World J Mens Health.
2019 Jan;37(1):45-54. 10.5534/wjmh.180036.
Testosterone Is a Contraceptive and Should Not Be Used in Men Who Desire Fertility
- Affiliations
-
- 1Department of Urology, University of Miami Miller School of Medicine, Miami, FL, USA. asp136@med.miami.edu
- 2Department of Urology, Thomas Jefferson University, Philadelphia, PA, USA.
- KMID: 2429721
- DOI: http://doi.org/10.5534/wjmh.180036
Abstract
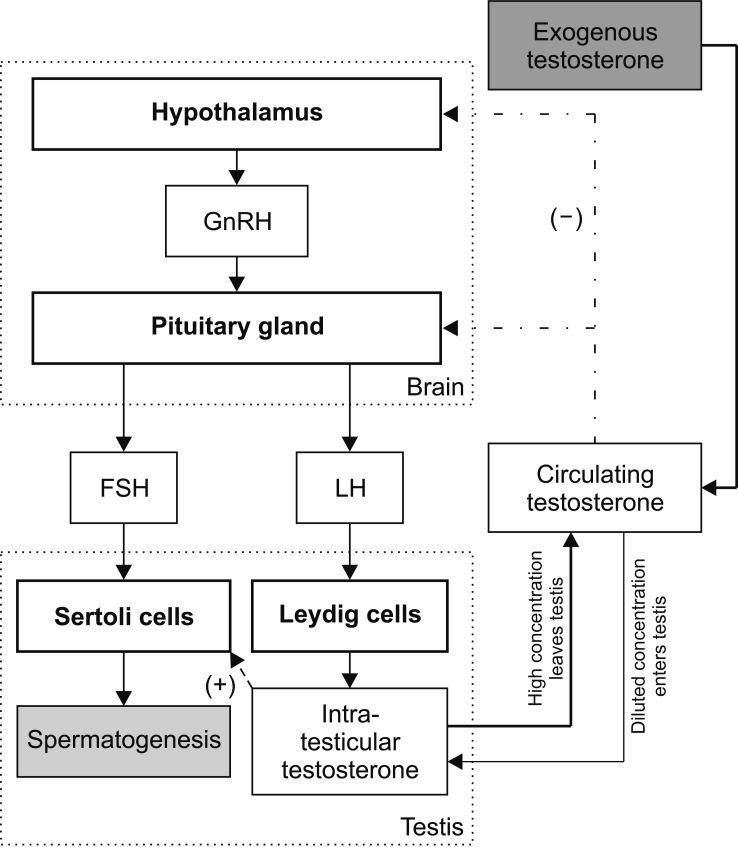
- Testosterone has a variety of functions and is commonly used in older men to treat symptoms of hypogonadism, such as decreased libido, decreased mood and erectile dysfunction. Despite its positive effects on sexual function, it has a negative effect on fertility. Exogenous testosterone therapy can negatively affect the hypothalamic-pituitary gonadal axis and inhibit the production of follicle stimulating hormone and luteinizing hormone. The purpose of this review is to discuss the contraceptive properties of testosterone therapy and to discuss strategies to increase testosterone in men with the desire to preserve fertility.
Keyword
MeSH Terms
Figure
Reference
-
1. Baillargeon J, Urban RJ, Ottenbacher KJ, Pierson KS, Goodwin JS. Trends in androgen prescribing in the United States, 2001 to 2011. JAMA Intern Med. 2013; 173:1465–1466. PMID: 23939517.
Article2. Westaby D, Ogle SJ, Paradinas FJ, Randell JB, Murray-Lyon IM. Liver damage from long-term methyltestosterone. Lancet. 1977; 2:262–263. PMID: 69876.
Article3. Mulhall JP, Trost LW, Brannigan RE, Kurtz EG, Redmon JB, Chiles KA, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018; 200:423–432. PMID: 29601923.
Article4. Bhasin S, Brito JP, Cunningham GR, Hayes FJ, Hodis HN, Matsumoto AM, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018; 103:1715–1744. PMID: 29562364.
Article6. Walker WH, Cheng J. FSH and testosterone signaling in Sertoli cells. Reproduction. 2005; 130:15–28. PMID: 15985628.
Article7. Walker WH. Non-classical actions of testosterone and spermatogenesis. Philos Trans R Soc Lond B Biol Sci. 2010; 365:1557–1569. PMID: 20403869.
Article8. Smith LB, Walker WH. The regulation of spermatogenesis by androgens. Semin Cell Dev Biol. 2014; 30:2–13. PMID: 24598768.
Article9. Shaha C, Tripathi R, Mishra DP. Male germ cell apoptosis: regulation and biology. Philos Trans R Soc Lond B Biol Sci. 2010; 365:1501–1515. PMID: 20403866.
Article10. O'Donnell L, Nicholls PK, O'Bryan MK, McLachlan RI, Stanton PG. Spermiation: the process of sperm release. Spermatogenesis. 2011; 1:14–35. PMID: 21866274.11. Wenker EP, Dupree JM, Langille GM, Kovac J, Ramasamy R, Lamb D, et al. The use of HCG-based combination therapy for recovery of spermatogenesis after testosterone use. J Sex Med. 2015; 12:1334–1337. PMID: 25904023.
Article12. Cornud F, Amar E, Hamida K, Thiounn N, Hélénon O, Moreau JF. Imaging in male hypofertility and impotence. BJU Int. 2000; 86(Suppl 1):153–163. PMID: 10961285.
Article13. Amory JK, Bremner W. Endocrine regulation of testicular function in men: implications for contraceptive development. Mol Cell Endocrinol. 2001; 182:175–179. PMID: 11514052.
Article14. Contraceptive efficacy of testosterone-induced azoospermia in normal men. World Health Organization Task Force on methods for the regulation of male fertility. Lancet. 1990; 336:955–959. PMID: 1977002.15. World Health Organization Task Force on Methods for the Regulation of Male Fertility. Contraceptive efficacy of testosterone-induced azoospermia and oligozoospermia in normal men. Fertil Steril. 1996; 65:821–829. PMID: 8654646.16. Kohn TP, Louis MR, Pickett SM, Lindgren MC, Kohn JR, Pastuszak AW, et al. Age and duration of testosterone therapy predict time to return of sperm count after human chorionic gonadotropin therapy. Fertil Steril. 2017; 107:351–357. PMID: 27855957.
Article17. Eftekhar M, Mohammadian F, Yousefnejad F, Molaei B, Aflatoonian A. Comparison of conventional IVF versus ICSI in non-male factor, normoresponder patients. Iran J Reprod Med. 2012; 10:131–136. PMID: 25242986.
Article18. Komsky-Elbaz A, Raziel A, Friedler S, Strassburger D, Kasterstein E, Komarovsky D, et al. Conventional IVF versus ICSI in sibling oocytes from couples with endometriosis and normozoospermic semen. J Assist Reprod Genet. 2013; 30:251–257. PMID: 23271211.
Article19. McBride JA, Coward RM. Recovery of spermatogenesis following testosterone replacement therapy or anabolic-androgenic steroid use. Asian J Androl. 2016; 18:373–380. PMID: 26908067.
Article20. Kost K, Singh S, Vaughan B, Trussell J, Bankole A. Estimates of contraceptive failure from the 2002 National Survey of Family Growth. Contraception. 2008; 77:10–21. PMID: 18082661.
Article21. Trussell J. Contraceptive failure in the United States. Contraception. 2011; 83:397–404. PMID: 21477680.
Article22. Heinemann K, Saad F, Wiesemes M, White S, Heinemann L. Attitudes toward male fertility control: results of a multinational survey on four continents. Hum Reprod. 2005; 20:549–556. PMID: 15608042.
Article23. Wang C, Swerdloff RS. Hormonal approaches to male contraception. Curr Opin Urol. 2010; 20:520–524. PMID: 20808223.
Article24. Nieschlag E, Hoogen H, Bölk M, Schuster H, Wickings EJ. Clinical trial with testosterone undecanoate for male fertility control. Contraception. 1978; 18:607–614. PMID: 219988.
Article25. Gu Y, Liang X, Wu W, Liu M, Song S, Cheng L, et al. Multicenter contraceptive efficacy trial of injectable testosterone undecanoate in Chinese men. J Clin Endocrinol Metab. 2009; 94:1910–1915. PMID: 19293262.
Article26. Liu PY, Swerdloff RS, Anawalt BD, Anderson RA, Bremner WJ, Elliesen J, et al. Determinants of the rate and extent of spermatogenic suppression during hormonal male contraception: an integrated analysis. J Clin Endocrinol Metab. 2008; 93:1774–1783. PMID: 18303073.
Article27. Kogan P, Wald M. Male contraception: history and development. Urol Clin North Am. 2014; 41:145–161. PMID: 24286773.28. Larkin A. Male hormonal contraceptive trial ending early [Internet]. Arlington (VT): CONRAD;c2011. cited 2018 Jan 11. Available from: http://www.conrad.org/news-pressreleases-63.html.29. Thirumalai ACJ, Amory JK, Swerdloff RS, Surampudi V, Liu PY, Bremner WJ, et al. Pharmacokinetic and pharmacodynamic effects of 28 days of oral dimethandrolone undecanoate in healthy men: A prototype male pill. J Investig Med. 2018; 66:A165–A166.30. Srinivas-Shankar U, Sharma D. Testosterone treatment in elderly men. Adv Ther. 2009; 26:25–39. PMID: 19198768.
Article31. Holyoak JD, Crawford ED, Meacham RB. Testosterone and the prostate: implications for the treatment of hypogonadal men. Curr Urol Rep. 2008; 9:500–505. PMID: 18947516.
Article32. Stahlman J, Britto M, Fitzpatrick S, McWhirter C, Testino SA, Brennan JJ, et al. Serum testosterone levels in non-dosed females after secondary exposure to 1.62% testosterone gel: effects of clothing barrier on testosterone absorption. Curr Med Res Opin. 2012; 28:291–301. PMID: 22188558.
Article33. Testosterone nasal gel (Natesto) for hypogonadism. Med Lett Drugs Ther. 2015; 57:73–74. PMID: 25941957.34. Conners W, Morgentaler A, Guidry M, Westfield G, Bryson N, Goldstein I. Preservation of normal concentrations of pituitary gonadotropins despite achievement of normal serum testoterone levels in hypogonadal men treated with a 4.5. nasal testosterone gel. The Journal of Urology. 2017; 197(4 Suppl):e1204.35. Rogol AD, Tkachenko N, Bryson N. NatestoTM, a novel testosterone nasal gel, normalizes androgen levels in hypogonadal men. Andrology. 2016; 4:46–54. PMID: 26695758.36. Ramasamy R. Natesto effects on testosterone, luteinizing hormone, follicle stimulating hormone and semen parameters [Internet]. Bethesda (MD): ClinicalTrials.gov;cited 2018 Jun 1. Available from: https://www.clinicaltrials.gov/ct2/show/NCT03203681?term=Intranasal+testosterone+semen&rank=1.37. Layton JB, Meier CR, Sharpless JL, Stürmer T, Jick SS, Brookhart MA. Comparative safety of testosterone dosage forms. JAMA Intern Med. 2015; 175:1187–1196. PMID: 25962056.
Article38. Di Luigi L, Sgrò P, Aversa A, Migliaccio S, Bianchini S, Botrè F, et al. Concerns about serum androgens monitoring during testosterone replacement treatments in hypogonadal male athletes: a pilot study. J Sex Med. 2012; 9:873–886. PMID: 22321254.
Article39. Snyder PJ, Lawrence DA. Treatment of male hypogonadism with testosterone enanthate. J Clin Endocrinol Metab. 1980; 51:1335–1339. PMID: 6777395.
Article40. Middleton T, Turner L, Fennell C, Savkovic S, Jayadev V, Conway AJ, et al. Complications of injectable testosterone undecanoate in routine clinical practice. Eur J Endocrinol. 2015; 172:511–517. PMID: 25637074.
Article41. Gonzalo IT, Swerdloff RS, Nelson AL, Clevenger B, Garcia R, Berman N, et al. Levonorgestrel implants (Norplant II) for male contraception clinical trials: combination with transdermal and injectable testosterone. J Clin Endocrinol Metab. 2002; 87:3562–3572. PMID: 12161475.
Article42. Ilani N, Roth MY, Amory JK, Swerdloff RS, Dart C, Page ST, et al. A new combination of testosterone and nestorone transdermal gels for male hormonal contraception. J Clin Endocrinol Metab. 2012; 97:3476–3486. PMID: 22791756.
Article43. George M, Yulia T, Svetlana K. Influence of testosterone gel treatment on spermatogenesis in men with hypogonadism. Gynecol Endocrinol. 2014; 30(Suppl 1):22–24. PMID: 25200823.
Article44. Bebb RA, Anawalt BD, Christensen RB, Paulsen CA, Bremner WJ, Matsumoto AM. Combined administration of levonorgestrel and testosterone induces more rapid and effective suppression of spermatogenesis than testosterone alone: a promising male contraceptive approach. J Clin Endocrinol Metab. 1996; 81:757–762. PMID: 8636300.
Article45. Kinniburgh D, Zhu H, Cheng L, Kicman AT, Baird DT, Anderson RA. Oral desogestrel with testosterone pellets induces consistent suppression of spermatogenesis to azoospermia in both Caucasian and Chinese men. Hum Reprod. 2002; 17:1490–1501. PMID: 12042267.
Article46. Brady BM, Walton M, Hollow N, Kicman AT, Baird DT, Anderson RA. Depot testosterone with etonogestrel implants result in induction of azoospermia in all men for long-term contraception. Hum Reprod. 2004; 19:2658–2667. PMID: 15333595.
Article47. Garrett C, Liu DY, McLachlan RI, Baker HW. Time course of changes in sperm morphometry and semen variables during testosterone-induced suppression of human spermatogenesis. Hum Reprod. 2005; 20:3091–3100. PMID: 16006469.
Article48. Kinniburgh D, Anderson RA, Baird DT. Suppression of spermatogenesis with desogestrel and testosterone pellets is not enhanced by addition of finasteride. J Androl. 2001; 22:88–95. PMID: 11191092.49. Rao PK, Boulet SL, Mehta A, Hotaling J, Eisenberg ML, Honig SC, et al. Trends in testosterone replacement therapy use from 2003 to 2013 among reproductive-age men in the United States. J Urol. 2017; 197:1121–1126. PMID: 27789218.
Article50. Liu PY, Swerdloff RS, Christenson PD, Handelsman DJ, Wang C. Hormonal Male Contraception Summit Group. Rate, extent, and modifiers of spermatogenic recovery after hormonal male contraception: an integrated analysis. Lancet. 2006; 367:1412–1420. PMID: 16650651.
Article51. Ramasamy R, Armstrong JM, Lipshultz LI. Preserving fertility in the hypogonadal patient: an update. Asian J Androl. 2015; 17:197–200. PMID: 25337850.
Article52. Coviello AD, Matsumoto AM, Bremner WJ, Herbst KL, Amory JK, Anawalt BD, et al. Low-dose human chorionic gonadotropin maintains intratesticular testosterone in normal men with testosterone-induced gonadotropin suppression. J Clin Endocrinol Metab. 2005; 90:2595–2602. PMID: 15713727.
Article53. Pelusi C, Giagulli VA, Baccini M, Fanelli F, Mezzullo M, Fazzini A, et al. Clomiphene citrate effect in obese men with low serum testosterone treated with metformin due to dysmetabolic disorders: a randomized, double-blind, placebocontrolled study. PLoS One. 2017; 12:e0183369. PMID: 28886024.
Article54. Rodriguez KM, Pastuszak AW, Lipshultz LI. Enclomiphene citrate for the treatment of secondary male hypogonadism. Expert Opin Pharmacother. 2016; 17:1561–1567. PMID: 27337642.
Article55. Kaminetsky J, Werner M, Fontenot G, Wiehle RD. Oral enclomiphene citrate stimulates the endogenous production of testosterone and sperm counts in men with low testosterone: comparison with testosterone gel. J Sex Med. 2013; 10:1628–1635. PMID: 23530575.
Article56. Chua ME, Escusa KG, Luna S, Tapia LC, Dofitas B, Morales M. Revisiting oestrogen antagonists (clomiphene or tamoxifen) as medical empiric therapy for idiopathic male infertility: a meta-analysis. Andrology. 2013; 1:749–757. PMID: 23970453.
Article57. Roth LW, Ryan AR, Meacham RB. Clomiphene citrate in the management of male infertility. Semin Reprod Med. 2013; 31:245–250. PMID: 23775379.
Article58. Tamunopriye J, Abiola OO. Human chorionic gonadotrophin (HCG) stimulation test and testosterone response in children with micropenis. Pediatr Endocrinol Rev. 2014; 12:42–45. PMID: 25345084.59. Choi J, Smitz J. Luteinizing hormone and human chorionic gonadotropin: origins of difference. Mol Cell Endocrinol. 2014; 383:203–213. PMID: 24365330.
Article60. Berger P, Gruschwitz M, Spoettl G, Dirnhofer S, Madersbacher S, Gerth R, et al. Human chorionic gonadotropin (hCG) in the male reproductive tract. Mol Cell Endocrinol. 2007; 260-262:190–196. PMID: 17097221.
Article61. Crosnoe-Shipley LE, Elkelany OO, Rahnema CD, Kim ED. Treatment of hypogonadotropic male hypogonadism: case-based scenarios. World J Nephrol. 2015; 4:245–253. PMID: 25949938.
Article62. Rastrelli G, Corona G, Mannucci E, Maggi M. Factors affecting spermatogenesis upon gonadotropin-replacement therapy: a meta-analytic study. Andrology. 2014; 2:794–808. PMID: 25271205.
Article63. Liu PY, Baker HW, Jayadev V, Zacharin M, Conway AJ, Handelsman DJ. Induction of spermatogenesis and fertility during gonadotropin treatment of gonadotropin-deficient infertile men: predictors of fertility outcome. J Clin Endocrinol Metab. 2009; 94:801–808. PMID: 19066302.
Article64. Petak SM, Nankin HR, Spark RF, Swerdloff RS, Rodriguez-Rigau LJ. American Association of Clinical Endocrinologists. American Association of Clinical Endocrinologists Medical Guidelines for clinical practice for the evaluation and treatment of hypogonadism in adult male patients--2002 update. Endocr Pract. 2002; 8:440–456. PMID: 15260010.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Management of Anabolic Steroid-Induced Infertility: Novel Strategies for Fertility Maintenance and Recovery
- Misuse of testosterone replacement therapy in men in infertile couples and its influence on infertility treatment
- Attitude of Korean Males toward Late-Onset Hypogonadism: Survey of Males in Their 40s and Above
- Correlation between Serum Total Testosterone and the AMS and IIEF Questionnaires in Patients with Erectile Dysfunction with Testosterone Deficiency Syndrome
- Testosterone Replacement Therapy and Prostate Cancer Incidence