Ann Lab Med.
2019 Mar;39(2):150-157. 10.3343/alm.2019.39.2.150.
Comparison of High Sensitivity and Conventional Flow Cytometry for Diagnosing Overt Paroxysmal Nocturnal Hemoglobinuria and Detecting Minor Paroxysmal Nocturnal Hemoglobinuria Clones
- Affiliations
-
- 1Department of Laboratory Medicine, University of Ulsan College of Medicine, Ulsan University Hospital, Ulsan, Korea. uucm77@gmail.com
- 2Department of Laboratory Medicine, Pusan National University Yangsan Hospital, Yangsan, Korea.
- 3Department of Hematology and Cellular Therapy, University of Ulsan College of Medicine, Ulsan University Hospital, Ulsan, Korea.
- KMID: 2425970
- DOI: http://doi.org/10.3343/alm.2019.39.2.150
Abstract
- BACKGROUND
High sensitivity flow cytometry (HS-FCM) was recently developed for diagnosing paroxysmal nocturnal hemoglobinuria (PNH). We compared its performance with conventional flow cytometry (C-FCM) for diagnosing overt PNH and detecting minor (0.1-1%) PNH clones in aplastic anemia (AA)/low-grade myelodysplastic syndrome (MDS) patients.
METHODS
C-FCM and HS-FCM were performed simultaneously on 41 samples from healthy controls and 23 peripheral blood samples from 15 AA/low-grade MDS and eight PNH patients, using a Navios flow cytometer (Beckman Coulter, Miami, FL, USA). Results were compared.
RESULTS
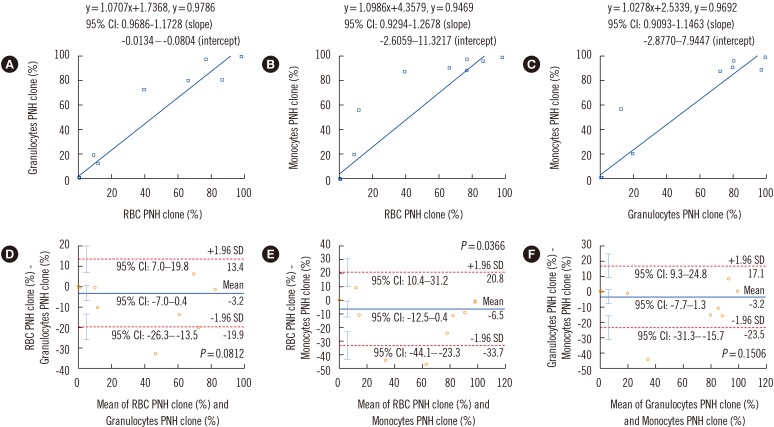
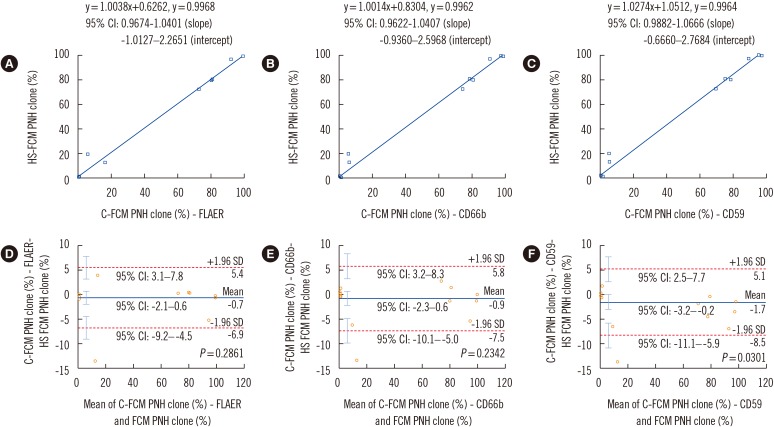
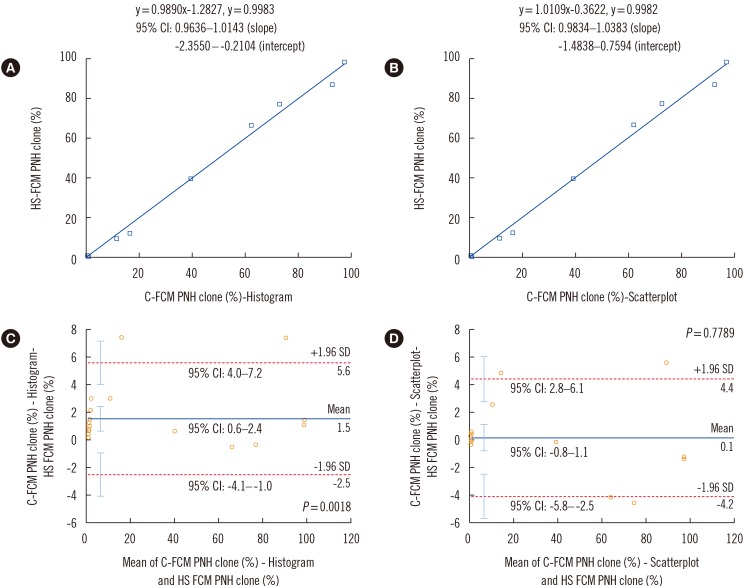
No healthy control samples had PNH clone size >0.01%. For granulocytes, C-FCM detected a smaller PNH clone size than HS-FCM (mean difference: 0.7-1.7%). In AA/low-grade MDS patients, three samples showed >1% PNH clones with C-FCM but not with HS-FCM. Seven samples showed minor PNH clones by C-FCM, but HS-FCM showed negative results for all these samples. In PNH patients, C-FCM detected a smaller PNH clone size than HS-FCM (mean difference: 1.9-5.0%). For red blood cells, C-FCM detected a greater PNH clone size than HS-FCM (mean difference: 1.5%). In AA/low-grade MDS patients, C-FCM showed >1% PNH clones in six samples, but HS-FCM showed >1% PNH clones in none of the samples. C-FCM detected minor PNH clones in nine samples, but six of them were negative by HS-FCM. In PNH patients, C-FCM detected a greater PNH clone size than HS-FCM (mean difference: 2.5%).
CONCLUSIONS
HS-FCM can sensitively detect minor PNH clones and reduce false-positive C-FCM minor PNH clone cases in AA/low-grade MDS patients.
MeSH Terms
Figure
Reference
-
1. Miyata T, Takeda J, Iida Y, Yamada N, Inoue N, Takahashi M, et al. The cloning of PIG-A, a component in the early step of GPI-anchor biosynthesis. Science. 1993; 259:1318–1320. PMID: 7680492.2. Takeda J, Miyata T, Kawagoe K, Iida Y, Endo Y, Fujita T, et al. Deficiency of the GPI anchor caused by a somatic mutation of the PIG-A gene in paroxysmal nocturnal hemoglobinuria. Cell. 1993; 73:703–711. PMID: 8500164.3. Hillmen P, Lewis SM, Bessler M, Luzzatto L, Dacie JV. Natural history of paroxysmal nocturnal hemoglobinuria. N Engl J Med. 1995; 333:1253–1258. PMID: 7566002.4. Parker CJ. Bone marrow failure syndromes: paroxysmal nocturnal hemoglobinuria. Hematol Oncol Clin North Am. 2009; 23:333–346. PMID: 19327587.5. Richards SJ, Rawstron AC, Hillmen P. Application of flow cytometry to the diagnosis of paroxysmal nocturnal hemoglobinuria. Cytometry. 2000; 42:223–233. PMID: 10934341.6. Raza A, Ravandi F, Rastogi A, Bubis J, Lim SH, Weitz I, et al. A prospective multicenter study of paroxysmal nocturnal hemoglobinuria cells in patients with bone marrow failure. Cytometry B Clin Cytom. 2014; 86:175–182. PMID: 24227693.7. Sugimori C, Chuhjo T, Feng X, Yamazaki H, Takami A, Teramura M, et al. Minor population of CD55–CD59-blood cells predicts response to immunosuppressive therapy and prognosis in patients with aplastic anemia. Blood. 2006; 107:1308–1314. PMID: 16179371.8. Dunn DE, Tanawattanacharoen P, Boccuni P, Nagakura S, Green SW, Kirby MR, et al. Paroxysmal nocturnal hemoglobinuria cells in patients with bone marrow failure syndromes. Ann Intern Med. 1999; 131:401–408. PMID: 10498555.9. Tichelli A, Gratwohl A, Nissen C, Speck B. Late clonal complications in severe aplastic anemia. Leuk Lymphoma. 1994; 12:167–175. PMID: 8167549.10. Parker C, Omine M, Richards S, Nishimura J, Bessler M, Ware R, et al. Diagnosis and management of paroxysmal nocturnal hemoglobinuria. Blood. 2005; 106:3699–3709. PMID: 16051736.11. Borowitz MJ, Craig FE, Digiuseppe JA, Illingworth AJ, Rosse W, Sutherland DR, et al. Guidelines for the diagnosis and monitoring of paroxysmal nocturnal hemoglobinuria and related disorders by flow cytometry. Cytometry B Clin Cytom. 2010; 78:211–230. PMID: 20533382.12. Sutherland DR, Keeney M, Illingworth A. Practical guidelines for the high-sensitivity detection and monitoring of paroxysmal nocturnal hemoglobinuria clones by flow cytometry. Cytometry B Clin Cytom. 2012; 82:195–208. PMID: 22499484.13. Sutherland DR, Kuek N, Azcona-Olivera J, Anderson T, Acton E, Barth D, et al. Use of a FLAER-based white blood cell assay in the primary screening of PNH clones. Am J Clin Pathol. 2009; 132:564–572. PMID: 19762534.14. Marinov I, Kohoutová M, Tkáčová V, Lysák D, Holubová M, Stehlíková O, et al. Intra- and interlaboratory variability of paroxysmal nocturnal hemoglobinuria testing by flow cytometry following the 2012 Practical Guidelines for high-sensitivity paroxysmal nocturnal hemoglobinuria testing. Cytometry B Clin Cytom. 2013; 84:229–236. PMID: 23325604.15. Sipol AA, Babenko EV, Borisov VI, Naumova EV, Boyakova EV, Yakunin DI, et al. An inter-laboratory comparison of PNH clone detection by high-sensitivity flow cytometry in a Russian cohort. Hematology. 2015; 20:31–38. PMID: 24666187.16. Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016; 127:2391–2405. PMID: 27069254.17. Illingworth A, Marinov I, Sutherland DR, Wagner-Ballon O, DelVecchio L. ICCS/ESCCA consensus guidelines to detect GPI-deficient cells in paroxysmal nocturnal hemoglobinuria (PNH) and related disorders part 3-data analysis, reporting and case studies. Cytometry B Clin Cytom. 2018; 94:49–66. PMID: 29236350.18. Sutherland DR, Illingworth A, Marinov I, Ortiz F, Andreasen J, Payne D, et al. ICCS/ESCCA consensus guidelines to detect GPI-deficient cells in paroxysmal nocturnal hemoglobinuria (PNH) and related disorders part 2-reagent selection and assay optimization for high-sensitivity testing. Cytometry B Clin Cytom. 2018; 94:23–48. PMID: 29236353.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Paroxysmal Nocturnal Hemoglobinuria
- Paroxysmal Nocturnal Hemoglobinuria Presenting as Recurrent Jejunitis
- Delivery in a Patient with Paroxysmal Nocturnal Hemoglobinuria Successfully Managed with Low Molecular Weight Heparin Therapy
- Progressive Moyamoya Syndrome Associated with Paroxysmal Nocturnal Hemoglobinuria
- Acute Renal Failure in Paroxysmal Nocturnal Hemoglobinuria