Korean J Nutr.
2009 Dec;42(8):714-722.
Effect of Leucine Intake on Body Weight Reduction in Rats Fed High Fat Diet
- Affiliations
-
- 1Department of Nutritional Science and Food Management, Ewha Womans University, Seoul 120-750, Korea. mkk@ewha.ac.kr
Abstract
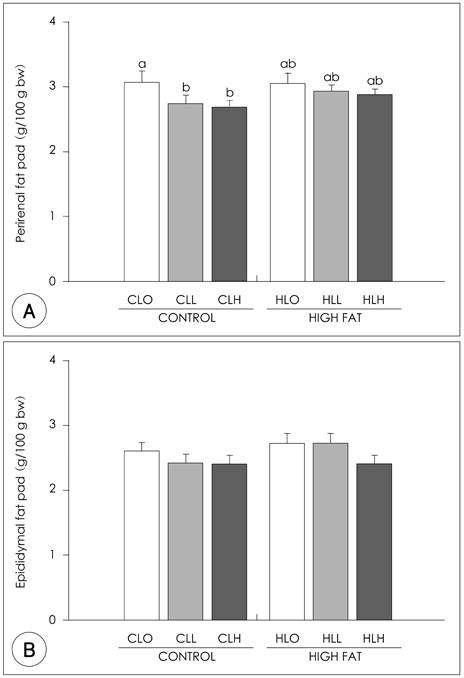
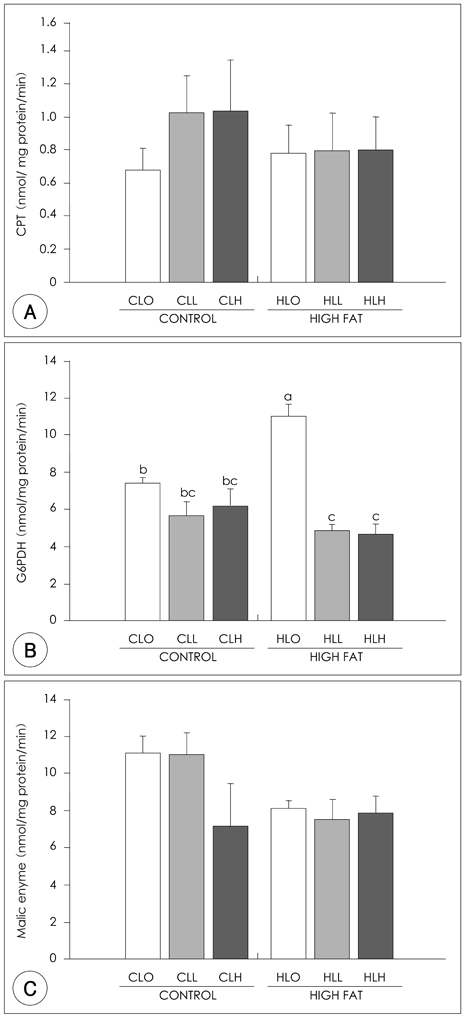
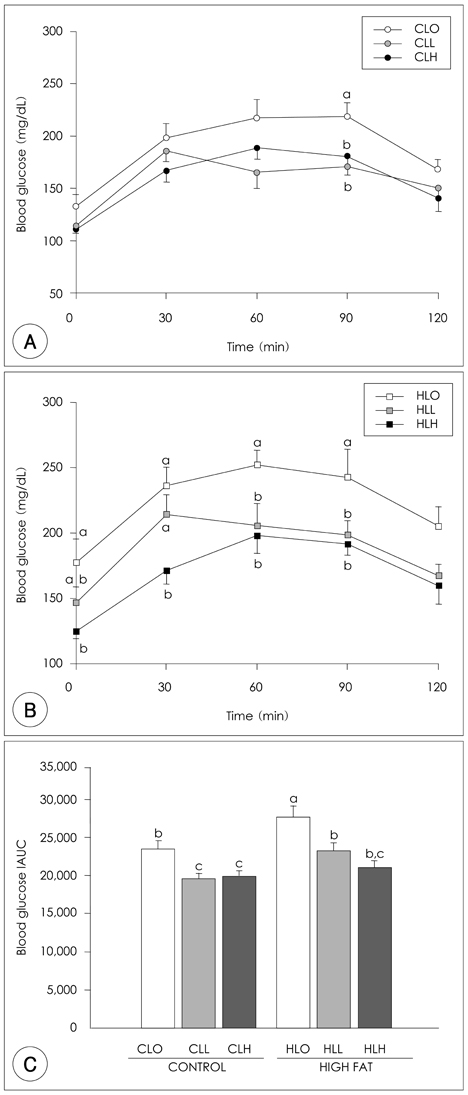
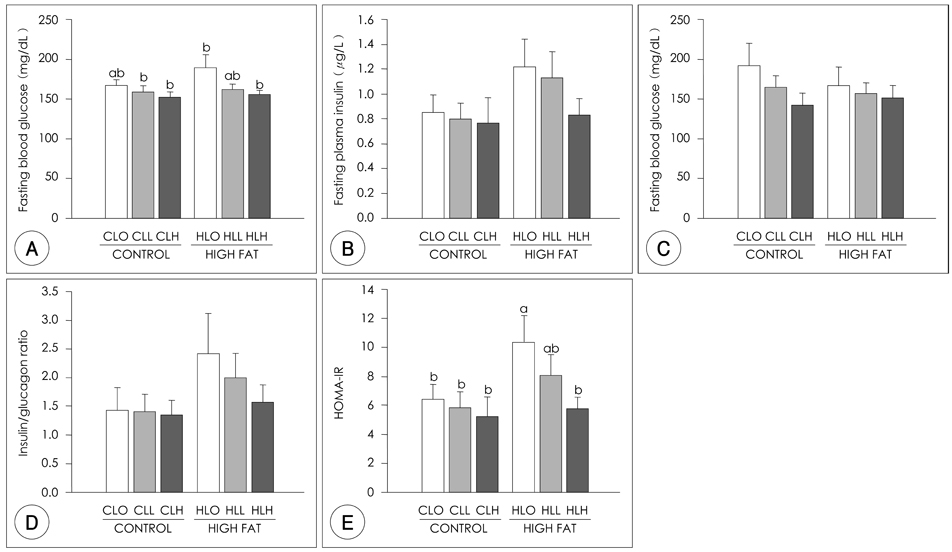
- The principal objective of this study was to determine the effects of leucine on body weight reduction in high fat diet-induced overweight rats. To induce overweight, six-month-old male Sprague-Dawley rats (n = 80) were divided into 8 groups; one group of 10 rats was fed on a normal fat diet and the remaining 70 rats were fed on a high-fat diet (40% of energy as fat) for 14 weeks. Then, 10 rats fed on the normal fat diet and another 10 rats fed on the high fat diet were sacrificed to identify overweight induction. The remaining 60 rats were divided randomly into 6 groups according to body weight and fed on one of the diets with different dietary fat levels (9.6% or 40% of energy as fat) and leucine levels (0, 0.6 or 1.2 g/kg BW) for the following 5 weeks of experiments. The body weight loss in the Leu-administered groups (0.6 g, 1.2 g/kg BW) was significantly higher than those of Leu non-administered groups. The perirenal fat pad weights in the Leu-administered groups were significantly lower than those of the Leu non-administered groups. Of the hepatic enzymes, glucose-6-phosphate dehydrogenase (G6PDH) activities were reduced significantly in the Leu-administered groups than in the Leu non-administered groups. With the oral glucose tolerance test (OGTT), the incremental areas under the curve of the glucose response (IAUC) of the Leu-administered groups were significantly lower than those of the Leu non-administered groups. The fasting glucose concentration and HOMA-IR of the Leu-administered groups were significantly lower than those of the Leu non-administered groups. In conclusion, the results of this study suggest that one of the possible mechanisms of leucine in the observed body weight reduction might involve the inhibition of lipogenic enzyme activities such as glucose-6-phosphate dehydrogenase, rather than the activation of lipolysis enzymes. Additionally, leucine adminstration resulted in improved glucose metabolism.
MeSH Terms
Figure
Reference
-
1. Park JW, Park HM, Ha NS. A study on the obesity and weight control methods of college students. J Korean Acad Psych Mental Health Nurs. 2004. 13(1):5–13.2. WHO. Fact sheet No 311. Obesity and overweight. 2006.3. Kim Eun Mi, Lee Jong Ho. Dietary protein and obesity. Korean J Obes. 2008. 17(3):101–109.4. Hu FB. Protein, body weight, and cardiovascular health. Am J Clin Nutr. 2005. 82:1 Suppl. 242S–247S.
Article5. Buchholz AC, Schoeller DA. Is a calorie a calorie? Am J Clin Nutr. 2004. 79(5):899S–906S.
Article6. Lau DC, Douketis JD, Morrison KM, Hramiak IM, Sharma AM, Ur E. Obesity Canada Clinical Practice Guidelines Expert Panel. 2006 canadian clinical practice guidelines on the management and prevention of obesity in adults and children [summary]. CMAJ. 2007. 176(8):S1–S13.
Article7. Zhang Y, Guo K, LeBlanc RE, Loh D, Schwartz GJ, Yu YH. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes. 2007. 56(6):1647–1654.
Article8. Kimball SR, Jefferson LS. Regulation of protein synthesis by branched-chain amino acids. Curr Opin Clin Nutr Metab Care. 2001. 4(1):39–43.
Article9. Baum JI, O'Connor JC, Seyler JE, Anthony TG, Freund GG, Layman DK. Leucine reduces the duration of insulin-induced PI 3-kinase activity in rat skeletal muscle. Am J Physiol Endocrinol Metab. 2005. 288(1):E86–E91.
Article10. Ruderman NB. Muscle amino acid metabolism and gluconeogenesis. Annu Rev Med. 1975. 26:245–258.
Article11. Donato J Jr, Pedrosa RG, Cruzat VF, Pires IS, Tirapegui J. Effects of leucine supplementation on the body composition and protein status of rats submitted to food restriction. Nutrition. 2006. 22(5):520–527.
Article12. Kim J, Park J, Hong S, Kim MK. Effect of corn gluten and its hydrolysa te consumptions on weight reduction in rats fed a high-fat diet. Nutr Res Prac. 2009. 3(3):200–207.
Article13. Bieber LL, Markwell MA. Peroxisomal and microsomal carnitine acetyltransferases. Methods Enzymol. 1981. 71 Pt C:351–358.14. Markwell MA, McGroarty EJ, Bieber LL, Tolbert NE. The subcellular distribution of carnitine acyltransferases in mammalian liver and kidney. A new peroxisomal enzyme. J Biol Chem. 1973. 248(10):3426–3432.
Article15. Geer BW, Krochko D, Williamson JH. Ontogeny, cell distribution, and the physiological role of NADP-malic enxyme in drosophila melanogaster. Biochem Genet. 1979. 17(9-10):867–879.
Article16. Noltmann EA, Gubler CJ, Kuby SA. Glucose 6-phosphate dehydrogenase (zwischenferment). I. isolation of the crystalline enzyme from yeast. J Biol Chem. 1961. 236:1225–1230.17. Layman DK, Boileau RA, Erickson DJ, Painter JE, Shiue H, Sather C, Christou DD. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J Nutr. 2003. 133(2):411–417.
Article18. Layman DK, Evans E, Baum JI, Seyler J, Erickson DJ, Boileau RA. Dietary protein and exercise have additive effects on body composition during weight loss in adult women. J Nutr. 2005. 135(8):1903–1910.
Article19. Cota D, Proulx K, Smith KA, Kozma SC, Thomas G, Woods SC, Seeley RJ. Hypothalamic mTOR signaling regulates food intake. Science. 2006. 312(5775):927–930.
Article20. Gropper SS, Smith JL, Groff JL. Chapter 4. Carbohydrates. Advanced nutrition and human metabolism. 2005. 4th Ed. CA: Thomson Learning;94–96.21. Layman DK, Walker DA. Potential importance of leucine in treatment of obesity and the metabolic syndrome. J Nutr. 2006. 136:319S–323S.
Article22. Garlick PJ. The role of leucine in the regulation of protein metabolism. J Nutr. 2005. 135:6 Suppl. 1553S–1556S.
Article23. Rossetti L, Rothman DL, DeFronzo RA, Shulman GI. Effect of dietary protein on in vivo insulin action and liver glycogen repletion. Am J Physiol. 1989. 257(2 Pt 1):E212–E219.
Article24. Ferrannini E, Bevilacqua S, Lanzone L, Bonadonna R, Brandi L, Oleggini M, Boni C, Buzzigoli G, Ciociaro D. Metabolic interactions of amino acids and glucose in healthy humans. Diab Nutr Metab. 1988. 3:175–186.25. Krebs M, Krssak M, Bernroider E, Anderwald C, Brehm A, Meyerspeer M, Nowotny P, Roth E, Waldhausl W, Roden M. Mechanism of amino acid-induced skeletal muscle insulin resistance in humans. Diabetes. 2002. 51(3):599–605.
Article26. Piatti PM, Monti F, Fermo I, Baruffaldi L, Nasser R, Santambrogio G, Librenti MC, Galli-Kienle M, Pontiroli AE, Pozza G. Hypocaloric high-protein diet improves glucose oxidation and spares lean body mass: Comparison to hypocaloric high-carbohydrate diet. Metabolism. 1994. 43(12):1481–1487.
Article27. Parker B, Noakes M, Luscombe N, Clifton P. Effect of a high-protein, high-monounsaturated fat weight loss diet on glycemic control and lipid levels in type 2 diabetes. Diabetes Care. 2002. 25(3):425–430.
Article28. Jungas RL, Halperin ML, Brosnan JT. Quantitative analysis of amino acid oxidation and related gluconeogenesis in humans. Physiol Rev. 1992. 72(2):419–448.
Article29. Patti ME, Brambilla E, Luzi L, Landaker EJ, Kahn CR. Bidirectional modulation of insulin action by amino acids. J Clin Invest. 1998. 101(7):1519–1529.
Article30. Nishitani S, Takehana K, Fujitani S, Sonaka I. Branched-chain amino acids improve glucose metabolism in rats with liver cirrhosis. Am J Physiol Gastrointest Liver Physiol. 2005. 288(6):G1292–G1300.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of corn gluten hydrolyzates, branched chain amino acids, and leucine on body weight reduction in obese rats induced by a high fat diet
- Effect of corn gluten and its hydrolysate consumptions on weight reduction in rats fed a high-fat diet
- Effects of Liquid Culture of Coriolus Versicolor on Lipid Metabolism and Enzyme Activities in Rats Fed High Fat Diet
- Effect of Dietary Grape Pomace on Lipid Oxidation and Related Enzyme Activities in Rats Fed High Fat Diet
- Effects of Liquid Culture of Agaricus blazei Murill on Lipid Metabolism and Enzyme Activities in Rats Fed High Fat Diet