Anesth Pain Med.
2018 Oct;13(4):349-362. 10.17085/apm.2018.13.4.349.
Derivation of pharmacokinetic equations
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. nohgj@amc.seoul.kr
- 2Department of Clinical Pharmacology and Therapeutics, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 2435992
- DOI: http://doi.org/10.17085/apm.2018.13.4.349
Abstract
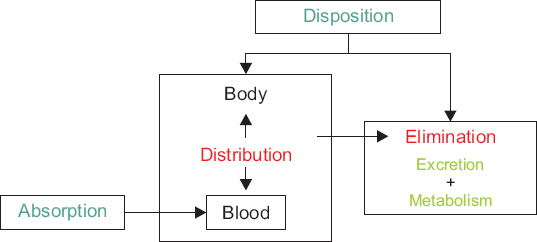
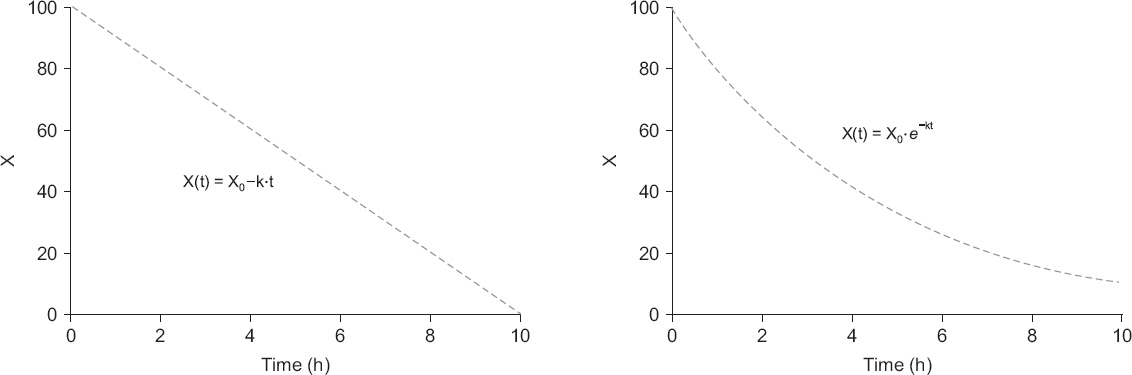
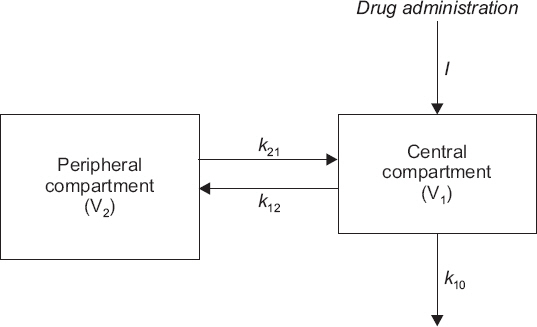
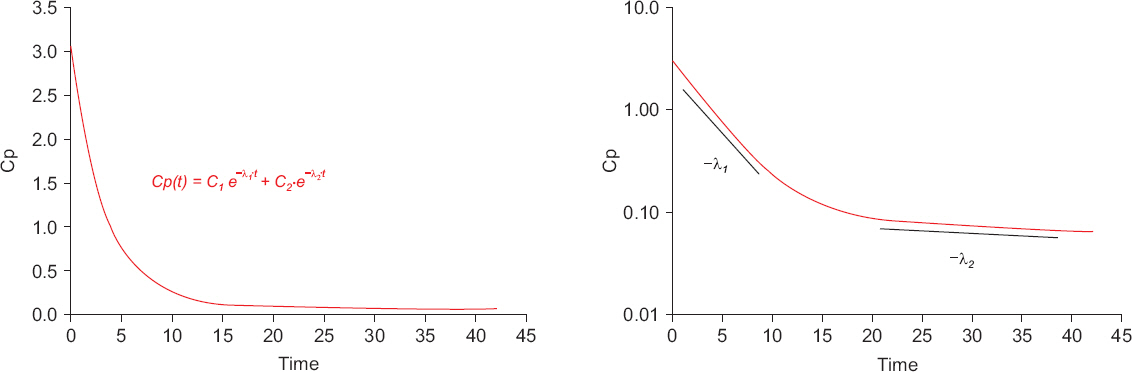
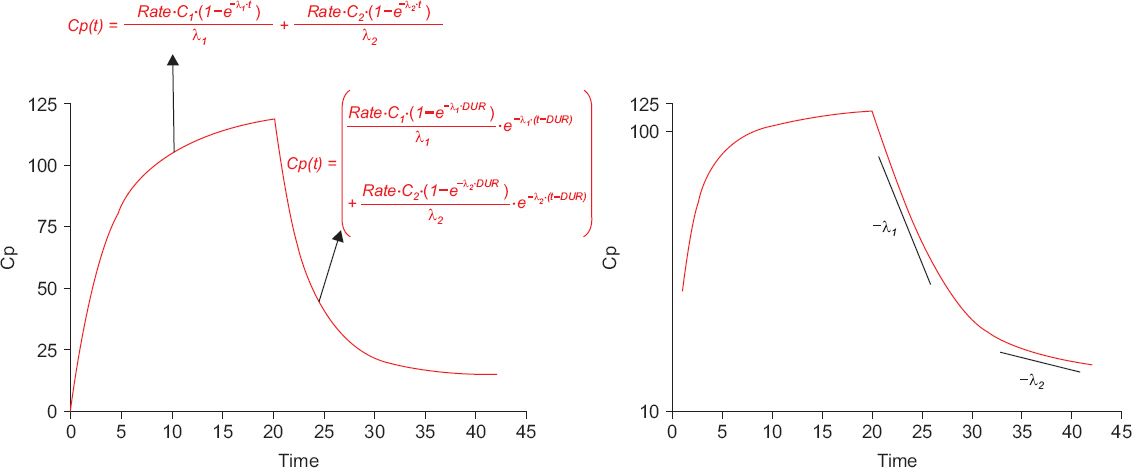
- A variety of drugs are continuously or intermittently administered to patients during general or regional anesthesia. Pharmacotherapy should also receive priority compared with several treatment modalities including nerve blocks for chronic pain control. Therefore, pharmacology may be fundamental to anesthesia as well as pain medicine. Pharmacokinetic equations quantitatively evaluating drug transfer in the body are essential to understanding pharmacological principles. In mammillary compartmental models, pharmacokinetic equations are easily derived from a few simple principles. The kinetics of drug transfer between compartments is determined initially. Ordinary, linear differential equations are constructed based on the kinetics. The Laplace transforms of these differential equations are used to derive functions for the calculation of drug amounts in the central or effect compartments in the Laplace domain. The inverse Laplace transforms of these functions are used to obtain pharmacokinetic equations in time domain. In this review, a two-compartment mammillary pharmacokinetic model is used to derive pharmacokinetic equations using the aforementioned principles.
Keyword
MeSH Terms
Figure
Reference
-
REFERENCE
1. Benet LZ, Turi JS. Use of general partial fraction theorem for obtaining inverse laplace transforms in pharmacokinetic analysis. J Pharm Sci. 1971; 60:1593–4. DOI: 10.1002/jps.2600601041.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Meta-Analysis on the Performance of Cystatin C- versus Creatinine-based eGFR Equations in Predicting Vancomycin Clearance
- PKconverter: R package to convert the pharmacokinetic parameters
- Development of Theory of Uncertainty on COVID-19: Theory Derivation Based on Uncertainty in Illness Theory
- Estimation of Maximum Tibia Length from Fragmentary Condition in Korean Populations
- What kind of medical equations and decision trees do physicians want in their daily activities? : Analysis of one-year MedCalc 3000(R) log data