J Vet Sci.
2016 Jun;17(2):137-144. 10.4142/jvs.2016.17.2.137.
Time-course changes of hippocalcin expression in the mouse hippocampus following pilocarpine-induced status epilepticus
- Affiliations
-
- 1Department of Pharmacy, College of Pharmacy, Dankook University, Cheonan 31116, Korea. anaphy@dankook.ac.kr
- KMID: 2413162
- DOI: http://doi.org/10.4142/jvs.2016.17.2.137
Abstract
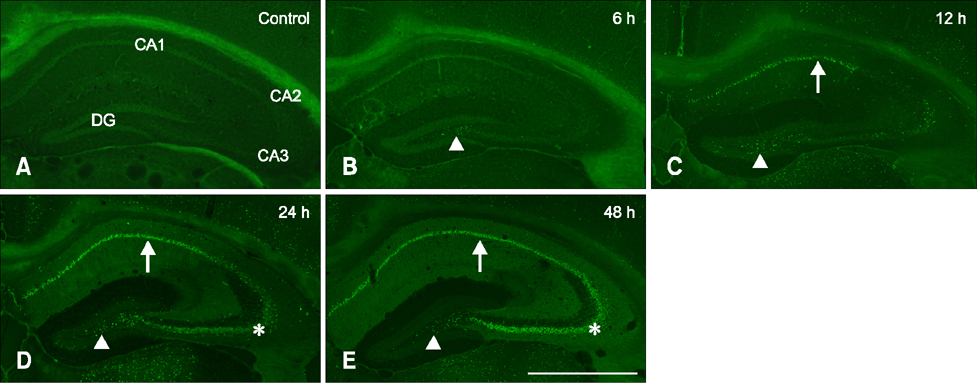
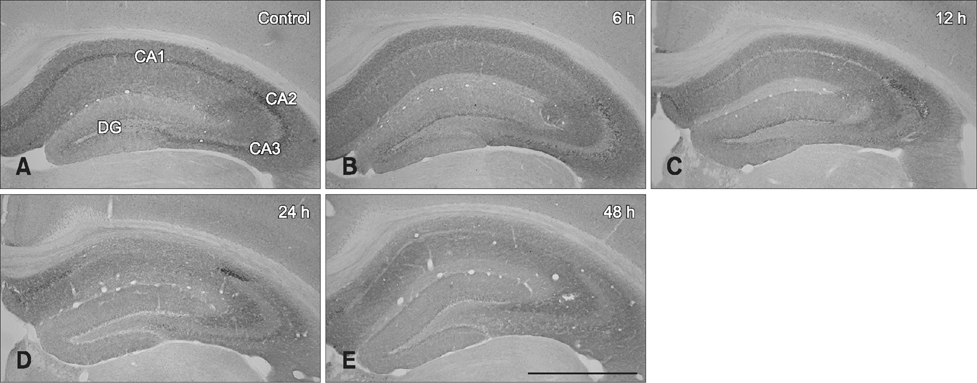
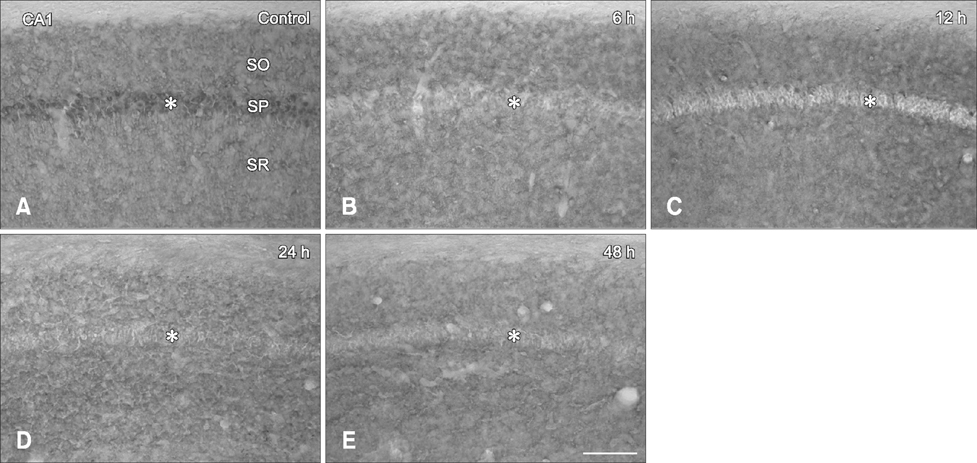
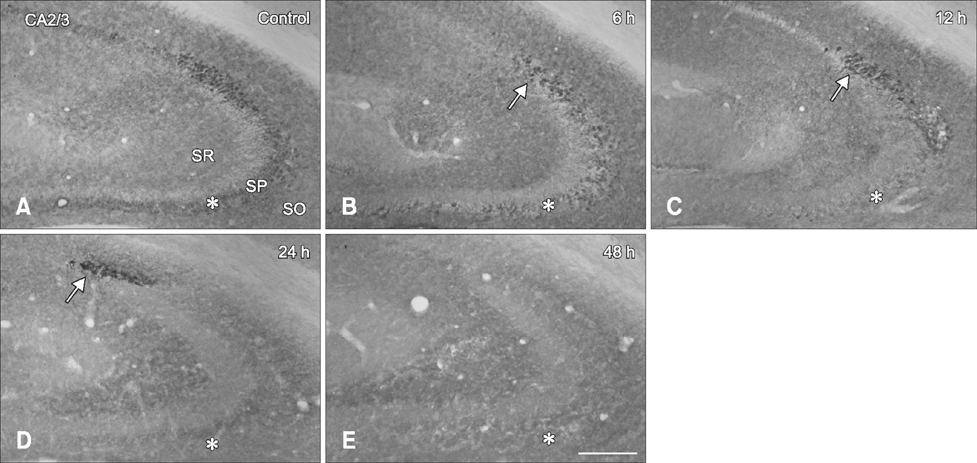
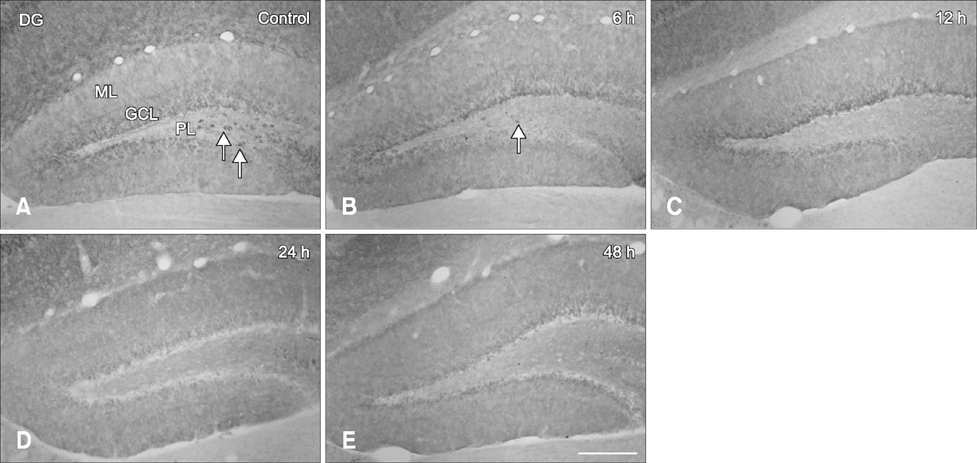
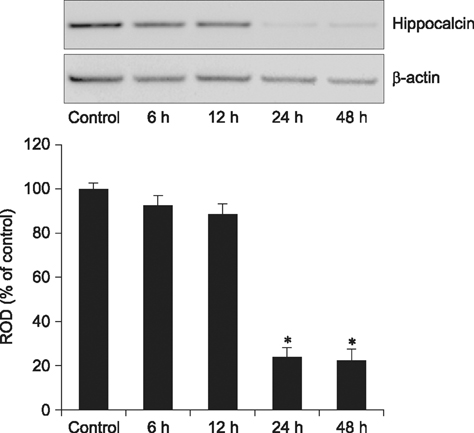
- Hippocalcin participates in the maintenance of neuronal calcium homeostasis. In the present study, we examined the time-course changes of neuronal degeneration and hippocalcin protein level in the mouse hippocampus following pilocarpine-induced status epilepticus (SE). Marked neuronal degeneration was observed in the hippocampus after SE in a time-dependent manner, although neuronal degeneration differed according to the hippocampal subregions. Almost no hippocalcin immunoreactivity was detected in the pyramidal neurons of the cornu ammonis 1 (CA1) region from 6 h after SE. However, many pyramidal neurons in the CA2 region showed hippocalcin immunoreactivity until 24 h after SE. In the CA3 region, only a few hippocalcin immunoreactive cells were observed at 12 h after SE, and almost no hippocalcin immunoreactivity was observed in the pyramidal neurons from 24 h after SE. Hippocalcin immunoreactivity in the polymorphic cells of the dentate gyrus was markedly decreased from 6 h after SE. In addition, hippocalcin protein level in the hippocampus began to decrease from 6 h after SE, and was significantly decreased at 24 h and 48 h after pilocarpine-induced SE. These results indicate that marked reduction of hippocalcin level may be closely related to neuronal degeneration in the hippocampus following pilocarpine-induced SE.
MeSH Terms
Figure
Reference
-
1. Al Sufiani F, Ang LC. Neuropathology of temporal lobe epilepsy. Epilepsy Res Treat. 2012; 2012:624519.
Article2. Borges K, Gearing M, McDermott DL, Smith AB, Almonte AG, Wainer BH, Dingledine R. Neuronal and glial pathological changes during epileptogenesis in the mouse pilocarpine model. Exp Neurol. 2003; 182:21–34.
Article3. Burgoyne RD, Weiss JL. The neuronal calcium sensor family of Ca2+-binding proteins. Biochem J. 2001; 353:1–12.4. Curia G, Longo D, Biagini G, Jones RS, Avoli M. The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods. 2008; 172:143–157.
Article5. Dichter MA. Emerging insights into mechanisms of epilepsy: implications for new antiepileptic drug development. Epilepsia. 1994; 35:Suppl 4. S51–S57.
Article6. do Nascimento AL, Dos Santos NF, Campos Pelágio F, Aparecida Teixeira S, de Moraes Ferrari EA, Langone F. Neuronal degeneration and gliosis time-course in the mouse hippocampal formation after pilocarpine-induced status epilepticus. Brain Res. 2012; 1470:98–110.
Article7. Duarte FS, Hoeller AA, Duzzioni M, Gavioli EC, Canteras NS, De Lima TCM. NK1 receptors antagonism of dorsal hippocampus counteract the anxiogenic-like effects induced by pilocarpine in non-convulsive Wistar rats. Behav Brain Res. 2014; 265:53–60.
Article8. Engel J Jr. ILAE classification of epilepsy syndromes. Epilepsy Res. 2006; 70:Suppl 1. S5–S10.
Article9. Franklin KBJ, Paxinos G. The Mouse Brain in Stereotaxic Coordinates. San Diego: Academic Press;1997.10. Kim JH, Lee DW, Choi BY, Sohn M, Lee SH, Choi HC, Song HK, Suh SW. Cytidine 5'-diphosphocholine (CDP-choline) adversely effects on pilocarpine seizure-induced hippocampal neuronal death. Brain Res. 2015; 1595:156–165.
Article11. Kobayashi M, Takamatsu K, Saitoh S, Noguchi T. Myristoylation of hippocalcin is linked to its calcium-dependent membrane association properties. J Biol Chem. 1993; 268:18898–18904.
Article12. Koh PO. Melatonin regulates the calcium-buffering proteins, parvalbumin and hippocalcin, in ischemic brain injury. J Pineal Res. 2012; 53:358–365.
Article13. Korhonen L, Hansson I, Kukkonen JP, Brännvall K, Kobayashi M, Takamatsu K, Lindholm D. Hippocalcin protects against caspase-12-induced and age-dependent neuronal degeneration. Mol Cell Neurosci. 2005; 28:85–95.
Article14. Lasoń W, Chlebicka M, Rejdak K. Research advances in basic mechanisms of seizures and antiepileptic drug action. Pharmacol Rep. 2013; 65:787–801.
Article15. Lee CH, Park JH, Yoo KY, Choi JH, Hwang IK, Ryu PD, Kim DH, Kwon YG, Kim YM, Won MH. Pre- and post-treatments with escitalopram protect against experimental ischemic neuronal damage via regulation of BDNF expression and oxidative stress. Exp Neurol. 2011; 229:450–459.
Article16. Masuo Y, Ogura A, Kobayashi M, Masaki T, Furuta Y, Ono T, Takamatsu K. Hippocalcin protects hippocampal neurons against excitotoxin damage by enhancing calcium extrusion. Neuroscience. 2007; 145:495–504.
Article17. Mercer EA, Korhonen L, Skoglösa Y, Olsson PA, Kukkonen JP, Lindholm D. NAIP interacts with hippocalcin and protects neurons against calcium-induced cell death through caspase-3-dependent and -independent pathways. EMBO J. 2000; 19:3597–3607.
Article18. Palmer CL, Lim W, Hastie PGR, Toward M, Korolchuk VI, Burbidge SA, Banting G, Collingridge GL, Isaac JTR, Henley JM. Hippocalcin functions as a calcium sensor in hippocampal LTD. Neuron. 2005; 47:487–494.
Article19. Poirier JL, Capek R, De Koninck Y. Differential progression of Dark Neuron and Fluoro-Jade labelling in the rat hippocampus following pilocarpine-induced status epilepticus. Neuroscience. 2000; 97:59–68.
Article20. Racine RJ. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol. 1972; 32:281–294.
Article21. Ravizza T, Rizzi M, Perego C, Richichi C, Velísková J, Moshé SL, De Simoni MG, Vezzani A. Inflammatory response and glia activation in developing rat hippocampus after status epilepticus. Epilepsia. 2005; 46:Suppl 5. 113–117.
Article22. Saitoh S, Takamatsu K, Kobayashi M, Noguchi T. Distribution of hippocalcin mRNA and immunoreactivity in rat brain. Neurosci Lett. 1993; 157:107–110.
Article23. Schmued LC, Hopkins KJ. Fluoro-Jade B: a high affinity fluorescent marker for the localization of neuronal degeneration. Brain Res. 2000; 874:123–130.
Article24. Smolders I, Khan GM, Manil J, Ebinger G, Michotte Y. NMDA receptor-mediated pilocarpine-induced seizures: characterization in freely moving rats by microdialysis. Br J Pharmacol. 1997; 121:1171–1179.
Article25. Syntichaki P, Tavernarakis N. The biochemistry of neuronal necrosis: rogue biology? Nat Rev Neurosci. 2003; 4:672–684.
Article26. Tzingounis AV, Kobayashi M, Takamatsu K, Nicoll RA. Hippocalcin gates the calcium activation of the slow afterhyperpolarization in hippocampal pyramidal cells. Neuron. 2007; 53:487–493.
Article27. Wang L, Liu YH, Huang YG, Chen LW. Time-course of neuronal death in the mouse pilocarpine model of chronic epilepsy using Fluoro-Jade C staining. Brain Res. 2008; 1241:157–167.
Article28. Weiergräber M, Stephani U, Köhling R. Voltage-gated calcium channels in the etiopathogenesis and treatment of absence epilepsy. Brain Res Rev. 2010; 62:245–271.
Article29. Yu SP, Canzoniero LMT, Choi DW. Ion homeostasis and apoptosis. Curr Opin Cell Biol. 2001; 13:405–411.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Spatiotemporal expression of RCAN1 and its isoform RCAN1-4 in the mouse hippocampus after pilocarpine-induced status epilepticus
- Neuroprotective effect of lithium after pilocarpine-induced status epilepticus in mice
- Differential Expression of Activating Transcription Factor-2 and c-Jun in the Immature and Adult Rat Hippocampus Following Lithium-Pilocarpine Induced Status Epilepticus
- Antiepileptic and Neuroprotective Effect of Ketamine in Lithium-Pilocarpine Induced Status Epilepticus Rat Model
- Effect of MK-801 on Neuronal Cell Loss and Fos Expression of Hippocampus in Lithium-Pilocarpine Induced Status Epilepticus