Nutr Res Pract.
2018 Aug;12(4):298-306. 10.4162/nrp.2018.12.4.298.
The effects of Brassica juncea L. leaf extract on obesity and lipid profiles of rats fed a high-fat/high-cholesterol diet
- Affiliations
-
- 1Department of Food and Nutrition, Chosun University, 309, Pilmun-daero, Dong-gu, Gwangju 61452, Korea. joominlee@chosun.ac.kr
- KMID: 2420995
- DOI: http://doi.org/10.4162/nrp.2018.12.4.298
Abstract
- BACKGROUND/OBJECTIVES
Obesity is a global health problem of significant importance which increases mortality. In place of anti-obesity drugs, natural products are being developed as alternative therapeutic materials. In this study, we investigated the effect of Brassica juncea L. leaf extract (BLE) on fat deposition and lipid profiles in high-fat, high-cholesterol diet (HFC)-induced obese rats.
MATERIALS/METHODS
Male Sprague-Dawley rats were divided into four groups (n = 8 per group) according to diet: normal diet group (ND), high-fat/high-cholesterol diet group (HFC), HFC with 3% BLE diet group (HFC-A1), and HFC with 5% BLE diet group (HFC-A2). Each group was fed for 6 weeks. Rat body and adipose tissue weights, serum biochemical parameters, and tissue lipid contents were determined. The expression levels of mRNA and proteins involved in lipid and cholesterol metabolism were determined by reverse transcription polymerase chain reaction and western blot analysis, respectively.
RESULTS
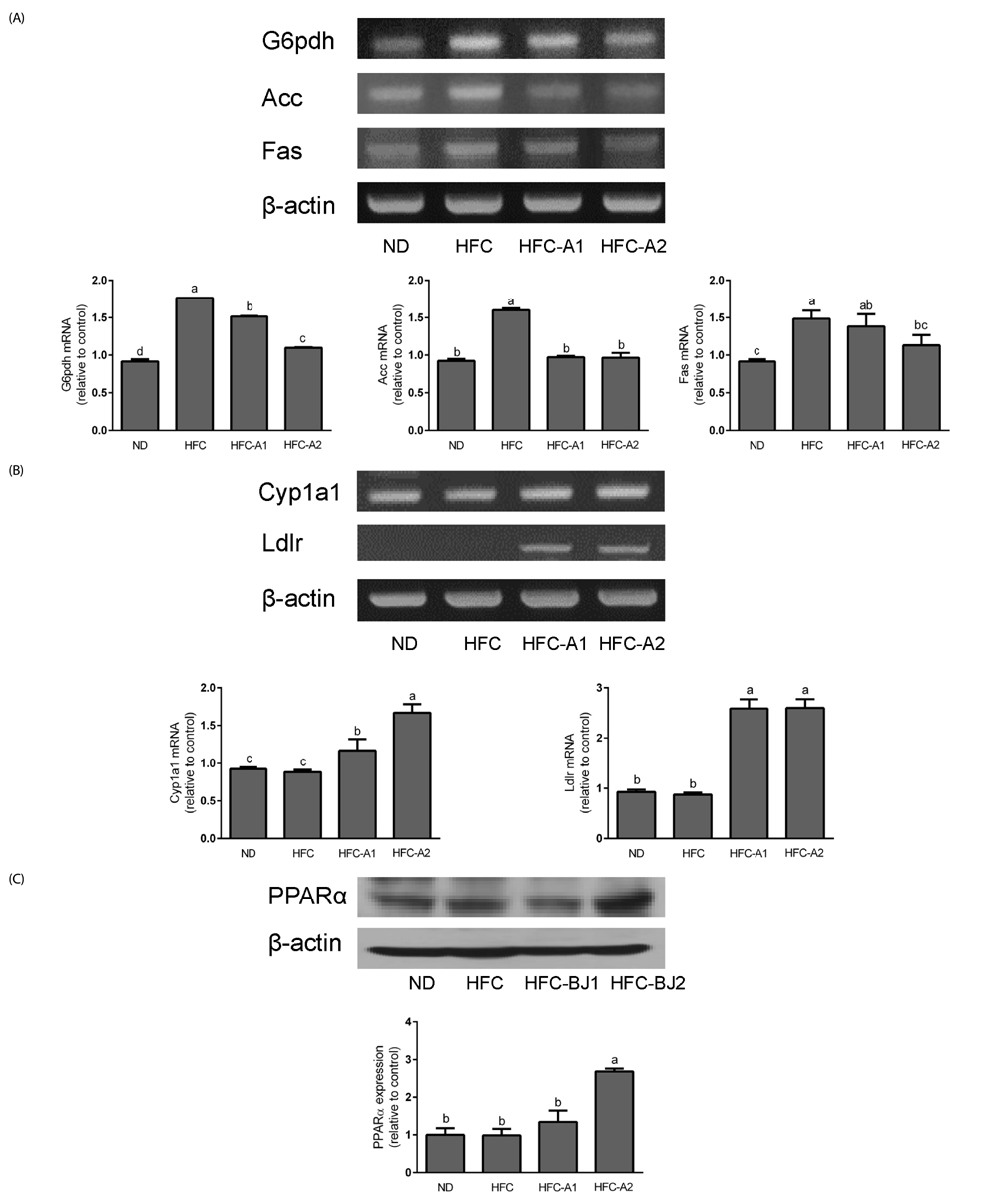
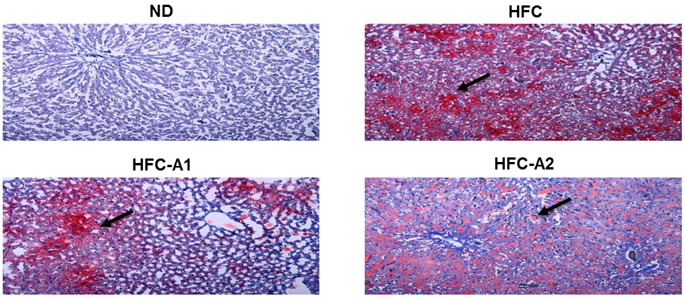
The HFC-A2 group showed significantly lower body weight gain and food efficiency ratio than the HFC group. BLE supplementation caused mesenteric, epididymal, and total adipose tissue weights to decrease. The serum levels of triglyceride, total cholesterol, and low-density lipoprotein cholesterol were significantly reduced, and high-density lipoprotein cholesterol was significantly increased in rats fed BLE. These results were related to lower glucose-6-phosphate dehydrogenase, acetyl-coA carboxylase, and fatty acid synthase mRNA expression, and to higher expression of the cholesterol 7α-hydroxylase and low density lipoprotein-receptor, as well as increased protein levels of peroxisome proliferator-activated receptor α. Histological analysis of the liver revealed decreased lipid droplets in HFC rats treated with BLE.
CONCLUSIONS
Supplementation of HFC with 3% or 5% BLE inhibited body fat accumulation, improved lipid profiles, and modulated lipogenesis- and cholesterol metabolism-related gene and protein expression.
Keyword
MeSH Terms
-
Acetyl-CoA Carboxylase
Adipose Tissue
Animals
Anti-Obesity Agents
Biological Products
Blotting, Western
Body Weight
Brassica*
Cholesterol
Diet*
Diet, High-Fat
Global Health
Glucosephosphate Dehydrogenase
Humans
Lipid Droplets
Lipoproteins
Liver
Male
Metabolism
Mortality
Mustard Plant*
Obesity*
Peroxisomes
Polymerase Chain Reaction
Rats*
Rats, Sprague-Dawley
Reverse Transcription
RNA, Messenger
Triglycerides
Weights and Measures
Acetyl-CoA Carboxylase
Anti-Obesity Agents
Biological Products
Cholesterol
Glucosephosphate Dehydrogenase
Lipoproteins
RNA, Messenger
Figure
Reference
-
1. Kopelman P. Health risks associated with overweight and obesity. Obes Rev. 2007; 8:Suppl 1. 13–17.
Article2. Michalakis K, Mintziori G, Kaprara A, Tarlatzis BC, Goulis DG. The complex interaction between obesity, metabolic syndrome and reproductive axis: a narrative review. Metabolism. 2013; 62:457–478.
Article3. Garg SK, Maurer H, Reed K, Selagamsetty R. Diabetes and cancer: two diseases with obesity as a common risk factor. Diabetes Obes Metab. 2014; 16:97–110.
Article4. Kopelman PG. Obesity as a medical problem. Nature. 2000; 404:635–643.
Article5. Karelis AD, St-Pierre DH, Conus F, Rabasa-Lhoret R, Poehlman ET. Metabolic and body composition factors in subgroups of obesity: what do we know? J Clin Endocrinol Metab. 2004; 89:2569–2575.
Article6. Esteghamati A, Mazaheri T, Vahidi Rad M, Noshad S. Complementary and alternative medicine for the treatment of obesity: a critical review. Int J Endocrinol Metab. 2015; 13:e19678.
Article7. Rucker D, Padwal R, Li SK, Curioni C, Lau DC. Long term pharmacotherapy for obesity and overweight: updated meta-analysis. BMJ. 2007; 335:1194–1199.
Article8. Kumar V, Thakur AK, Barothia ND, Chatterjee SS. Therapeutic potentials of Brassica juncea: an overview. Tang. 2011; 1:2.1–2.16.9. Manohar RP, Pushpan R, Rohini S. Mustard and its uses in Ayurveda. Indian J Tradit Knowl. 2009; 8:400–404.10. Kumar S, Andy A. Health promoting bioactive phytochemicals from Brassica. Int Food Res J. 2012; 19:59–71.11. Lee J. Physicochemical characteristics and antioxidant effects of red mustard (Brassica juncea L.) leaf using different drying methods. Korean J Community Living Sci. 2017; 28:515–524.
Article12. Hill CB, Williams PH, Carlson DG, Tookey HL. Variation in glucosinolates in oriental Brassica vegetables. J Am Soc Hortic Sci. 1987; 112:309–313.13. McNaughton SA, Marks GC. Development of a food composition database for the estimation of dietary intakes of glucosinolates, the biologically active constituents of cruciferous vegetables. Br J Nutr. 2003; 90:687–697.
Article14. Miyata S, Inoue J, Shimizu M, Sato R. Allyl isothiocyanate suppresses the proteolytic activation of sterol regulatory element-binding proteins and de novo fatty acid and cholesterol synthesis. Biosci Biotechnol Biochem. 2016; 80:1006–1011.
Article15. Choi KM, Lee YS, Kim W, Kim SJ, Shin KO, Yu JY, Lee MK, Lee YM, Hong JT, Yun YP, Yoo HS. Sulforaphane attenuates obesity by inhibiting adipogenesis and activating the AMPK pathway in obese mice. J Nutr Biochem. 2014; 25:201–207.
Article16. Thakur AK, Chatterjee SS, Kumar V. Antidepressant-like effects of Brassica juncea L. leaves in diabetic rodents. Indian J Exp Biol. 2014; 52:613–622.17. Kim HY, Yokozawa T, Cho EJ, Cheigh HS, Choi JS, Chung HY. In vitro and in vivo antioxidant effects of mustard leaf (Brassica juncea). Phytother Res. 2003; 17:465–471.18. Yokozawa T, Kim HY, Cho EJ, Yamabi N, Choi JS. Protective effects of mustard leaf (Brassica juncea) against diabetic oxidative stress. J Nutr Sci Vitaminol (Tokyo). 2003; 49:87–93.
Article19. Valvala VK, Vangipurapu RK, Banam VR, Pulukurthy UM, Turlapati NR. Effect of mustard (Brassica juncea) leaf extract on streptozotocin-induced diabetic cataract in Wistar rats. J Food Biochem. 2011; 35:109–124.20. Kwak Y, Lee J, Ju J. Anti-cancer activities of Brassica juncea leaves in vitro. EXCLI J. 2016; 15:699–710.21. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18:499–502.
Article22. Rosenfeld L. Lipoprotein analysis. Early methods in the diagnosis of atherosclerosis. Arch Pathol Lab Med. 1989; 113:1101–1110.23. Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957; 226:497–509.
Article24. Wease DF, Espinosa ES, Anderson YJ. New system for automated extraction and simultaneous determination of serum cholesterol and triglycerides. Clin Chem. 1975; 21:1430–1436.
Article25. Zlatkis A, Zak B. Study of a new cholesterol reagent. Anal Biochem. 1969; 29:143–148.
Article26. Haglund O, Luostarinen R, Wallin R, Wibell L, Saldeen T. The effects of fish oil on triglycerides, cholesterol, fibrinogen and malondialdehyde in humans supplemented with vitamin E. J Nutr. 1991; 121:165–169.
Article27. Khan BA, Abraham A, Leelamma S. Role of Murraya koenigii (curry leaf) and Brassica juncea (mustard) in lipid peroxidation. Indian J Physiol Pharmacol. 1996; 40:155–158.28. Yadav SP, Vats V, Ammini AC, Grover JK. Brassica juncea (Rai) significantly prevented the development of insulin resistance in rats fed fructose-enriched diet. J Ethnopharmacol. 2004; 93:113–116.
Article29. Park Y, Storkson JM, Liu W, Albright KJ, Cook ME, Pariza MW. Structure-activity relationship of conjugated linoleic acid and its cognates in inhibiting heparin-releasable lipoprotein lipase and glycerol release from fully differentiated 3T3-L1 adipocytes. J Nutr Biochem. 2004; 15:561–568.
Article30. Rinella ME. Nonalcoholic fatty liver disease: a systematic review. JAMA. 2015; 313:2263–2273.31. Panchal SK, Poudyal H, Iyer A, Nazer R, Alam A, Diwan V, Kauter K, Sernia C, Campbell F, Ward L, Gobe G, Fenning A, Brown L. High-carbohydrate high-fat diet-induced metabolic syndrome and cardiovascular remodeling in rats. J Cardiovasc Pharmacol. 2011; 57:51–64.
Article32. Huang GJ, Deng JS, Huang SS, Lin SS, Shao YY, Chen CC, Hou WC, Kuo YH. Protective effect of antrosterol from Antrodia camphorata submerged whole broth against carbon tetrachloride-induced acute liver injury in mice. Food Chem. 2012; 132:709–716.
Article33. Cousin B, Munoz O, Andre M, Fontanilles AM, Dani C, Cousin JL, Laharrague P, Casteilla L, Pénicaud L. A role for preadipocytes as macrophage-like cells. FASEB J. 1999; 13:305–312.
Article34. Tiniakos DG, Vos MB, Brunt EM. Nonalcoholic fatty liver disease: pathology and pathogenesis. Annu Rev Pathol. 2010; 5:145–171.
Article35. Bechmann LP, Hannivoort RA, Gerken G, Hotamisligil GS, Trauner M, Canbay A. The interaction of hepatic lipid and glucose metabolism in liver diseases. J Hepatol. 2012; 56:952–964.
Article36. Beck F, Plummer S, Senior PV, Byrne S, Green S, Brammar WJ. The ontogeny of peroxisome-proliferator-activated receptor gene expression in the mouse and rat. Proc Biol Sci. 1992; 247:83–87.
Article37. Costet P, Legendre C, Moré J, Edgar A, Galtier P, Pineau T. Peroxisome proliferator-activated receptor α-isoform deficiency leads to progressive dyslipidemia with sexually dimorphic obesity and steatosis. J Biol Chem. 1998; 273:29577–29585.
Article38. Astrup A, Buemann B, Christensen NJ, Toubro S. Failure to increase lipid oxidation in response to increasing dietary fat content in formerly obese women. Am J Physiol. 1994; 266:E592–E599.
Article39. Goldstein JL, Brown MS. The LDL receptor. Arterioscler Thromb Vasc Biol. 2009; 29:431–438.
Article40. Reena MB, Gowda LR, Lokesh BR. Enhanced hypocholesterolemic effects of interesterified oils are mediated by upregulating LDL receptor and cholesterol 7-α-hydroxylase gene expression in rats. J Nutr. 2011; 141:24–30.
Article41. Chiang JY. Bile acids: regulation of synthesis. J Lipid Res. 2009; 50:1955–1966.
Article42. Hebanowska A. Mechanisms of bile acid biosynthesis regulation-autoregulation by bile acids. Postepy Biochem. 2011; 57:314–323.43. Li T, Owsley E, Matozel M, Hsu P, Novak CM, Chiang JY. Transgenic expression of cholesterol 7α-hydroxylase in the liver prevents high-fat diet-induced obesity and insulin resistance in mice. Hepatology. 2010; 52:678–690.
Article44. Miyake JH, Duong-Polk XT, Taylor JM, Du EZ, Castellani LW, Lusis AJ, Davis RA. Transgenic expression of cholesterol-7-α-hydroxylase prevents atherosclerosis in C57BL/6J mice. Arterioscler Thromb Vasc Biol. 2002; 22:121–126.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Soyoligosaccharide on Lipid Metabolism in Rats Fed the High Fat or Low Fat Diet
- Effects of Genistein Supplementation on Fatty Liver and Lipid Metabolism in Rats Fed High Fat Diet
- Effects of Liquid Culture of Coriolus Versicolor on Lipid Metabolism and Enzyme Activities in Rats Fed High Fat Diet
- Effects of Liquid Culture of Agaricus blazei Murill on Lipid Metabolism and Enzyme Activities in Rats Fed High Fat Diet
- alpha-Lipoic acid reduced weight gain and improved the lipid profile in rats fed with high fat diet