J Breast Cancer.
2018 Dec;21(4):382-390. 10.4048/jbc.2018.21.e48.
PIK3CA Mutations and Neoadjuvant Therapy Outcome in Patients with Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer: A Sequential Analysis
- Affiliations
-
- 1Department of Pathology and Translational Genomics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. eunyoon.cho@samsung.com
- 2Division of Hematology-Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Department of Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 2429816
- DOI: http://doi.org/10.4048/jbc.2018.21.e48
Abstract
- PURPOSE
PIK3CA mutation is considered to be a possible cause for resistance to neoadjuvant chemotherapy (NAC) in human epidermal growth factor receptor 2 (HER2)-positive breast cancer. We investigated the association between PIK3CA mutations and the outcome of NAC in HER2-positive breast cancers.
METHODS
A total of 100 HER2-positive breast cancer patients who had undergone NAC and surgery between 2004 and 2016 were examined. Mutation status was sequentially assessed in pre-NAC, post-NAC, and recurrent specimens taken from these patients.
RESULTS
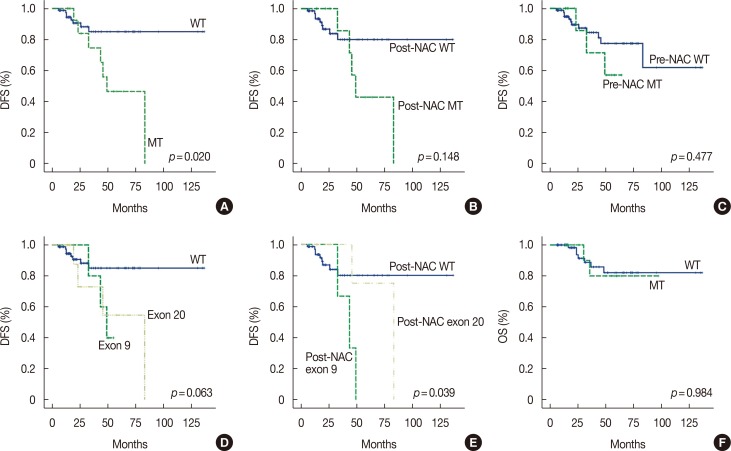
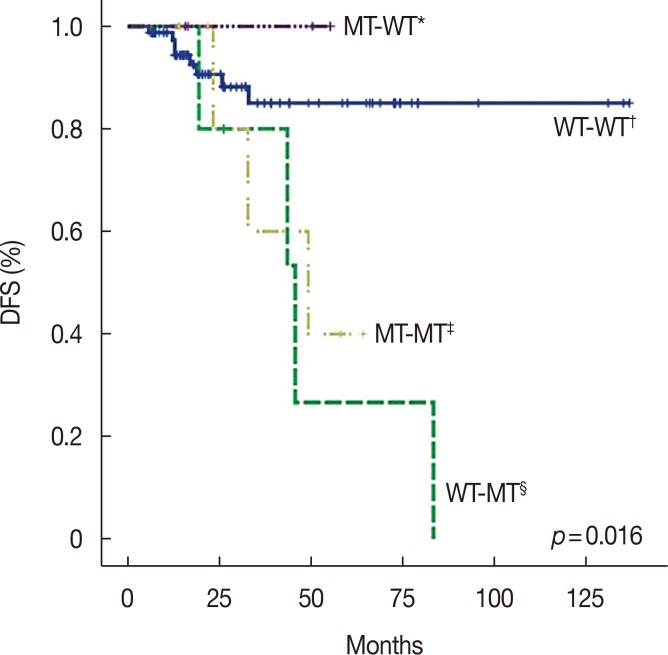
PIK3CA mutations were identified in the sequential specimens of 17 patients (17.0%). These 17 patients experienced shorter disease-free survival (DFS) than the rest of the patients (58.3 months vs. 119.3 months, p=0.020); however, there was no significant difference in pathologic complete response (pCR) and overall survival (OS) (pCR, 17.6% vs. 33.7%, p=0.191; OS, 84.5 months vs. 118.0 months, p=0.984). While there was no difference in pCR between the wild-type and mutant PIK3CA groups in pre-NAC specimens (25.0% vs. 31.8%, p=0.199), PIK3CA mutations correlated with lower pCR in post-NAC specimens (0.0% vs. 24.3%, p < 0.001). Multivariate analysis revealed significantly worse DFS in the mutant PIK3CA group than in the wild-type group (hazard ratio, 3.540; 95% confidence interval, 1.001-12.589; p=0.050). Moreover, the DFS curves of the change of PIK3CA mutation status in sequential specimens were significantly different (p=0.016).
CONCLUSION
PIK3CA mutation in HER2-positive breast cancer was correlated with a lower pCR rate and shorter DFS. These results suggest that PIK3CA mutation is a prognostic marker for NAC in HER2-positive breast cancer, especially in post-NAC specimens.
Keyword
MeSH Terms
-
Breast
Breast Neoplasms
Disease-Free Survival
Drug Therapy
Epidermal Growth Factor*
Humans
Humans*
Multivariate Analysis
Neoadjuvant Therapy*
Phosphatidylinositol 3-Kinases
Polymerase Chain Reaction
Receptor, Epidermal Growth Factor*
Receptor, ErbB-2
Epidermal Growth Factor
Phosphatidylinositol 3-Kinases
Receptor, Epidermal Growth Factor
Receptor, ErbB-2
Figure
Reference
-
1. Hennessy BT, Smith DL, Ram PT, Lu Y, Mills GB. Exploiting the PI3K/AKT pathway for cancer drug discovery. Nat Rev Drug Discov. 2005; 4:988–1004. PMID: 16341064.
Article2. Yang SX, Polley E, Lipkowitz S. New insights on PI3K/AKT pathway alterations and clinical outcomes in breast cancer. Cancer Treat Rev. 2016; 45:87–96. PMID: 26995633.
Article3. Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012; 490:61–70. PMID: 23000897.4. Stemke-Hale K, Gonzalez-Angulo AM, Lluch A, Neve RM, Kuo WL, Davies M, et al. An integrative genomic and proteomic analysis of PIK3CA, PTEN, and AKT mutations in breast cancer. Cancer Res. 2008; 68:6084–6091. PMID: 18676830.
Article5. Toi M, Iwata H, Fujiwara Y, Ito Y, Nakamura S, Tokuda Y, et al. Lapatinib monotherapy in patients with relapsed, advanced, or metastatic breast cancer: efficacy, safety, and biomarker results from Japanese patients phase II studies. Br J Cancer. 2009; 101:1676–1682. PMID: 19844234.
Article6. Xu BH, Jiang ZF, Chua D, Shao ZM, Luo RC, Wang XJ, et al. Lapatinib plus capecitabine in treating HER2-positive advanced breast cancer: efficacy, safety, and biomarker results from Chinese patients. Chin J Cancer. 2011; 30:327–335. PMID: 21527065.
Article7. Loibl S, Majewski I, Guarneri V, Nekljudova V, Holmes E, Bria E, et al. PIK3CA mutations are associated with reduced pathological complete response rates in primary HER2-positive breast cancer: pooled analysis of 967 patients from five prospective trials investigating lapatinib and trastuzumab. Ann Oncol. 2016; 27:1519–1525. PMID: 27177864.
Article8. Ibrahim EM, Kazkaz GA, Al-Mansour MM, Al-Foheidi ME. The predictive and prognostic role of phosphatase phosphoinositol-3 (PI3) kinase (PIK3CA) mutation in HER2-positive breast cancer receiving HER2-targeted therapy: a meta-analysis. Breast Cancer Res Treat. 2015; 152:463–476. PMID: 26105797.
Article9. Berns K, Horlings HM, Hennessy BT, Madiredjo M, Hijmans EM, Beelen K, et al. A functional genetic approach identifies the PI3K pathway as a major determinant of trastuzumab resistance in breast cancer. Cancer Cell. 2007; 12:395–402. PMID: 17936563.
Article10. Mills GB, Kohn E, Lu Y, Eder A, Fang X, Wang H, et al. Linking molecular diagnostics to molecular therapeutics: targeting the PI3K pathway in breast cancer. Semin Oncol. 2003; 30(5 Suppl 16):93–104. PMID: 14613030.
Article11. Yuan H, Chen J, Liu Y, Ouyang T, Li J, Wang T, et al. Association of PIK3CA mutation status before and after neoadjuvant chemotherapy with response to chemotherapy in women with breast cancer. Clin Cancer Res. 2015; 21:4365–4372. PMID: 25979484.
Article12. Engels CC, Kiderlen M, Bastiaannet E, van Eijk R, Mooyaart A, Smit VT, et al. The clinical value of HER-2 overexpression and PIK3CA mutations in the older breast cancer population: a FOCUS study analysis. Breast Cancer Res Treat. 2016; 156:361–370. PMID: 26968397.
Article13. Toomey S, Eustace AJ, Fay J, Sheehan KM, Carr A, Milewska M, et al. Impact of somatic PI3K pathway and ERBB family mutations on pathological complete response (pCR) in HER2-positive breast cancer patients who received neoadjuvant HER2-targeted therapies. Breast Cancer Res. 2017; 19:87. PMID: 28750640.
Article14. Pogue-Geile KL, Song N, Jeong JH, Gavin PG, Kim SR, Blackmon NL, et al. Intrinsic subtypes, PIK3CA mutation, and the degree of benefit from adjuvant trastuzumab in the NSABP B-31 trial. J Clin Oncol. 2015; 33:1340–1347. PMID: 25559813.
Article15. Loibl S, de la Pena L, Nekljudova V, Zardavas D, Michiels S, Denkert C, et al. Neoadjuvant buparlisib plus trastuzumab and paclitaxel for women with HER2+ primary breast cancer: a randomised, double-blind, placebo-controlled phase II trial (NeoPHOEBE). Eur J Cancer. 2017; 85:133–145. PMID: 28923573.
Article16. Cizkova M, Dujaric ME, Lehmann-Che J, Scott V, Tembo O, Asselain B, et al. Outcome impact of PIK3CA mutations in HER2-positive breast cancer patients treated with trastuzumab. Br J Cancer. 2013; 108:1807–1809. PMID: 23612454.
Article17. Wang L, Zhang Q, Zhang J, Sun S, Guo H, Jia Z, et al. PI3K pathway activation results in low efficacy of both trastuzumab and lapatinib. BMC Cancer. 2011; 11:248. PMID: 21676217.
Article18. Kalinsky K, Jacks LM, Heguy A, Patil S, Drobnjak M, Bhanot UK, et al. PIK3CA mutation associates with improved outcome in breast cancer. Clin Cancer Res. 2009; 15:5049–5059. PMID: 19671852.
Article19. Majewski IJ, Nuciforo P, Mittempergher L, Bosma AJ, Eidtmann H, Holmes E, et al. PIK3CA mutations are associated with decreased benefit to neoadjuvant human epidermal growth factor receptor 2-targeted therapies in breast cancer. J Clin Oncol. 2015; 33:1334–1339. PMID: 25559818.
Article20. Goel S, Krop IE. PIK3CA mutations in HER2-positive breast cancer: an ongoing conundrum. Ann Oncol. 2016; 27:1368–1372. PMID: 27358378.
Article21. Wang Y, Liu Y, Du Y, Yin W, Lu J. The predictive role of phosphatase and tensin homolog (PTEN) loss, phosphoinositol-3 (PI3) kinase (PIK3CA) mutation, and PI3K pathway activation in sensitivity to trastuzumab in HER2-positive breast cancer: a meta-analysis. Curr Med Res Opin. 2013; 29:633–642. PMID: 23574264.
Article22. Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010; 28:2784–2795. PMID: 20404251.
Article23. Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol. 2013; 31:3997–4013. PMID: 24101045.
Article24. Bachman KE, Argani P, Samuels Y, Silliman N, Ptak J, Szabo S, et al. The PIK3CA gene is mutated with high frequency in human breast cancers. Cancer Biol Ther. 2004; 3:772–775. PMID: 15254419.
Article25. Rexer BN, Chanthaphaychith S, Dahlman K, Arteaga CL. Direct inhibition of PI3K in combination with dual HER2 inhibitors is required for optimal antitumor activity in HER2+ breast cancer cells. Breast Cancer Res. 2014; 16:R9. PMID: 24451154.
Article26. Tolaney S, Burris H, Gartner E, Mayer IA, Saura C, Maurer M, et al. Phase I/II study of pilaralisib (SAR245408) in combination with trastuzumab or trastuzumab plus paclitaxel in trastuzumab-refractory HER2-positive metastatic breast cancer. Breast Cancer Res Treat. 2015; 149:151–161. PMID: 25537644.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Concomitant PIK3CA and TP53 Mutations in Breast Cancer: An Analysis of Clinicopathologic and Mutational Features, Neoadjuvant Therapeutic Response, and Prognosis
- Human Epidermal Growth Factor Receptor 2-positive Mucinous Carcinoma with Signet Ring Cell Differentiation, Which Showed Complete Response after Neoadjuvant Chemotherapy
- Prevalence and Prognostic Role of PIK3CA/AKT1 Mutations in Chinese Breast Cancer Patients
- Recent Advances in the Neoadjuvant Treatment of Breast Cancer
- PIK3CA Mutation is Associated with Poor Response to HER2-Targeted Therapy in Breast Cancer Patients