Intest Res.
2018 Jul;16(3):467-474. 10.5217/ir.2018.16.3.467.
Clinical significance of carcinoembryonic antigen in peritoneal fluid detected during operation in stage I–III colorectal cancer patients
- Affiliations
-
- 1Department of Gastroenterology, Kosin University College of Medicine, Busan, Korea. parksj6406@daum.net
- 2Department of Colorectal Surgery, Kosin University College of Medicine, Busan, Korea.
- KMID: 2417659
- DOI: http://doi.org/10.5217/ir.2018.16.3.467
Abstract
- BACKGROUND/AIMS
Early diagnosis of peritoneal metastases in patients with colorectal cancer (CRC) can influence patient prognosis. The aim of this study was to identify the clinical significance of carcinoembryonic antigen (CEA) in peritoneal fluid detected during operation in stage I-III CRC patients.
METHODS
Between April 2009 and April 2015, we reviewed medical records from a total of 60 stage I-III CRC patients who had peritoneal fluid collected during operation. Patients who had positive cytology in the assessment of peritoneal fluid were excluded. We evaluated the values of CEA in peritoneal fluid (pCEA) to predict the long-term outcomes of these patients using Kaplan-Meier curves and Cox regression models.
RESULTS
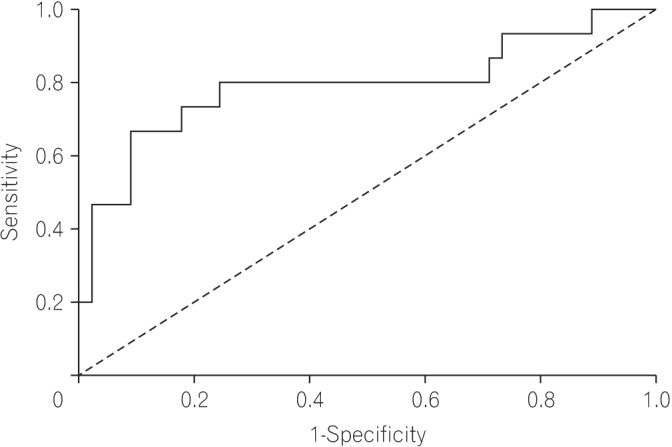
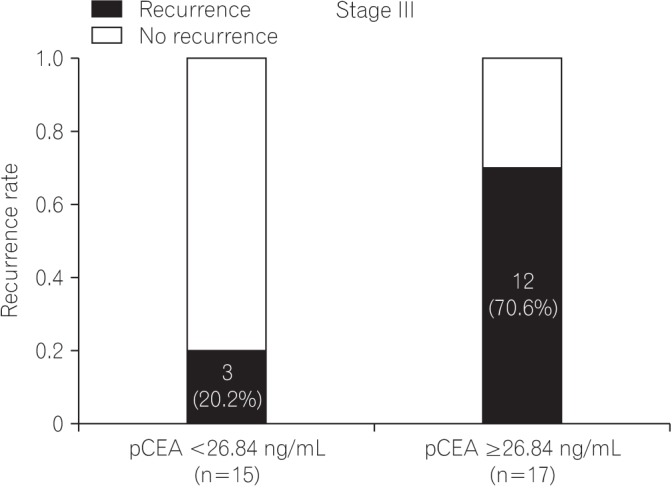
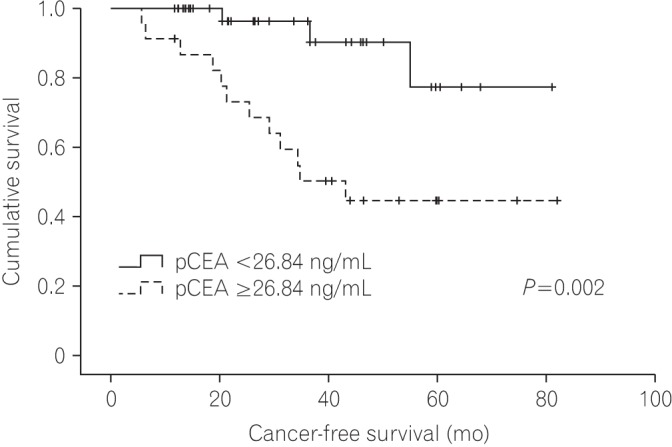
The median follow-up duration was 37 months (interquartile range, 21-50 months). On receiver operating characteristic analysis, pCEA had the largest area under the curve (0.793; 95% confidence interval, 0.635-0.950; P=0.001) with an optimal cutoff value of 26.84 (sensitivity, 80.0%; specificity, 76.6%) for predicting recurrence. The recurrence rate was 8.1% in patients with low pCEA ( < 26.84 ng/mL, n=37), and 52.2% in patients with high pCEA (≥26.84 ng/mL, n=23). In multivariate Cox regression analysis, high pCEA (≥26.84 ng/mL) was a risk factor for poor cancer-free survival (CFS) in stage I-III patients.
CONCLUSIONS
In this study, we determined that high pCEA (≥26.84 ng/mL) detected during operation was helpful for the prediction of poor CFS in patients with stage I-III CRC.
MeSH Terms
Figure
Reference
-
1. Edwards BK, Ward E, Kohler BA, et al. Annual report to the nation on the status of cancer, 1975–2006, featuring colorectal cancer trends and impact of interventions (risk factors, screening, and treatment) to reduce future rates. Cancer. 2010; 116:544–573. PMID: 19998273.
Article2. Ferlay J, Autier P, Boniol M, Heanue M, Colombet M, Boyle P. Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol. 2007; 18:581–592. PMID: 17287242.
Article3. Brodsky JT, Cohen AM. Peritoneal seeding following potentially curative resection of colonic carcinoma: implications for adjuvant therapy. Dis Colon Rectum. 1991; 34:723–727. PMID: 1855433.
Article4. Segelman J, Granath F, Holm T, Machado M, Mahteme H, Martling A. Incidence, prevalence and risk factors for peritoneal carcinomatosis from colorectal cancer. Br J Surg. 2012; 99:699–705. PMID: 22287157.
Article5. Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136:E359–E386. DOI: 10.1002/ijc.29210. PMID: 25220842.
Article6. Loupakis F, Cremolini C, Masi G, et al. Initial therapy with FOLFOXIRI and bevacizumab for metastatic colorectal cancer. N Engl J Med. 2014; 371:1609–1618. PMID: 25337750.
Article7. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66:7–30. PMID: 26742998.
Article8. Lemmens VE, Klaver YL, Verwaal VJ, Rutten HJ, Coebergh JW, de Hingh IH. Predictors and survival of synchronous peritoneal carcinomatosis of colorectal origin: a population-based study. Int J Cancer. 2011; 128:2717–2725. PMID: 20715167.
Article9. Chu DZ, Lang NP, Thompson C, Osteen PK, Westbrook KC. Peritoneal carcinomatosis in nongynecologic malignancy: a prospective study of prognostic factors. Cancer. 1989; 63:364–367. PMID: 2910444.
Article10. Runyon BA. Care of patients with ascites. N Engl J Med. 1994; 330:337–342. PMID: 8277955.
Article11. Sangisetty SL, Miner TJ. Malignant ascites: a review of prognostic factors, pathophysiology and therapeutic measures. World J Gastrointest Surg. 2012; 4:87–95. PMID: 22590662.
Article12. Norton JA. Carcinoembryonic antigen: new applications for an old marker. Ann Surg. 1991; 213:95–97. PMID: 1992947.
Article13. Kaleta EJ, Tolan NV, Ness KA, O'Kane D, Algeciras-Schimnich A. CEA, AFP and CA 19-9 analysis in peritoneal fluid to differentiate causes of ascites formation. Clin Biochem. 2013; 46:814–818. PMID: 23454392.
Article14. Liu F, Kong X, Dou Q, et al. Evaluation of tumor markers for the differential diagnosis of benign and malignant ascites. Ann Hepatol. 2014; 13:357–363. PMID: 24756011.
Article15. Ahadi M, Tehranian S, Memar B, et al. Diagnostic value of carcinoembryonic antigen in malignancy-related ascites: systematic review and meta-analysis. Acta Gastroenterol Belg. 2014; 77:418–424. PMID: 25682632.16. Lee IK, Kim DH, Gorden DL, et al. Prognostic value of CEA and CA 19-9 tumor markers combined with cytology from peritoneal fluid in colorectal cancer. Ann Surg Oncol. 2009; 16:861–870. PMID: 19189191.
Article17. Kanellos I, Zacharakis E, Kanellos D, Pramateftakis MG, Betsis D. Prognostic significance of CEA levels and positive cytology in peritoneal washings in patients with colorectal cancer. Colorectal Dis. 2006; 8:436–440. PMID: 16684089.
Article18. Nishikawa T, Sunami E, Tanaka T, et al. Incidence and prognostic significance of positive peritoneal lavage in colorectal cancer. Surg Today. 2015; 45:1073–1081. PMID: 25373362.
Article19. Hess KR, Varadhachary GR, Taylor SH, et al. Metastatic patterns in adenocarcinoma. Cancer. 2006; 106:1624–1633. PMID: 16518827.
Article20. Bosanquet DC, Harris DA, Evans MD, Beynon J. Systematic review an meta-analysis of intraoperative peritoneal lavage for colorectal cancer staging. Br J Surg. 2013; 100:853–862. PMID: 23536330.
Article21. Passot G, Mohkam K, Cotte E, Glehen O. Intra-operative peritoneal lavage for colorectal cancer. World J Gastroenterol. 2014; 20:1935–1939. PMID: 24616569.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Clinical Significance of Carcinoembryonic Antigen and CA72-4 Assays of Peritoneal Fluid in Colorectal Carcinomas
- Individualized Cutoff Value of the Serum Carcinoembryonic Antigen Level According to TNM Stage in Colorectal Cancer
- Serum Carcinoembryonic Antigen for Recurrence in Colorectal Cancer Patients
- Clinical Significance of Detection of Carcinoembryonic Antigen in Peritoneal Fluid by Reverse Transcription-Polymerase Chain Reaction in Patients with Gastric Cancer
- Changes of serum carcinoembryonic antigen in patients with colorectal cancer