Yonsei Med J.
2018 Dec;59(10):1214-1221. 10.3349/ymj.2018.59.10.1214.
Serum Periostin Levels: A Potential Serologic Marker for Toluene Diisocyanate-Induced Occupational Asthma
- Affiliations
-
- 1Department of Internal Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea.
- 2Department of Allergy and Clinical Immunology, Ajou University School of Medicine, Suwon, Korea. hspark@ajou.ac.kr
- 3Department of Pulmonary, Allergy and Critical Care Medicine, Kangdong Sacred Heart Hospital, Hallym University College of Medicine, Seoul, Korea.
- 4Division of Medical Biochemistry, Department of Biomolecular Sciences, Saga Medical School, Saga, Japan.
- KMID: 2426336
- DOI: http://doi.org/10.3349/ymj.2018.59.10.1214
Abstract
- PURPOSE
Toluene diisocyanate (TDI) is a leading cause of occupational asthma (OA). Periostin is a matricellular protein implicated in type 2 immunity-driven asthma. Its pathogenic role in TDI-OA has not been completely elucidated. The present study was performed to investigate the role of periostin in TDI-OA.
MATERIALS AND METHODS
Serum periostin levels were measured in subjects with TDI-OA, asymptomatic TDI-exposure controls (AECs), non-occupational asthmatics (NAs), and unexposed normal controls (NCs). To understand the mechanism by which TDI induces periostin production, primary small airway epithelial cells (SAECs) were cultured under stimulation of TDI and neutrophils from asthmatic patients.
RESULTS
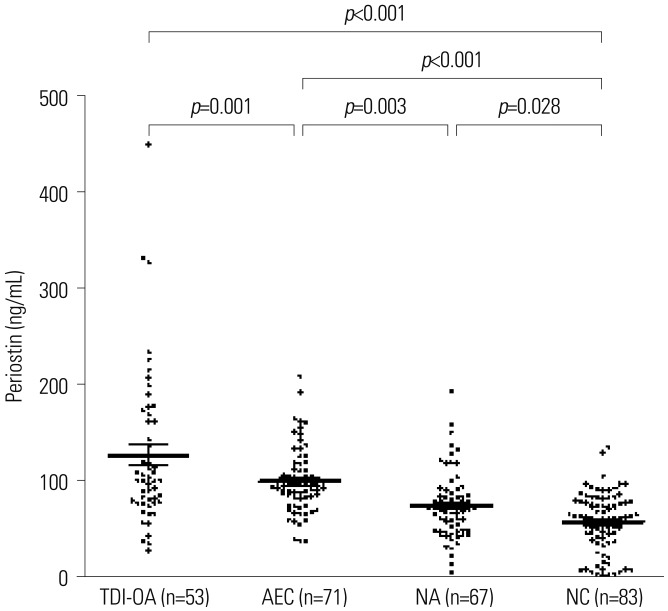
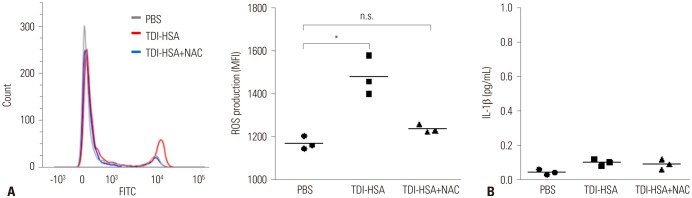
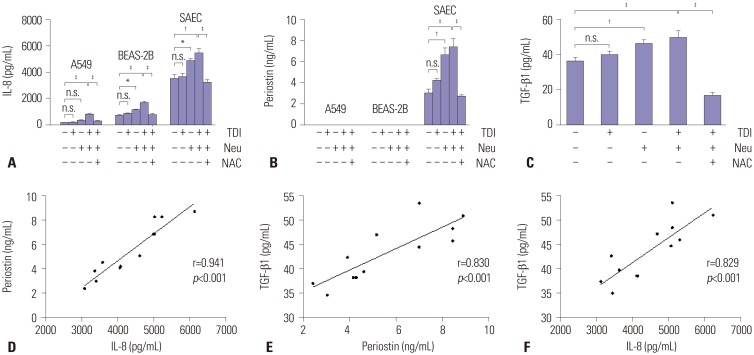
Fifty-three subjects with TDI-OA, 71 AECs, 67 NAs, and 83 NCs were enrolled. Serum periostin levels were significantly higher in TDI-OA subjects than in AECs (p=0.001), NAs (p < 0.001), and NCs (p < 0.001). In TDI-exposed subjects (TDI-OA and AEC), the PC20 methacholine levels were significantly lower in subjects with a higher periostin level than in those with a lower periostin level. TDI exposure did not increase periostin production directly by SAECs; however, periostin production increased significantly after co-culture with TDI and neutrophils, which was suppressed by an antioxidant. In addition, increased release of TGF-β1 was noted from SAECs when exposed to TDI and neutrophils, which was also suppressed by an antioxidant.
CONCLUSION
These results suggest that an increased periostin level may contribute to the progression of airway inflammation to remodeling in TDI-exposed workers. A high serum periostin level is a potential serologic marker of the phenotype of TDI-OA.
MeSH Terms
Figure
Reference
-
1. Torén K, Blanc PD. Asthma caused by occupational exposures is common - a systematic analysis of estimates of the population-attributable fraction. BMC Pulm Med. 2009; 9:7. PMID: 19178702.
Article2. Cassidy LD, Doney B, Wang ML, Kurth L, Conner PR, Collins JJ, et al. Medical monitoring for occupational asthma among toluene diisocyanate production workers in the United States. J Occup Environ Med. 2017; 59(Suppl 12):S13–S21. PMID: 29200134.
Article3. Rüegger M, Droste D, Hofmann M, Jost M, Miedinger D. Diisocyanate-induced asthma in Switzerland: long-term course and patients' self-assessment after a 12-year follow-up. J Occup Med Toxicol. 2014; 9:21. PMID: 24949081.
Article4. Collins JJ, Anteau S, Conner PR, Cassidy LD, Doney B, Wang ML, et al. Incidence of occupational asthma and exposure to toluene diisocyanate in the United States toluene diisocyanate production industry. J Occup Environ Med. 2017; 59(Suppl 12):S22–S27. PMID: 29200135.
Article5. Diller WF. Frequency and trends of occupational asthma due to toluene diisocyanate: a critical review. Appl Occup Environ Hyg. 2002; 17:872–877. PMID: 12495598.
Article6. Vandenplas O. Occupational asthma: etiologies and risk factors. Allergy Asthma Immunol Res. 2011; 3:157–167. PMID: 21738881.
Article7. Parulekar AD, Atik MA, Hanania NA. Periostin, a novel biomarker of TH2-driven asthma. Curr Opin Pulm Med. 2014; 20:60–65. PMID: 24247042.
Article8. Takayama G, Arima K, Kanaji T, Toda S, Tanaka H, Shoji S, et al. Periostin: a novel component of subepithelial fibrosis of bronchial asthma downstream of IL-4 and IL-13 signals. J Allergy Clin Immunol. 2006; 118:98–104. PMID: 16815144.
Article9. Ferrando M, Bagnasco D, Varricchi G, Bernardi S, Bragantini A, Passalacqua G, et al. Personalized medicine in allergy. Allergy Asthma Immunol Res. 2017; 9:15–24. PMID: 27826958.
Article10. Liu AY, Zheng H, Ouyang G. Periostin, a multifunctional matricellular protein in inflammatory and tumor microenvironments. Matrix Biol. 2014; 37:150–156. PMID: 24813586.
Article11. Wardzyńska A, Makowska JS, Pawełczyk M, Piechota-Polańczyk A, Kurowski M, Kowalski ML. Periostin in exhaled breath condensate and in serum of asthmatic patients: relationship to upper and lower airway disease. Allergy Asthma Immunol Res. 2017; 9:126–132. PMID: 28102057.
Article12. Kim DW, Cho SH. Emerging endotypes of chronic rhinosinusitis and its application to precision medicine. Allergy Asthma Immunol Res. 2017; 9:299–306. PMID: 28497916.
Article13. Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, et al. Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000; 161:309–329. PMID: 10619836.14. Park HS, Kim HY, Nahm DH, Son JW, Kim YY. Specific IgG, but not specific IgE, antibodies to toluene diisocyanate-human serum albumin conjugate are associated with toluene diisocyanate bronchoprovocation test results. J Allergy Clin Immunol. 1999; 104(4 Pt 1):847–851. PMID: 10518831.
Article15. Mhike M, Hettick JM, Chipinda I, Law BF, Bledsoe TA, Lemons AR, et al. Characterization and comparative analysis of 2,4-toluene diisocyanate and 1,6-hexamethylene diisocyanate haptenated human serum albumin and hemoglobin. J Immunol Methods. 2016; 431:38–44. PMID: 26853746.
Article16. Pham DL, Ban GY, Kim SH, Shin YS, Ye YM, Chwae YJ, et al. Neutrophil autophagy and extracellular DNA traps contribute to airway inflammation in severe asthma. Clin Exp Allergy. 2017; 47:57–70. PMID: 27883241.
Article17. Ban GY, Pham DL, Trinh TH, Lee SI, Suh DH, Yang EM, et al. Autophagy mechanisms in sputum and peripheral blood cells of patients with severe asthma: a new therapeutic target. Clin Exp Allergy. 2016; 46:48–59. PMID: 26112695.
Article18. Izuhara K, Matsumoto H, Ohta S, Ono J, Arima K, Ogawa M. Recent developments regarding periostin in bronchial asthma. Allergol Int. 2015; 64(Suppl):S3–S10. PMID: 26344077.
Article19. Izuhara K, Ohta S, Ono J. Using periostin as a biomarker in the treatment of asthma. Allergy Asthma Immunol Res. 2016; 8:491–498. PMID: 27582399.
Article20. Yuyama N, Davies DE, Akaiwa M, Matsui K, Hamasaki Y, Suminami Y, et al. Analysis of novel disease-related genes in bronchial asthma. Cytokine. 2002; 19:287–296. PMID: 12421571.
Article21. Woodruff PG, Modrek B, Choy DF, Jia G, Abbas AR, Ellwanger A, et al. T-helper type 2-driven inflammation defines major subphenotypes of asthma. Am J Respir Crit Care Med. 2009; 180:388–395. PMID: 19483109.
Article22. Peters MC, Mekonnen ZK, Yuan S, Bhakta NR, Woodruff PG, Fahy JV. Measures of gene expression in sputum cells can identify TH2-high and TH2-low subtypes of asthma. J Allergy Clin Immunol. 2014; 133:388–394. PMID: 24075231.
Article23. Corren J, Lemanske RF, Hanania NA, Korenblat PE, Parsey MV, Arron JR, et al. Lebrikizumab treatment in adults with asthma. N Engl J Med. 2011; 365:1088–1098. PMID: 21812663.
Article24. Hanania NA, Wenzel S, Rosén K, Hsieh HJ, Mosesova S, Choy DF, et al. Exploring the effects of omalizumab in allergic asthma: an analysis of biomarkers in the EXTRA study. Am J Respir Crit Care Med. 2013; 187:804–811. PMID: 23471469.25. Kanemitsu Y, Matsumoto H, Izuhara K, Tohda Y, Kita H, Horiguchi T, et al. Increased periostin associates with greater airflow limitation in patients receiving inhaled corticosteroids. J Allergy Clin Immunol. 2013; 132:305–312. PMID: 23791506.
Article26. Kim MA, Izuhara K, Ohta S, Ono J, Yoon MK, Ban GY, et al. Association of serum periostin with aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2014; 113:314–320. PMID: 25037608.
Article27. Shin YS, Kim MA, Pham LD, Park HS. Cells and mediators in diisocyanate-induced occupational asthma. Curr Opin Allergy Clin Immunol. 2013; 13:125–131. PMID: 23324746.
Article28. Lange RW, Day BW, Lemus R, Tyurin VA, Kagan VE, Karol MH. Intracellular S-glutathionyl adducts in murine lung and human bronchoepithelial cells after exposure to diisocyanatotoluene. Chem Res Toxicol. 1999; 12:931–936. PMID: 10525268.
Article29. Muti AD, Pârvu AE, Muti LA, Moldovan R, Mureşan A. Vitamin E effect in a rat model of toluene diisocyanate-induced asthma. Clujul Med. 2016; 89:499–505. PMID: 27857519.
Article30. Park HS, Kim SR, Lee YC. Impact of oxidative stress on lung diseases. Respirology. 2009; 14:27–38. PMID: 19144046.
Article31. Qu J, Li Y, Zhong W, Gao P, Hu C. Recent developments in the role of reactive oxygen species in allergic asthma. J Thorac Dis. 2017; 9:E32–E43. PMID: 28203435.
Article32. Lee YM, Kim HA, Park HS, Lee SK, Nahm DH. Exposure to toluene diisocyanate (TDI) induces IL-8 production from bronchial epithelial cells: effect of pro-inflammatory cytokines. J Korean Med Sci. 2003; 18:809–812. PMID: 14676436.
Article33. Winterbourn CC, Kettle AJ, Hampton MB. Reactive oxygen species and neutrophil function. Annu Rev Biochem. 2016; 85:765–792. PMID: 27050287.
Article34. Ojiaku CA, Yoo EJ, Panettieri RA Jr. Transforming growth factor β1 function in airway remodeling and hyperresponsiveness. The missing link? Am J Respir Cell Mol Biol. 2017; 56:432–442. PMID: 27854509.
Article35. Gabehart KE, Royce SG, Maselli DJ, Miyasato SK, Davis EC, Tang ML, et al. Airway hyperresponsiveness is associated with airway remodeling but not inflammation in aging Cav1−/− mice. Respir Res. 2013; 14:110. PMID: 24138138.
Article36. Sidhu SS, Yuan S, Innes AL, Kerr S, Woodruff PG, Hou L, et al. Roles of epithelial cell-derived periostin in TGF-beta activation, collagen production, and collagen gel elasticity in asthma. Proc Natl Acad Sci U S A. 2010; 107:14170–14175. PMID: 20660732.37. Park HS, Kim HA, Jung JW, Kim YK, Lee SK, Kim SS, et al. Metalloproteinase-9 is increased after toluene diisocyanate exposure in the induced sputum from patients with toluene diisocyanate-induced asthma. Clin Exp Allergy. 2003; 33:113–118. PMID: 12534559.
Article38. Okamoto M, Hoshino T, Kitasato Y, Sakazaki Y, Kawayama T, Fujimoto K, et al. Periostin, a matrix protein, is a novel biomarker for idiopathic interstitial pneumonias. Eur Respir J. 2011; 37:1119–1127. PMID: 21177844.
Article39. Uchida M, Shiraishi H, Ohta S, Arima K, Taniguchi K, Suzuki S, et al. Periostin, a matricellular protein, plays a role in the induction of chemokines in pulmonary fibrosis. Am J Respir Cell Mol Biol. 2012; 46:677–686. PMID: 22246863.
Article40. Pham DL, Trinh TH, Ban GY, Kim SH, Park HS. Epithelial folliculin is involved in airway inflammation in workers exposed to toluene diisocyanate. Exp Mol Med. 2017; 49:e395. PMID: 29147010.
Article41. Choi GS, Trinh HKT, Yang EM, Ye YM, Shin YS, Kim SH, et al. Role of clusterin/progranulin in toluene diisocyanate-induced occupational asthma. Exp Mol Med. 2018; 50:58. PMID: 29717106.42. Izuhara K, Nunomura S, Nanri Y, Ogawa M, Ono J, Mitamura Y, et al. Periostin in inflammation and allergy. Cell Mol Life Sci. 2017; 74:4293–4303. PMID: 28887633.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A case of occupational asthma with systemic illness due to toluene diisocyanate and diphenylmethane diisocyanate
- Biomarkers Predicting Isocyanate-Induced Asthma
- Occupational Asthma due to Styrene and Toluene Diisocyanate
- Clinical significance of specific IgG and IgE antibodies to toluene diisocyanate ( TDi ) - human - serum albumin ( HSA ) conjugate in TDI - induceed occupational asthma
- Evidence for neutrophil activation in induced sputum from patients with TDI - induced occupational asthma