Ann Rehabil Med.
2018 Oct;42(5):643-651. 10.5535/arm.2018.42.5.643.
Immediate Effects of a Single Exercise on Behavior and Memory in the Early Period of Traumatic Brain Injury in Rats
- Affiliations
-
- 1Department of Physical and Rehabilitation Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University, School of Medicine, Seoul, Korea.
- 2Medical Research Institute, Regenerative and Neuroscience Laboratory, Kangbuk Samsung Hospital, Sungkyunkwan University, School of Medicine, Seoul, Korea.
- 3Department of Rehabilitation Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. dykimmart@gmail.com
- KMID: 2429193
- DOI: http://doi.org/10.5535/arm.2018.42.5.643
Abstract
OBJECTIVE
To evaluate the immediate effect of single exercise on physical performance and memory in the early stage of traumatic brain injury (TBI) in rats.
METHODS
Ninety TBI rats were randomly assigned to T0 (sedentary), T10 (treadmill 10 m/min for 30 minutes), or T20 (treadmill 20 m/min for 30 minutes) groups, on day 3 (D3), D7, and D14 after TBI, respectively. Rotarod (RR), Barnes maze (BM), brain magnetic resonance imaging (MRI) and MR spectroscopy were performed immediately before and 6 hours after exercise. Rats were sacrificed for immunohistochemistry with heat shock protein 70 (Hsp70) and glial fibrillary acidic protein (GFAP).
RESULTS
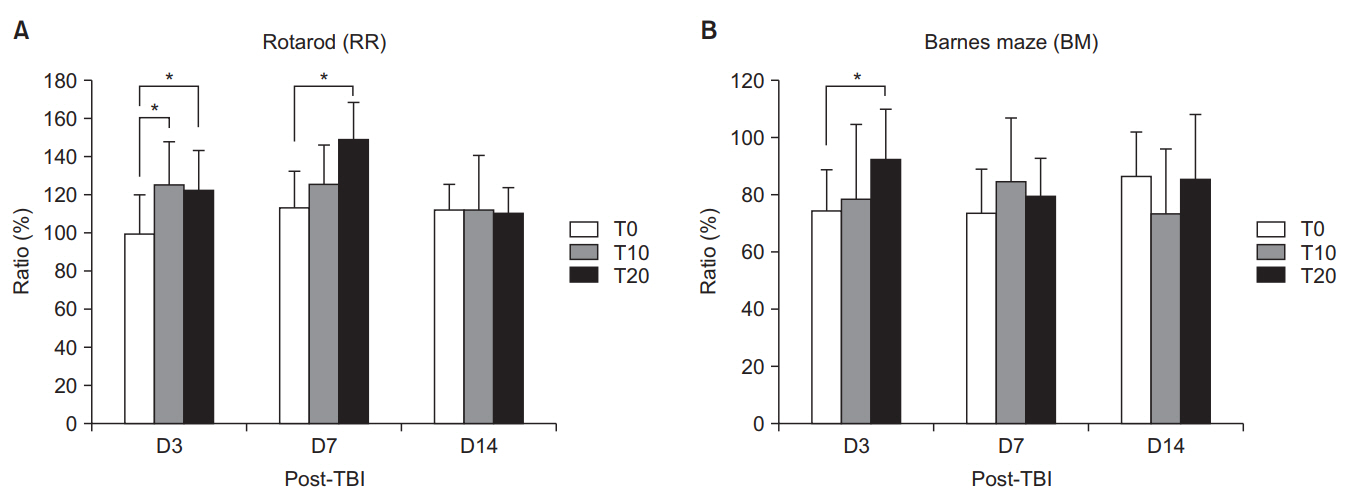
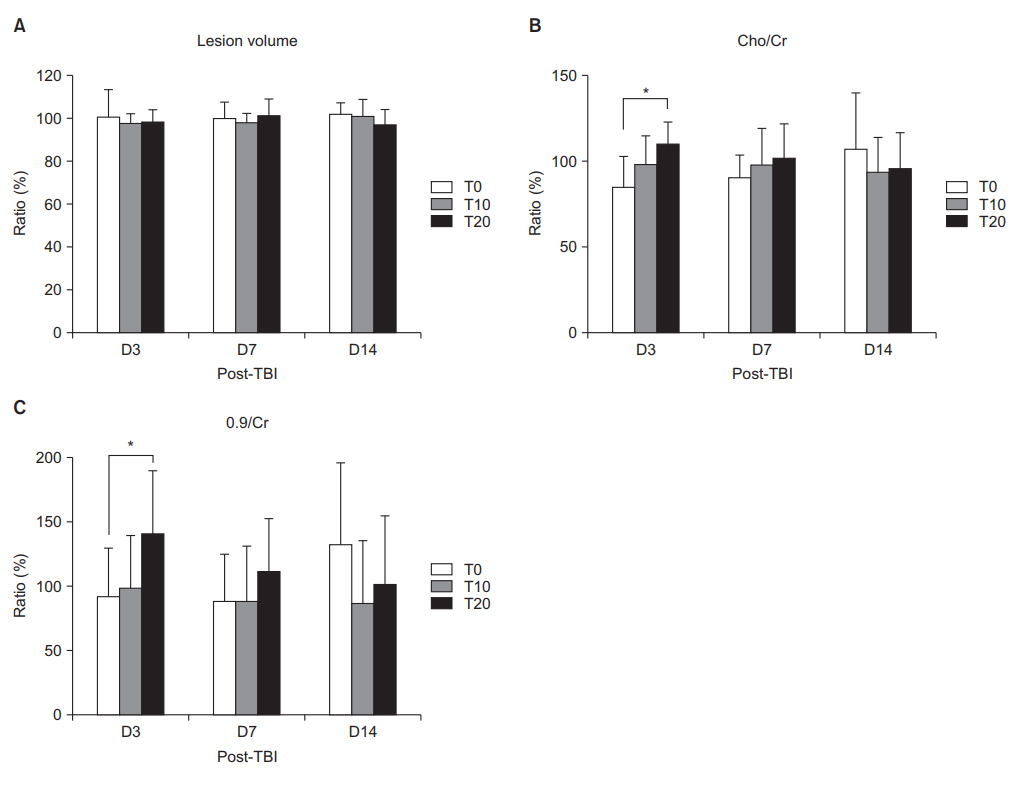
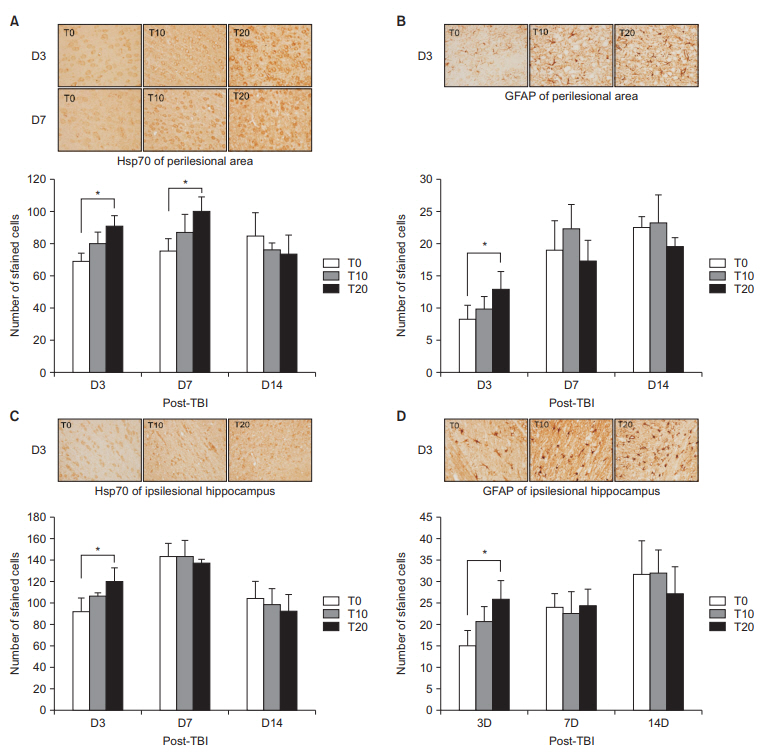
On D3, the T10 and T20 groups demonstrated significant improvement in RR (p < 0.05). On D7, only the T20 group showed significantly enhanced RR (p < 0.05). In BM on D3, the T20 group showed significant deterioration compared with the other groups (p < 0.05). Lesion volume did not significantly differ among the groups. MR spectroscopy on D3 showed that only the T20 group had significantly increased choline/creatine and 0.9/creatine (p < 0.05). In the perilesional area on D3, only T20 had a significantly higher Hsp70 and GFAP than the T0 group. On D7, Hsp70 was significantly higher in the T20 group than in the T0 group (p < 0.05). In the ipsilesional hippocampus on D3, the T20 group showed a significantly higher Hsp70 and GFAP than the T0 group (p < 0.05).
CONCLUSION
A single session of low-intensity exercise in the early period of TBI improves behavioral performance without inducing cognitive deficits. However, high-intensity exercise can exacerbate cognitive function in the early period after TBI. Therefore, the optimal timing of rehabilitation and exercise intensity are crucial in behavior and memory recovery after TBI.
Keyword
MeSH Terms
Figure
Reference
-
1. Hyder AA, Wunderlich CA, Puvanachandra P, Gururaj G, Kobusingye OC. The impact of traumatic brain injuries: a global perspective. NeuroRehabilitation. 2007; 22:341–53.
Article2. Ramanathan DM, McWilliams N, Schatz P, Hillary FG. Epidemiological shifts in elderly traumatic brain injury: 18-year trends in Pennsylvania. J Neurotrauma. 2012; 29:1371–8.
Article3. Brazinova A, Rehorcikova V, Taylor MS, Buckova V, Majdan M, Psota M, et al. Epidemiology of traumatic brain injury in Europe: a living systematic Review. J Neurotrauma. 2016; Aug. 25. [Epub]. http://10.1089/neu.2015.4126.
Article4. Menon DK, Maas AI. Traumatic brain injury in 2014: progress, failures and new approaches for TBI research. Nat Rev Neurol. 2015; 11:71–2.5. Swain RA, Harris AB, Wiener EC, Dutka MV, Morris HD, Theien BE, et al. Prolonged exercise induces angiogenesis and increases cerebral blood volume in primary motor cortex of the rat. Neuroscience. 2003; 117:1037–46.
Article6. van Praag H, Kempermann G, Gage FH. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci. 1999; 2:266–70.
Article7. Bernardi C, Tramontina AC, Nardin P, Biasibetti R, Costa AP, Vizueti AF, et al. Treadmill exercise induces hippocampal astroglial alterations in rats. Neural Plast. 2013; 2013:709732.
Article8. Saur L, Baptista PP, de Senna PN, Paim MF, do Nascimento P, Ilha J, et al. Physical exercise increases GFAP expression and induces morphological changes in hippocampal astrocytes. Brain Struct Funct. 2014; 219:293–302.
Article9. Keeler BE, Liu G, Siegfried RN, Zhukareva V, Murray M, Houle JD. Acute and prolonged hindlimb exercise elicits different gene expression in motoneurons than sensory neurons after spinal cord injury. Brain Res. 2012; 1438:8–21.
Article10. Kozlowski DA, James DC, Schallert T. Use-dependent exaggeration of neuronal injury after unilateral sensorimotor cortex lesions. J Neurosci. 1996; 16:4776–86.
Article11. Humm JL, Kozlowski DA, James DC, Gotts JE, Schallert T. Use-dependent exacerbation of brain damage occurs during an early post-lesion vulnerable period. Brain Res. 1998; 783:286–92.
Article12. Griesbach GS. Exercise after traumatic brain injury: is it a double-edged sword? PM R. 2011; 3(6 Suppl 1):S64–72.
Article13. Chang YK, Labban JD, Gapin JI, Etnier JL. The effects of acute exercise on cognitive performance: a metaanalysis. Brain Res. 2012; 1453:87–101.
Article14. McDonnell MN, Buckley JD, Opie GM, Ridding MC, Semmler JG. A single bout of aerobic exercise promotes motor cortical neuroplasticity. J Appl Physiol (1985). 2013; 114:1174–82.
Article15. McIntosh TK, Vink R, Noble L, Yamakami I, Fernyak S, Soares H, et al. Traumatic brain injury in the rat: characterization of a lateral fluid-percussion model. Neuroscience. 1989; 28:233–44.
Article16. Fonoff ET, Pereira JF Jr, Camargo LV, Dale CS, Pagano RL, Ballester G, et al. Functional mapping of the motor cortex of the rat using transdural electrical stimulation. Behav Brain Res. 2009; 202:138–41.
Article17. Yoon KJ, Oh BM, Kim DY. Functional improvement and neuroplastic effects of anodal transcranial direct current stimulation (tDCS) delivered 1 day vs. 1 week after cerebral ischemia in rats. Brain Res. 2012; 1452:61–72.18. Garnett MR, Blamire AM, Corkill RG, Cadoux-Hudson TA, Rajagopalan B, Styles P. Early proton magnetic resonance spectroscopy in normal-appearing brain correlates with outcome in patients following traumatic brain injury. Brain. 2000; 123(Pt 10):2046–54.
Article19. Li R, Fujitani N, Jia JT, Kimura H. Immunohistochemical indicators of early brain injury: an experimental study using the fluid-percussion model in cats. Am J Forensic Med Pathol. 1998; 19:129–36.
Article20. Zanier ER, Lee SM, Vespa PM, Giza CC, Hovda DA. Increased hippocampal CA3 vulnerability to low-level kainic acid following lateral fluid percussion injury. J Neurotrauma. 2003; 20:409–20.
Article21. Ginsberg MD, Sternau LL, Globus MY, Dietrich WD, Busto R. Therapeutic modulation of brain temperature: relevance to ischemic brain injury. Cerebrovasc Brain Metab Rev. 1992; 4:189–225.22. Borer KT, Bestervelt LL, Mannheim M, Brosamer MB, Thompson M, Swamy U, et al. Stimulation by voluntary exercise of adrenal glucocorticoid secretion in mature female hamsters. Physiol Behav. 1992; 51:713–8.
Article23. Smith-Swintosky VL, Pettigrew LC, Sapolsky RM, Phares C, Craddock SD, Brooke SM, et al. Metyrapone, an inhibitor of glucocorticoid production, reduces brain injury induced by focal and global ischemia and seizures. J Cereb Blood Flow Metab. 1996; 16:585–98.
Article24. DeVries AC, Joh HD, Bernard O, Hattori K, Hurn PD, Traystman RJ, et al. Social stress exacerbates stroke outcome by suppressing Bcl-2 expression. Proc Natl Acad Sci U S A. 2001; 98:11824–8.
Article25. Vanderwolf CH, Cain DP. The behavioral neurobiology of learning and memory: a conceptual reorientation. Brain Res Brain Res Rev. 1994; 19:264–97.
Article26. Qu M, Mittmann T, Luhmann HJ, Schleicher A, Zilles K. Long-term changes of ionotropic glutamate and GABA receptors after unilateral permanent focal cerebral ischemia in the mouse brain. Neuroscience. 1998; 85:29–43.
Article27. Griesbach GS, Hovda DA, Molteni R, Wu A, Gomez-Pinilla F. Voluntary exercise following traumatic brain injury: brain-derived neurotrophic factor upregulation and recovery of function. Neuroscience. 2004; 125:129–39.
Article28. Hoffman-Goetz L, Spagnuolo PA, Guan J. Repeated exercise in mice alters expression of IL-10 and TNFalpha in intestinal lymphocytes. Brain Behav Immun. 2008; 22:195–9.29. McEwen BS, Magarinos AM. Stress and hippocampal plasticity: implications for the pathophysiology of affective disorders. Hum Psychopharmacol. 2001; 16(S1):S7–S19.
Article30. Hansson AC, Sommer WH, Metsis M, Stromberg I, Agnati LF, Fuxe K. Corticosterone actions on the hippocampal brain-derived neurotrophic factor expression are mediated by exon IV promoter. J Neuroendocrinol. 2006; 18:104–14.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Characteristics of Memory Disturbance in Patients with Traumatic Brain Injury Referred for Mental Disabilitiy Evaluation
- Neuropsychiatric Treatment of Patients with Traumatic Brain Injury
- The use of Amantadine in Traumatic Brain Injury Patients
- The Impact of Anxiety Symptoms on Cognitive Function in Patients with Mild Traumatic Brain Injury
- Therapeutic Effect of Amantadine in Traumatic Brain Injury Patients: Two Cases and Review