–26.
Article6. Kim KM, Jang HC, Lim S. Differences among skeletal muscle mass indices derived from height-, weight-, and body mass index-adjusted models in assessing sarcopenia. Korean J Intern Med. 2016; 31:643–650.
Article7. Yoo KH, Kim YS, Sheen SS, et al. Prevalence of chronic obstructive pulmonary disease in Korea: the fourth Korean National Health and Nutrition Examination Survey, 2008. Respirology. 2011; 16:659–665.
Article8. Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019; 48:16–31.
Article9. Studenski SA, Peters KW, Alley DE, et al. The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci. 2014; 69:547–558.
Article10. Visser M, Schaap LA. Consequences of sarcopenia. Clin Geriatr Med. 2011; 27:387–399.
Article11. Choi KM. Sarcopenia and sarcopenic obesity. Endocrinol Metab (Seoul). 2013; 28:86–89.
Article12. Roubenoff R, Parise H, Payette HA, et al. Cytokines, insulin-like growth factor 1, sarcopenia, and mortality in very old community-dwelling men and women: the Framingham Heart Study. Am J Med. 2003; 115:429–435.
Article13. Lim S, Kim JH, Yoon JW, et al. Sarcopenic obesity: prevalence and association with metabolic syndrome in the Korean Longitudinal Study on Health and Aging (KLoSHA). Diabetes Care. 2010; 33:1652–1654.
Article14. American Lung Association. Trends in COPD (Chronic Bronchitis and Emphysema): Morbidity and mortality. 2013. cited by 2016 Nov 23. Available from: https://www.lung.org/assets/documents/research/copd-trend-report.pdf.15. Spruit MA, Singh SJ, Garvey C, et al. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013; 188:e13–e64.16. Garvey C, Fullwood MD, Rigler J. Pulmonary rehabilitation exercise prescription in chronic obstructive lung disease: US survey and review of guidelines and clinical practices. J Cardiopulm Rehabil Prev. 2013; 33:314–322.
Article17. Celli BR, Cote CG, Marin JM, et al. The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med. 2004; 350:1005–1012.
Article18. Maltais F, Decramer M, Casaburi R, et al. An official American Thoracic Society/European Respiratory Society statement: update on limb muscle dysfunction in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2014; 189:e15–e62.
Article19. Vestbo J, Prescott E, Almdal T, et al. Body mass, fat-free body mass, and prognosis in patients with chronic obstructive pulmonary disease from a random population sample: findings from the Copenhagen City Heart Study. Am J Respir Crit Care Med. 2006; 173:79–83.
Article20. Coronell C, Orozco-Levi M, Gea J. COPD and body weight in a Mediterranean population. Clin Nutr. 2002; 21:437. author reply -8.21. Schols AM, Broekhuizen R, Weling-Scheepers CA, et al. Body composition and mortality in chronic obstructive pulmonary disease. Am J Clin Nutr. 2005; 82:53–59.
Article22. Bernard S, LeBlanc P, Whittom F, et al. Peripheral muscle weakness in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998; 158:629–634.
Article23. Gosker HR, van Mameren H, van Dijk PJ, et al. Skeletal muscle fibre-type shifting and metabolic profile in patients with chronic obstructive pulmonary disease. Eur Respir J. 2002; 19:617–625.
Article24. Richardson RS, Sheldon J, Poole DC, et al. Evidence of skeletal muscle metabolic reserve during whole body exercise in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1999; 159:881–885.
Article25. Koechlin C, Couillard A, Simar D, et al. Does oxidative stress alter quadriceps endurance in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2004; 169:1022–1027.
Article26. Koechlin C, Maltais F, Saey D, et al. Hypoxaemia enhances peripheral muscle oxidative stress in chronic obstructive pulmonary disease. Thorax. 2005; 60:834–841.
Article27. Couillard A, Maltais F, Saey D, et al. Exercise-induced quadriceps oxidative stress and peripheral muscle dysfunction in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2003; 167:1664–1669.
Article28. Allaire J, Maltais F, Doyon JF, et al. Peripheral muscle endurance and the oxidative profile of the quadriceps in patients with COPD. Thorax. 2004; 59:673–678.
Article29. Mador MJ, Bozkanat E, Kufel TJ. Quadriceps fatigue after cycle exercise in patients with COPD compared with healthy control subjects. Chest. 2003; 123:1104–1111.
Article30. Saey D, Côté CH, Mador MJ, et al. Assessment of muscle fatigue during exercise in chronic obstructive pulmonary disease. Muscle Nerve. 2006; 34:62–71.
Article31. Gan WQ, Man SF, Senthilselvan A, et al. Association between chronic obstructive pulmonary disease and systemic inflammation: a systematic review and a meta-analysis. Thorax. 2004; 59:574–580.
Article32. Byun MK, Cho EN, Chang J, et al. Sarcopenia correlates with systemic inflammation in COPD. Int J Chron Obstruct Pulmon Dis. 2017; 12:669–675.
Article33. Spruit MA, Gosselink R, Troosters T, et al. Muscle force during an acute exacerbation in hospitalised patients with COPD and its relationship with CXCL8 and IGF-I. Thorax. 2003; 58:752–756.
Article34. Marquis K, Debigaré R, Lacasse Y, et al. Midthigh muscle cross-sectional area is a better predictor of mortality than body mass index in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2002; 166:809–813.
Article35. Wagner PD. Possible mechanisms underlying the development of cachexia in COPD. Eur Respir J. 2008; 31:492–501.
Article36. Jones SE, Maddocks M, Kon SS, et al. Sarcopenia in COPD: prevalence, clinical correlates and response to pulmonary rehabilitation. Thorax. 2015; 70:213–218.
Article37. Jang HC. How to diagnose sarcopenia in Korean older adults. Ann Geriatr Med Res. 2018; 22:73–79.
Article38. Limpawattana P, Putraveephong S, Inthasuwan P, et al. Frailty syndrome in ambulatory patients with COPD. Int J Chron Obstruct Pulmon Dis. 2017; 12:1193–1198.
Article39. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global Strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report. GOLD executive summary. Am J Respir Crit Care Med. 2017; 195:557–582.
Article40. Suetta C, Magnusson SP, Beyer N, et al. Effect of strength training on muscle function in elderly hospitalized patients. Scand J Med Sci Sports. 2007; 17:464–472.
Article41. Levinger I, Goodman C, Hare DL, et al. The reliability of the 1RM strength test for untrained middle-aged individuals. J Sci Med Sport. 2009; 12:310–316.
Article42. Binder EF, Yarasheski KE, Steger-May K, et al. Effects of progressive resistance training on body composition in frail older adults: results of a randomized, controlled trial. J Gerontol A Biol Sci Med Sci. 2005; 60:1425–1431.
Article43. Orr R, Raymond J, Fiatarone Singh M. Efficacy of progressive resistance training on balance performance in older adults: a systematic review of randomized controlled trials. Sports Med. 2008; 38:317–343.
Article44. Low DC, Walsh GS, Arkesteijn M. Effectiveness of exercise interventions to improve postural control in older adults: a systematic review and meta-analyses of centre of pressure measurements. Sports Med. 2017; 47:101–112.
Article45. American College of Sports Medicine. ACSM's guidelines for exercise testing and prescription. 10th ed. Philadelphia, PA: Wolters Kluwer;2018.46. Kendrick KR, Baxi SC, Smith RM. Usefulness of the modified 0-10 Borg scale in assessing the degree of dyspnea in patients with COPD and asthma. J Emerg Nurs. 2000; 26:216–222.
Article47. Ries AL, Bauldoff GS, Carlin BW, et al. Pulmonary rehabilitation: Joint ACCP/AACVPR evidence-based clinical practice guidelines. Chest. 2007; 131:4s–42s.48. van Gestel AJ, Clarenbach CF, Stöwhas AC, et al. Prevalence and prediction of exercise-induced oxygen desaturation in patients with chronic obstructive pulmonary disease. Respiration. 2012; 84:353–359.
Article49. Johannson KA, Vittinghoff E, Lee K, et al. Acute exacerbation of idiopathic pulmonary fibrosis associated with air pollution exposure. Eur Respir J. 2014; 43:1124–1131.
Article50. Ministry of Health and Welfare, The Korean Nutrition Society. Dietary reference intakes for Koreans 2015. Sejong: Ministry of Health and Welfare, The Korean Nutrition Society;2015.51. Houston DK, Nicklas BJ, Ding J, et al. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: the Health, Aging, and Body Composition (Health ABC) Study. Am J Clin Nutr. 2008; 87:150–155.
Article52. Bauer JM, Verlaan S, Bautmans I, et al. Effects of a vitamin D and leucine-enriched whey protein nutritional supplement on measures of sarcopenia in older adults, the PROVIDE study: a randomized, double-blind, placebo-controlled trial. J Am Med Dir Assoc. 2015; 16:740–747.
Article53. Verreijen AM, Verlaan S, Engberink MF, et al. A high whey protein-, leucine-, and vitamin D-enriched supplement preserves muscle mass during intentional weight loss in obese older adults: a double-blind randomized controlled trial. Am J Clin Nutr. 2015; 101:279–286.
Article54. Tieland M, Dirks ML, van der Zwaluw N, et al. Protein supplementation increases muscle mass gain during prolonged resistance-type exercise training in frail elderly people: a randomized, double-blind, placebo-controlled trial. J Am Med Dir Assoc. 2012; 13:713–719.
Article55. Ferreira IM, Brooks D, White J, et al. Nutritional supplementation for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012; 12:Cd000998.
Article56. Bauer J, Biolo G, Cederholm T, et al. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE Study Group. J Am Med Dir Assoc. 2013; 14:542–559.
Article57. Boland R. Role of vitamin D in skeletal muscle function. Endocr Rev. 1986; 7:434–448.
Article58. Kuchuk NO, Pluijm SM, van Schoor NM, et al. Relationships of serum 25-hydroxyvitamin D to bone mineral density and serum parathyroid hormone and markers of bone turnover in older persons. J Clin Endocrinol Metab. 2009; 94:1244–1250.
Article59. Wilson GJ, Wilson JM, Manninen AH. Effects of beta-hydroxy-beta-methylbutyrate (HMB) on exercise performance and body composition across varying levels of age, sex, and training experience: A review. Nutr Metab (Lond). 2008; 5:1.
Article60. Wu H, Xia Y, Jiang J, et al. Effect of beta-hydroxy-beta-methylbutyrate supplementation on muscle loss in older adults: a systematic review and meta-analysis. Arch Gerontol Geriatr. 2015; 61:168–175.
Article61. Molfino A, Gioia G, Rossi Fanelli F, et al. Beta-hydroxy-beta-methylbutyrate supplementation in health and disease: a systematic review of randomized trials. Amino Acids. 2013; 45:1273–1292.
Article62. Dalton JT, Taylor RP, Mohler ML, et al. Selective androgen receptor modulators for the prevention and treatment of muscle wasting associated with cancer. Curr Opin Support Palliat Care. 2013; 7:345–351.
Article63. Kolosova NG, Vitovtov AO, Stefanova NA. Metformin reduces the signs of sarcopenia in old OXYS rats. Adv Gerontol. 2016; 6:70–74.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cor Pulmonale with Particular Reference to Chronic Obstructive Pulmonary Disease and Pulmonary Tuberculosis
- Association of sarcopenia with osteoporosis in patients with chronic obstructive pulmonary disease
- Review of Epidemiology, Diagnosis, and Treatment of Osteosarcopenia in Korea
- Management of Chronic Obstructive Pulmonary Disease
- Pathophysiology of Chronic Obstructive Pulmonary Disease