Clin Exp Vaccine Res.
2019 Jan;8(1):54-63. 10.7774/cevr.2019.8.1.54.
Nucleoprotein vaccine induces cross-protective cytotoxic T lymphocytes against both lineages of influenza B virus
- Affiliations
-
- 1Graduate School of Pharmaceutical Sciences, Ewha Womans University, Seoul, Korea. TCELL@EWHA.AC.KR
- KMID: 2438964
- DOI: http://doi.org/10.7774/cevr.2019.8.1.54
Abstract
- PURPOSE
The influenza B virus diverges into two antigenically distinct lineages: B/Yamagata and B/Victoria. Influenza B is the dominant circulating virus during some influenza seasons, and recent data demonstrated that influenza A and B infection similarly cause severe clinical symptoms in hospitalized patients. Nucleoprotein (NP) is a good target for a universal influenza vaccine. This study investigated whether NP epitope variation within two lineages affects the dominant cytotoxic T lymphocyte (CTL) responses induced by vaccination and the resultant protective immunity.
MATERIALS AND METHODS
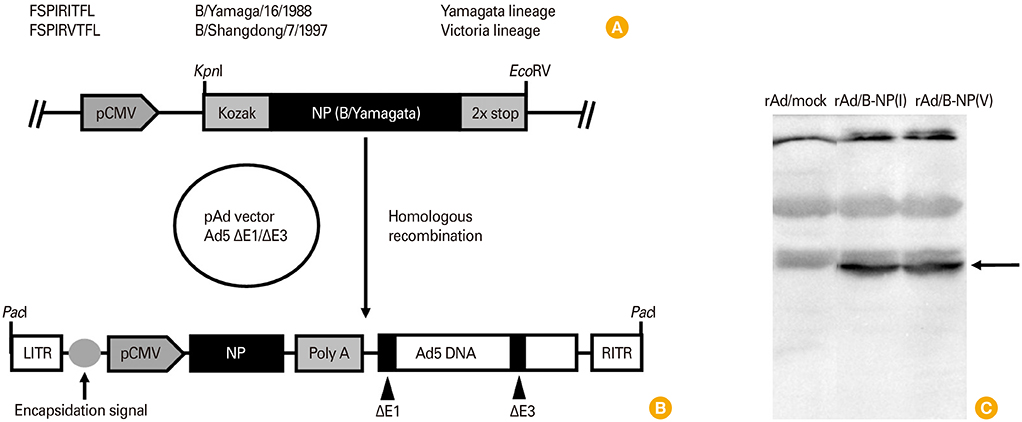
The NP of B/Yamagata/16/1988, the representative strain of the Yamagata lineage, includes a dominant CTL epitope, FSPIRITFL, while B/Shangdong/7/1997 from the Victoria lineage has one amino acid difference in this sequence, FSPIRVTFL. Two recombinant replication-deficient adenovirus (rAd)-vectored vaccines expressing either NP were prepared (rAd/B-NP(I) and rAd/B-NP(V), respectively) and administered to BALB/c mice intranasally. To examine the efficacy of vaccination, antibody responses, CTL responses, and morbidity/mortality after challenge were measured.
RESULTS
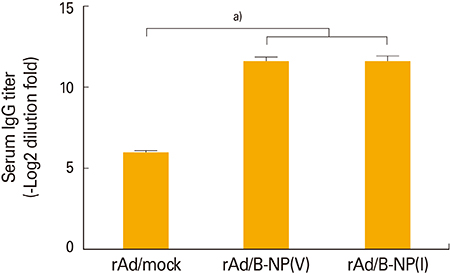
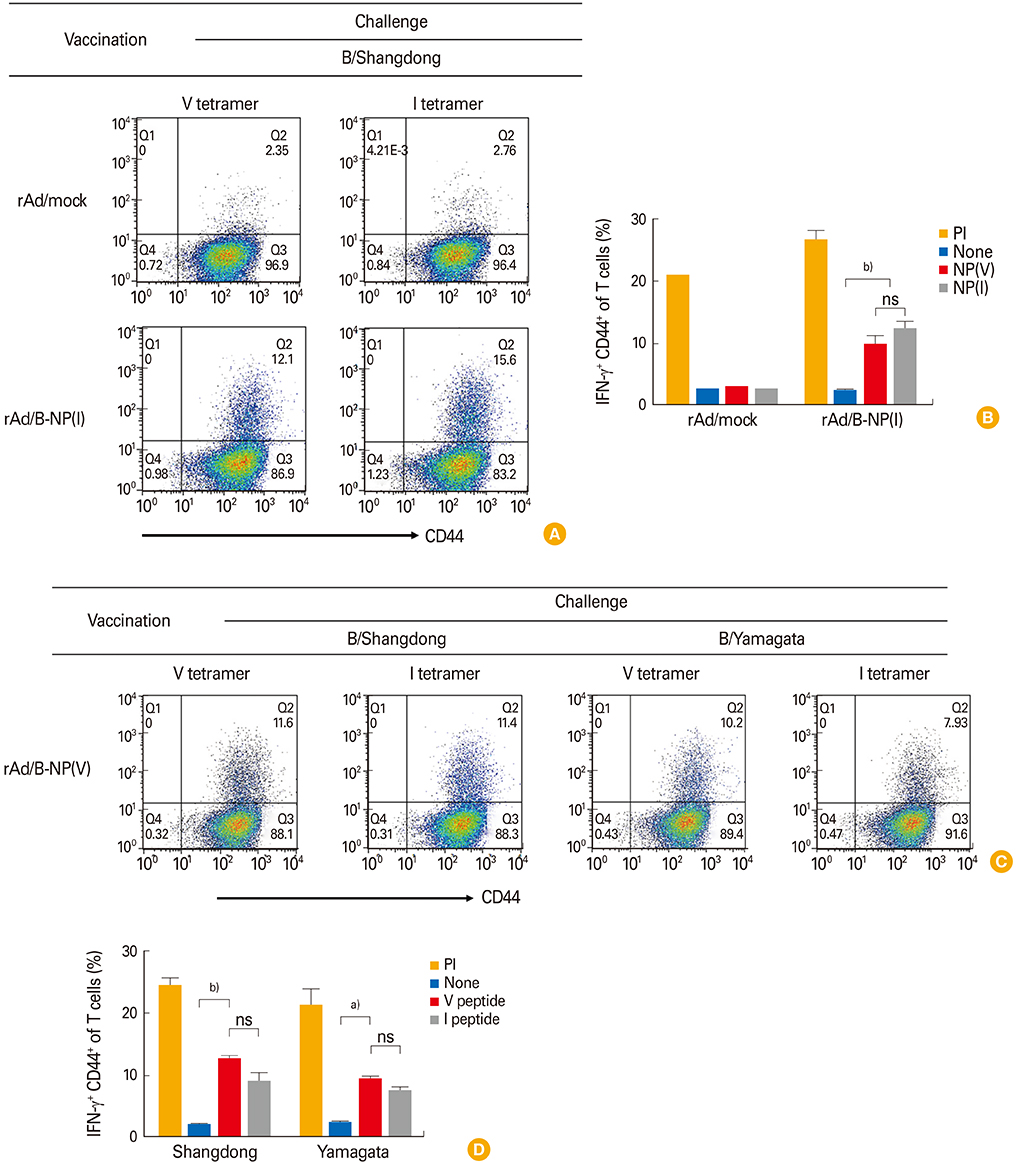
Both vaccines induce similar antibody and CD8 T-cell responses cross-reacting to both epitopes, and also confer cross-protection against both lineages regardless of amino acid difference.
CONCLUSION
The rAd-vectored vaccine expressing the NP could be developed as universal influenza B vaccine which provides broader protection.
Keyword
MeSH Terms
Figure
Reference
-
1. Fiore AE, Uyeki TM, Broder K, et al. Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Recomm Rep. 2010; 59:1–62.2. McCullers JA, Saito T, Iverson AR. Multiple genotypes of influenza B virus circulated between 1979 and 2003. J Virol. 2004; 78:12817–12828.
Article3. Suzuki Y. Sialobiology of influenza: molecular mechanism of host range variation of influenza viruses. Biol Pharm Bull. 2005; 28:399–408.
Article4. Furuse Y, Suzuki A, Kishi M, et al. Occurrence of mixed populations of influenza A viruses that can be maintained through transmission in a single host and potential for reassortment. J Clin Microbiol. 2010; 48:369–374.
Article5. Parrish CR, Kawaoka Y. The origins of new pandemic viruses: the acquisition of new host ranges by canine parvovirus and influenza A viruses. Annu Rev Microbiol. 2005; 59:553–586.
Article6. Rota PA, Wallis TR, Harmon MW, Rota JS, Kendal AP, Nerome K. Cocirculation of two distinct evolutionary lineages of influenza type B virus since 1983. Virology. 1990; 175:59–68.
Article7. Hay AJ, Gregory V, Douglas AR, Lin YP. The evolution of human influenza viruses. Philos Trans R Soc Lond B Biol Sci. 2001; 356:1861–1870.
Article8. Huang SS, Banner D, Paquette SG, Leon AJ, Kelvin AA, Kelvin DJ. Pathogenic influenza B virus in the ferret model establishes lower respiratory tract infection. J Gen Virol. 2014; 95:2127–2139.
Article9. Nobusawa E, Sato K. Comparison of the mutation rates of human influenza A and B viruses. J Virol. 2006; 80:3675–3678.
Article10. Paul Glezen W, Schmier JK, Kuehn CM, Ryan KJ, Oxford J. The burden of influenza B: a structured literature review. Am J Public Health. 2013; 103:e43–e51.
Article11. Alirol E, Getaz L, Stoll B, Chappuis F, Loutan L. Urbanisation and infectious diseases in a globalised world. Lancet Infect Dis. 2011; 11:131–141.
Article12. Belshe RB, Coelingh K, Ambrose CS, Woo JC, Wu X. Efficacy of live attenuated influenza vaccine in children against influenza B viruses by lineage and antigenic similarity. Vaccine. 2010; 28:2149–2156.
Article13. Influenza report 2006 [Internet]. Paris: Flying Publisher;2006. cited 2018 Dec 5. Available from: http://www.influenzareport.com.14. Assarsson E, Bui HH, Sidney J, et al. Immunomic analysis of the repertoire of T-cell specificities for influenza A virus in humans. J Virol. 2008; 82:12241–12251.
Article15. Hillaire ML, van Trierum SE, Kreijtz JH, et al. Cross-protective immunity against influenza pH1N1 2009 viruses induced by seasonal influenza A (H3N2) virus is mediated by virus-specific T-cells. J Gen Virol. 2011; 92(Pt 10):2339–2349.
Article16. Jameson J, Cruz J, Ennis FA. Human cytotoxic T-lymphocyte repertoire to influenza A viruses. J Virol. 1998; 72:8682–8689.
Article17. Pogue RR, Eron J, Frelinger JA, Matsui M. Amino-terminal alteration of the HLA-A*0201-restricted human immunodeficiency virus pol peptide increases complex stability and in vitro immunogenicity. Proc Natl Acad Sci U S A. 1995; 92:8166–8170.
Article18. Tian S, Maile R, Collins EJ, Frelinger JA. CD8+ T cell activation is governed by TCR-peptide/MHC affinity, not dissociation rate. J Immunol. 2007; 179:2952–2960.
Article19. Flynn KJ, Belz GT, Altman JD, Ahmed R, Woodland DL, Doherty PC. Virus-specific CD8+ T cells in primary and secondary influenza pneumonia. Immunity. 1998; 8:683–691.
Article20. Robbins PA, Rota PA, Shapiro SZ. A broad cytotoxic T lymphocyte response to influenza type B virus presented by multiple HLA molecules. Int Immunol. 1997; 9:815–823.
Article21. Kim MH, Kang JO, Kim JY, Jung HE, Lee HK, Chang J. Single mucosal vaccination targeting nucleoprotein provides broad protection against two lineages of influenza B virus. Antiviral Res. 2019; 163:19–28.
Article22. Cheng C, Gall JG, Kong WP, et al. Mechanism of ad5 vaccine immunity and toxicity: fiber shaft targeting of dendritic cells. PLoS Pathog. 2007; 3:e25.
Article23. Price GE, Soboleski MR, Lo CY, et al. Single-dose mucosal immunization with a candidate universal influenza vaccine provides rapid protection from virulent H5N1, H3N2 and H1N1 viruses. PLoS One. 2010; 5:e13162.
Article24. Kim SH, Kim JY, Choi Y, Nguyen HH, Song MK, Chang J. Mucosal vaccination with recombinant adenovirus encoding nucleoprotein provides potent protection against influenza virus infection. PLoS One. 2013; 8:e75460.
Article25. He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B. A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci U S A. 1998; 95:2509–2514.
Article26. Ulmer JB, Fu TM, Deck RR, et al. Protective CD4+ and CD8+ T cells against influenza virus induced by vaccination with nucleoprotein DNA. J Virol. 1998; 72:5648–5653.
Article27. Zheng M, Liu F, Shen Y, et al. Cross-protection against influenza virus infection by intranasal administration of nucleoprotein-based vaccine with compound 48/80 adjuvant. Hum Vaccin Immunother. 2015; 11:397–406.
Article28. Barefoot BE, Sample CJ, Ramsburg EA. Recombinant vesicular stomatitis virus expressing influenza nucleoprotein induces CD8 T-cell responses that enhance antibody-mediated protection after lethal challenge with influenza virus. Clin Vaccine Immunol. 2009; 16:488–498.
Article29. Roose K, Fiers W, Saelens X. Pandemic preparedness: toward a universal influenza vaccine. Drug News Perspect. 2009; 22:80–92.
Article30. van de Sandt CE, Dou Y, Vogelzang-van Trierum SE, et al. Influenza B virus-specific CD8+ T-lymphocytes strongly cross-react with viruses of the opposing influenza B lineage. J Gen Virol. 2015; 96:2061–2073.
Article31. Kreijtz JH, Bodewes R, van Amerongen G, et al. Primary influenza A virus infection induces cross-protective immunity against a lethal infection with a heterosubtypic virus strain in mice. Vaccine. 2007; 25:612–620.
Article32. Yap KL, Ada GL. The recovery of mice from influenza A virus infection: adoptive transfer of immunity with influenza virus-specific cytotoxic T lymphocytes recognizing a common virion antigen. Scand J Immunol. 1978; 8:413–420.
Article33. Lukacher AE, Braciale VL, Braciale TJ. In vivo effector function of influenza virus-specific cytotoxic T lymphocyte clones is highly specific. J Exp Med. 1984; 160:814–826.
Article34. Guo H, Santiago F, Lambert K, Takimoto T, Topham DJ. T cell-mediated protection against lethal 2009 pandemic H1N1 influenza virus infection in a mouse model. J Virol. 2011; 85:448–455.
Article35. Tatsis N, Ertl HC. Adenoviruses as vaccine vectors. Mol Ther. 2004; 10:616–629.
Article36. Holmberg K, Mariathasan S, Ohteki T, Ohashi PS, Gascoigne NR. TCR binding kinetics measured with MHC class I tetramers reveal a positive selecting peptide with relatively high affinity for TCR. J Immunol. 2003; 171:2427–2434.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A “Prime and Deploy” Strategy for Universal Influenza Vaccine Targeting Nucleoprotein Induces Lung-Resident Memory CD8 T cells
- Vaccine Strategy That Enhances the Protective Efficacy of Systemic Immunization by Establishing LungResident Memory CD8 T Cells Against Influenza Infection
- Mechanisms of Cross-protection by Influenza Virus M2-based Vaccines
- Enhancement of DNA Vaccine-induced Immune Responses by Influenza Virus NP Gene
- Influenza virus vaccine for neglected hosts: horses and dogs