Yonsei Med J.
2018 Mar;59(2):279-286. 10.3349/ymj.2018.59.2.279.
The Role of Serotonin in Ventricular Repolarization in Pregnant Mice
- Affiliations
-
- 1Division of Cardiology, Department of Internal Medicine, Severance Cardiovascular Hospital, Yonsei University College of Medicine, Seoul, Korea. cby6908@yuhs.ac
- 2Brain Korea 21 PLUS Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea.
- 3Department of Biochemistry and Molecular Biology, Yonsei University, Seoul, Korea.
- 4Graduate School of Medical Science and Engineering, KAIST, Daejeon, Korea.
- 5Department of Biomedical Engineering, Duke University, Durham, NC, USA.
- KMID: 2418792
- DOI: http://doi.org/10.3349/ymj.2018.59.2.279
Abstract
- PURPOSE
The mechanisms underlying repolarization abnormalities during pregnancy are not fully understood. Although maternal serotonin (5-hydroxytryptamine, 5-HT) production is an important determinant for normal fetal development in mice, its role in mothers remains unclear. We evaluated the role of serotonin in ventricular repolarization in mice hearts via 5Htr3 receptor (Htr3a) and investigated the mechanism of QT-prolongation during pregnancy.
MATERIALS AND METHODS
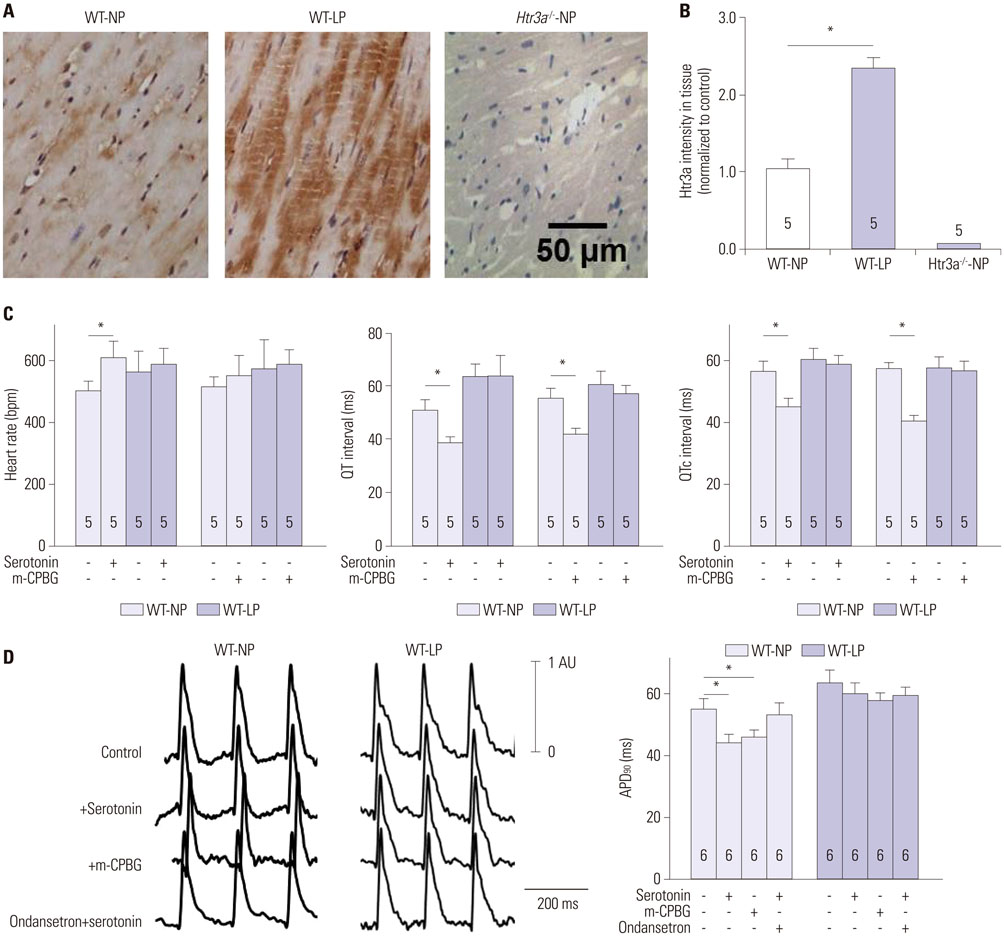
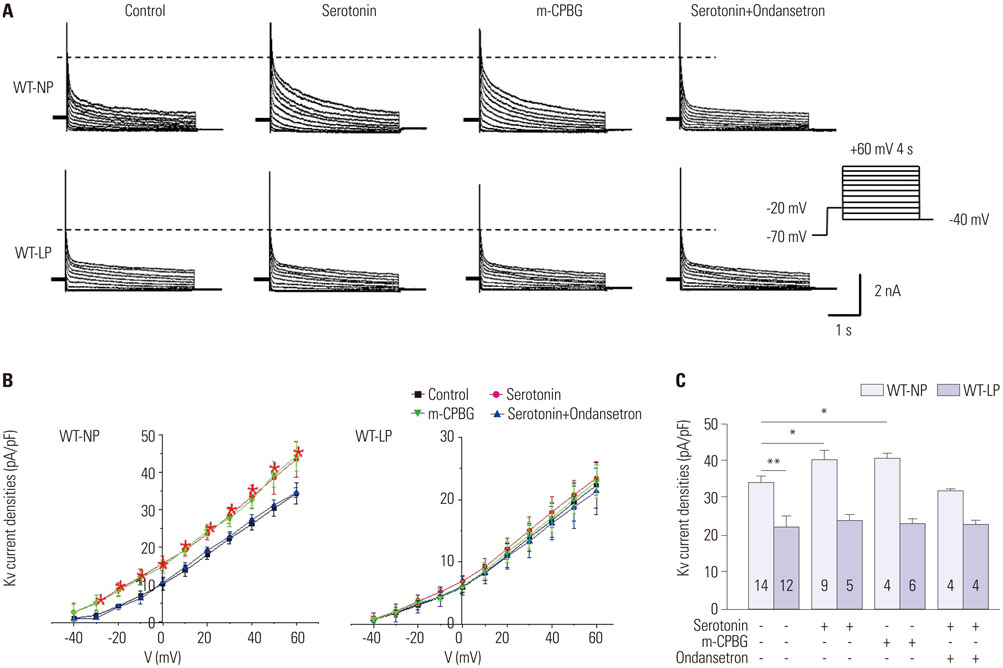
We measured current amplitudes and the expression levels of voltage-gated K⺠(Kv) channels in freshly-isolated left ventricular myocytes from wild-type non-pregnant (WT-NP), late-pregnant (WT-LP), and non-pregnant Htr3a homozygous knockout mice (Htr3a(−/−)-NP).
RESULTS
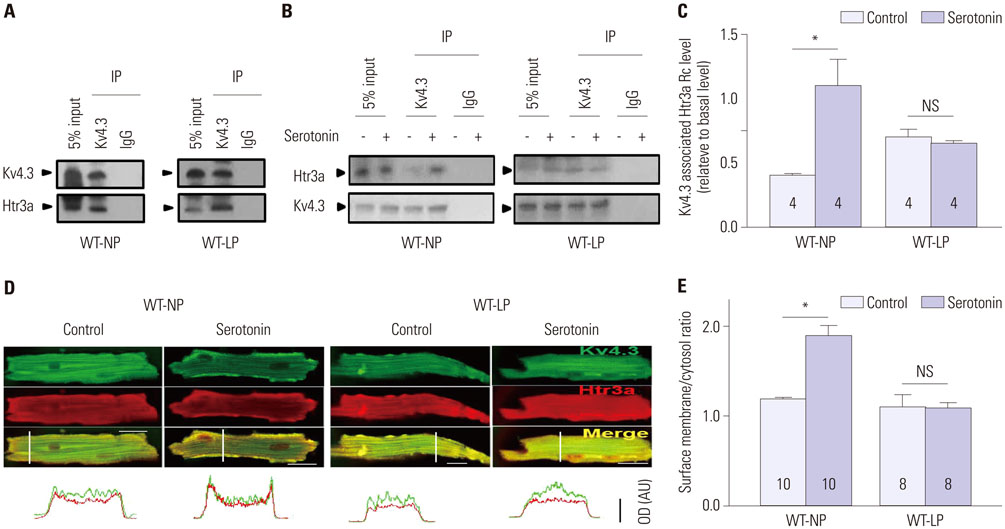
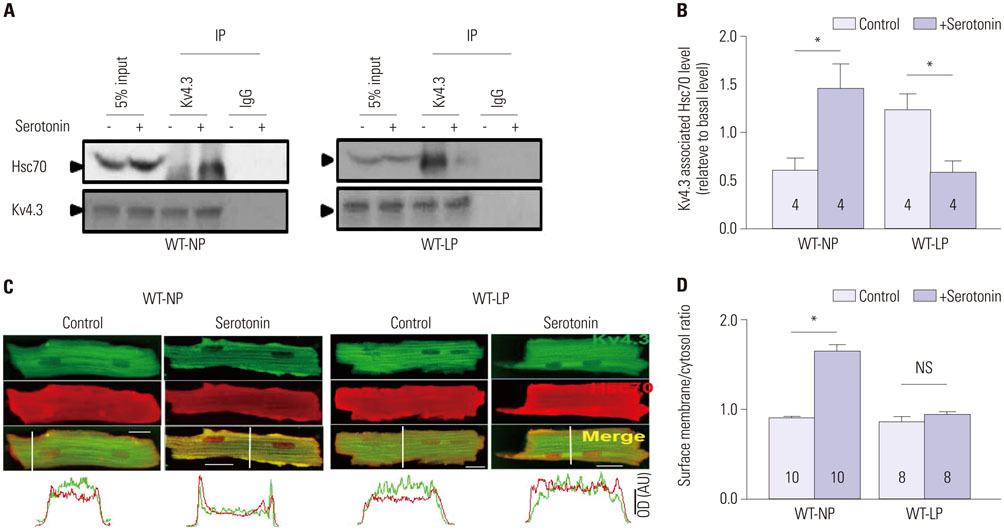
During pregnancy, serotonin and tryptophan hydroxylase 1, a rate-limiting enzyme for the synthesis of serotonin, were markedly increased in hearts and serum. Serotonin increased Kv current densities concomitant with the shortening of the QT interval in WT-NP mice, but not in WT-LP and Htr3a(−/−)-NP mice. Ondansetron, an Htr3 antagonist, decreased Kv currents in WT-LP mice, but not in WT-NP mice. Kv4.3 directly interacted with Htr3a, and this binding was facilitated by serotonin. Serotonin increased the trafficking of Kv4.3 channels to the cellular membrane in WT-NP.
CONCLUSION
Serotonin increases repolarizing currents by augmenting Kv currents. Elevated serotonin levels during pregnancy counterbalance pregnancy-related QT prolongation by facilitating Htr3-mediated Kv currents.
Keyword
MeSH Terms
-
*Action Potentials/drug effects
Animals
Cell Membrane/drug effects/metabolism
Disease Models, Animal
Electrocardiography
Female
HSC70 Heat-Shock Proteins/metabolism
HSP90 Heat-Shock Proteins/metabolism
Heart Ventricles/drug effects/*metabolism
Mice, Inbred C57BL
Mice, Knockout
Myocytes, Cardiac/drug effects/metabolism
Potassium Channels/metabolism
Pregnancy
Rabbits
Rats, Sprague-Dawley
Receptors, Serotonin, 5-HT3/metabolism
Serotonin/*metabolism
Serotonin 5-HT3 Receptor Agonists/pharmacology
HSC70 Heat-Shock Proteins
HSP90 Heat-Shock Proteins
Potassium Channels
Receptors, Serotonin, 5-HT3
Serotonin 5-HT3 Receptor Agonists
Serotonin
Figure
Reference
-
1. Pham TV, Rosen MR. Sex, hormones, and repolarization. Cardiovasc Res. 2002; 53:740–751.
Article2. Odening KE, Choi BR, Liu GX, Hartmann K, Ziv O, Chaves L, et al. Estradiol promotes sudden cardiac death in transgenic long QT type 2 rabbits while progesterone is protective. Heart Rhythm. 2012; 9:823–832.
Article3. Sauer AJ, Moss AJ, McNitt S, Peterson DR, Zareba W, Robinson JL, et al. Long QT syndrome in adults. J Am Coll Cardiol. 2007; 49:329–337.
Article4. Eghbali M, Deva R, Alioua A, Minosyan TY, Ruan H, Wang Y, et al. Molecular and functional signature of heart hypertrophy during pregnancy. Circ Res. 2005; 96:1208–1216.
Article5. Fiorica-Howells E, Maroteaux L, Gershon MD. Serotonin and the 5-HT(2B) receptor in the development of enteric neurons. J Neurosci. 2000; 20:294–305.6. Buznikov GA, Lambert HW, Lauder JM. Serotonin and serotonin-like substances as regulators of early embryogenesis and morphogenesis. Cell Tissue Res. 2001; 305:177–186.
Article7. Yavarone MS, Shuey DL, Tamir H, Sadler TW, Lauder JM. Serotonin and cardiac morphogenesis in the mouse embryo. Teratology. 1993; 47:573–584.
Article8. Côté F, Fligny C, Bayard E, Launay JM, Gershon MD, Mallet J, et al. Maternal serotonin is crucial for murine embryonic development. Proc Natl Acad Sci U S A. 2007; 104:329–334.
Article9. Havrilla PL, Kane-Gill SL, Verrico MM, Seybert AL, Reis SE. Coronary vasospasm and atrial fibrillation associated with ondansetron therapy. Ann Pharmacother. 2009; 43:532–536.
Article10. Park H, Oh CM, Park J, Park H, Cui S, Kim HS, et al. Deletion of the serotonin receptor type 3A in mice leads to sudden cardiac death during pregnancy. Circ J. 2015; 79:1807–1815.
Article11. Xu H, Guo W, Nerbonne JM. Four kinetically distinct depolarization-activated K+ currents in adult mouse ventricular myocytes. J Gen Physiol. 1999; 113:661–678.
Article12. Anneken L, Baumann S, Vigneault P, Biliczki P, Friedrich C, Xiao L, et al. Estradiol regulates human QT-interval: acceleration of cardiac repolarization by enhanced KCNH2 membrane trafficking. Eur Heart J. 2016; 37:640–650.
Article13. Lehmann MH, Hardy S, Archibald D, quart B, MacNeil DJ. Sex difference in risk of torsade de pointes with d,l-sotalol. Circulation. 1996; 94:2535–2541.
Article14. Zhang Y, Ouyang P, Post WS, Dalal D, Vaidya D, Blasco-Colmenares E, et al. Sex-steroid hormones and electrocardiographic QT-interval duration: findings from the third National Health and Nutrition Examination Survey and the Multi-Ethnic Study of Atherosclerosis. Am J Epidemiol. 2011; 174:403–411.
Article15. Zhou Z, Gong Q, Epstein ML, January CT. HERG channel dysfunction in human long QT syndrome. Intracellular transport and functional defects. J Biol Chem. 1998; 273:21061–21066.16. Ficker E, Dennis AT, Wang L, Brown AM. Role of the cytosolic chaperones Hsp70 and Hsp90 in maturation of the cardiac potassium channel HERG. Circ Res. 2003; 92:e87–100.
Article17. Walker VE, Atanasiu R, Lam H, Shrier A. Co-chaperone FKBP38 promotes HERG trafficking. J Biol Chem. 2007; 282:23509–23516.
Article18. Kuryshev YA, Brown AM, Wang L, Benedict CR, Rampe D. Interactions of the 5-hydroxytryptamine 3 antagonist class of antiemetic drugs with human cardiac ion channels. J Pharmacol Exp Ther. 2000; 295:614–620.19. Nerbonne JM, Nichols CG, Schwarz TL, Escande D. Genetic manipulation of cardiac K(+) channel function in mice: what have we learned, and where do we go from here. Circ Res. 2001; 89:944–956.20. Eghbali M, Wang Y, Toro L, Stefani E. Heart hypertrophy during pregnancy: a better functioning heart. Trends Cardiovasc Med. 2006; 16:285–291.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Ondansetron Inhibits Voltage-Gated K⺠Current of Ventricular Myocytes from Pregnant Mouse
- Early Repolarization Syndrome with Idiopathic Ventricular Fibrillation
- Changes in ventricular repolarization after balloon valvuloplasty in patients with pulmonary stenosis
- Left Ventricular Anomalous Muscle Band and Electrocardiographic Repolarization Changes in a 5 Year Old Girl
- Dispersion of QT Interval and Other Repolarization Indexes in Acute Myocardial Infarction