Korean J Physiol Pharmacol.
2018 Mar;22(2):203-213. 10.4196/kjpp.2018.22.2.203.
Hypoxia-dependent mitochondrial fission regulates endothelial progenitor cell migration, invasion, and tube formation
- Affiliations
-
- 1Department of Physiology, Laboratory for Vascular Medicine and Stem Cell Biology, Convergence Stem Cell Research Center, Medical Research Institute, Pusan National University School of Medicine, Yangsan 50612, Korea. smkwon323@pusan.ac.kr
- 2School of Life Sciences, Ulsan National Institute of Science and Technology, Ulsan 44919, Korea.
- 3Department of Medical Science, Pusan National University School of Medicine, Yangsan 50612, Korea.
- 4Division of Hemato-Oncology, Department of Internal Medicine, Pusan National University Hospital Medical Research Institute, Busan 49241, Korea. hemon@pusan.ac.kr
- KMID: 2410110
- DOI: http://doi.org/10.4196/kjpp.2018.22.2.203
Abstract
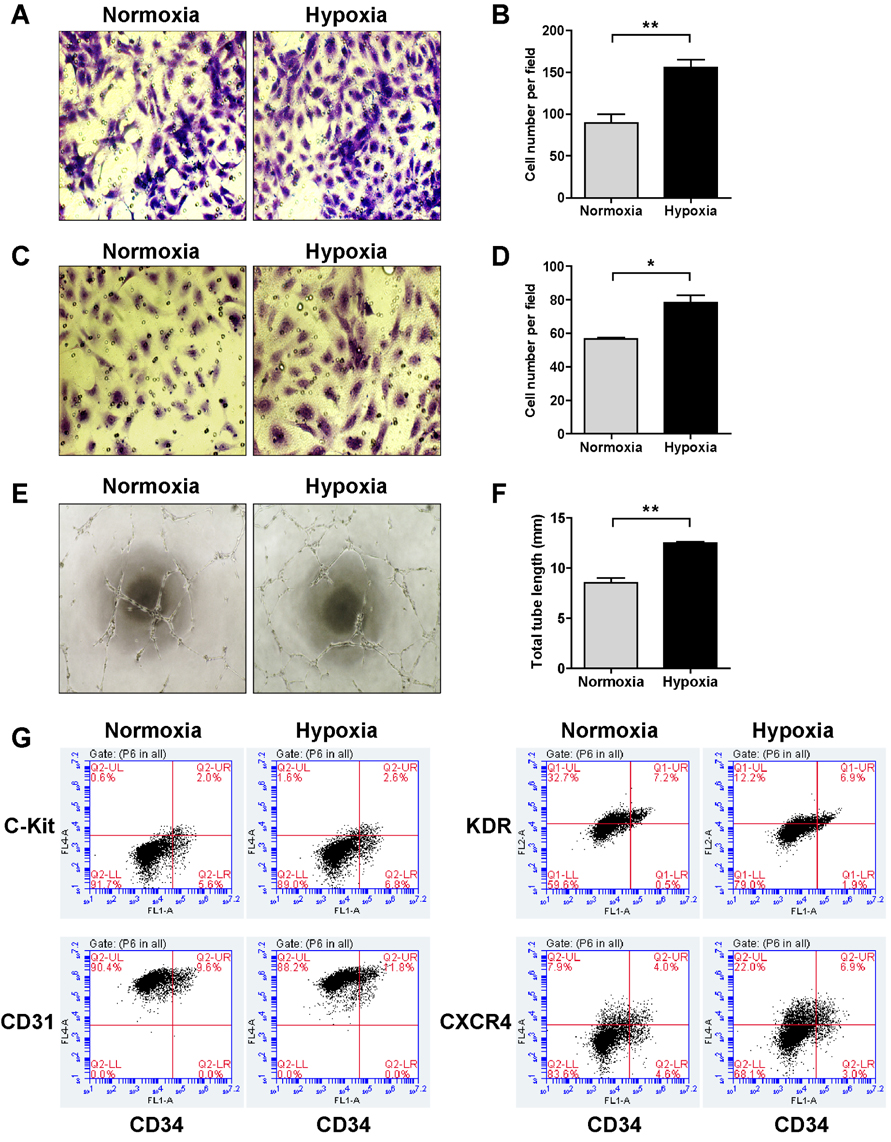
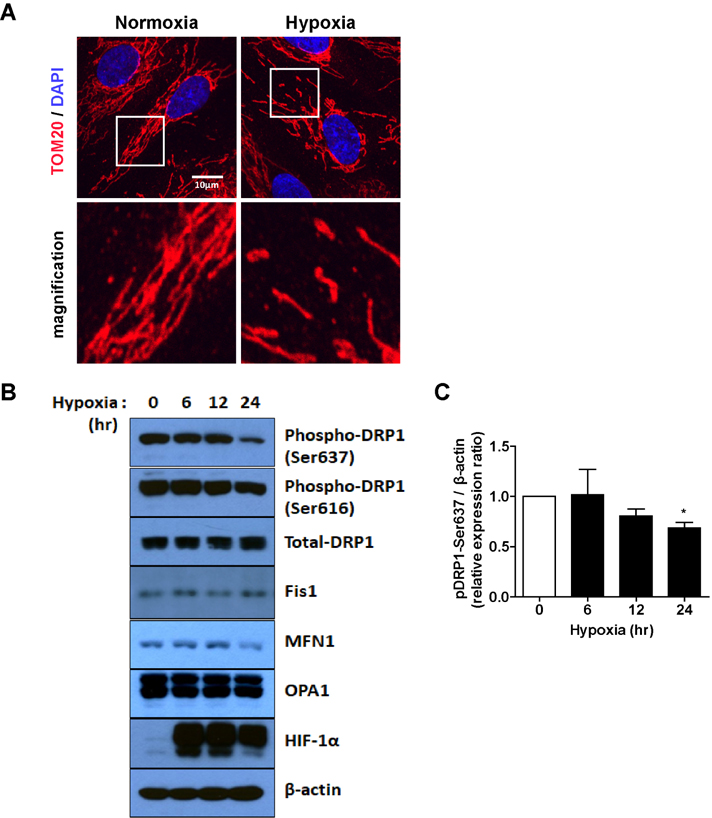
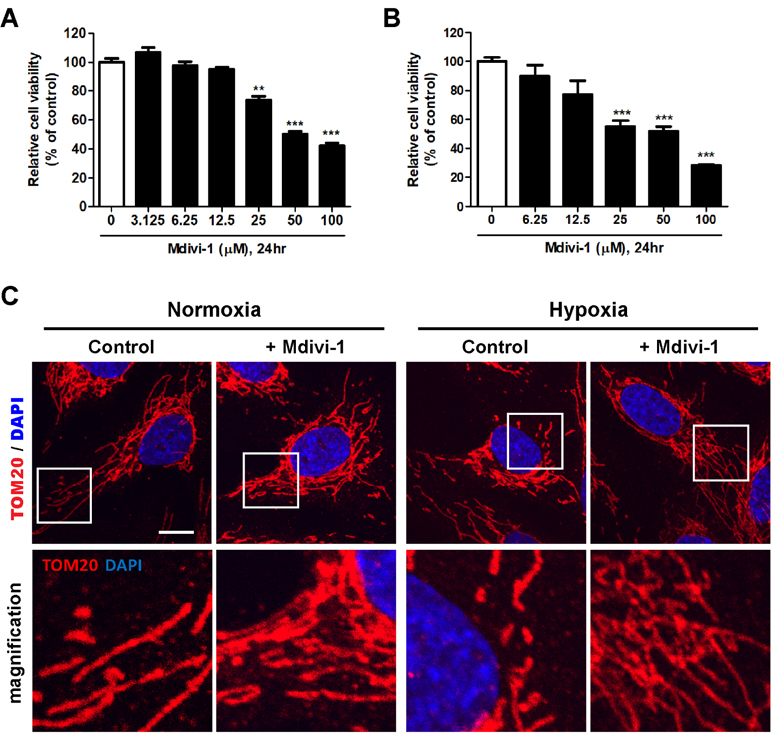
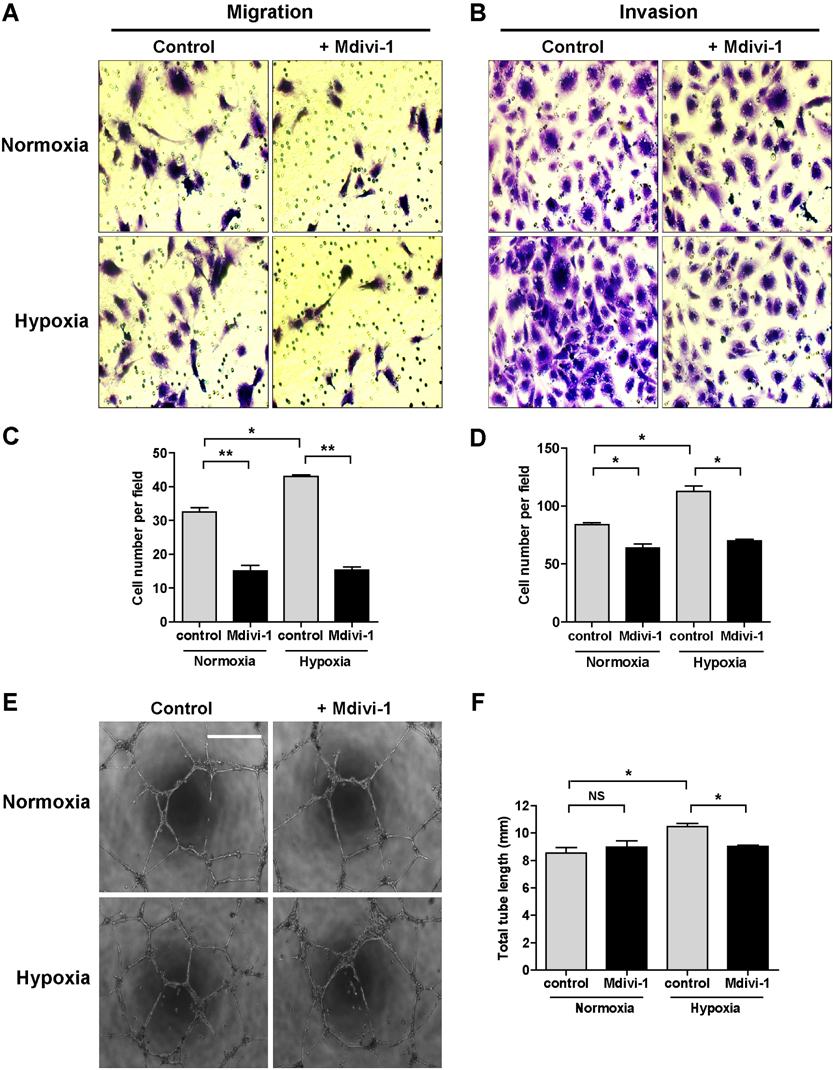
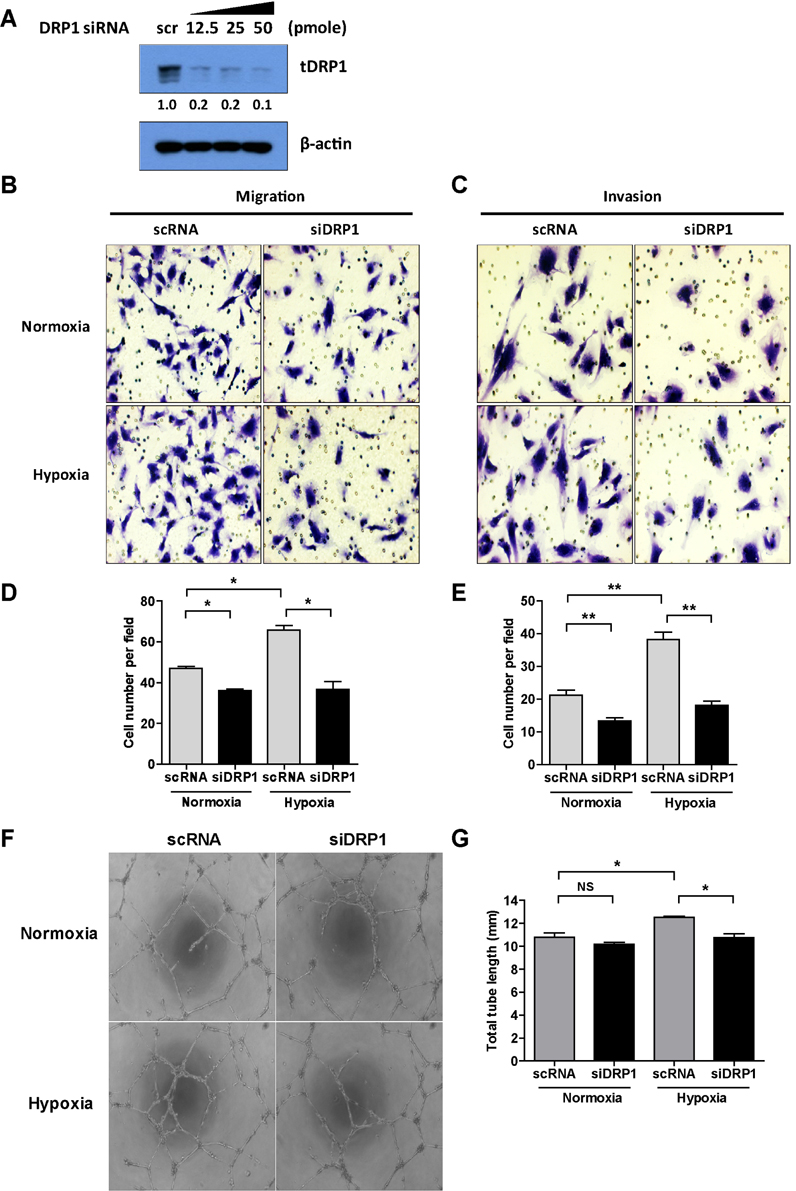
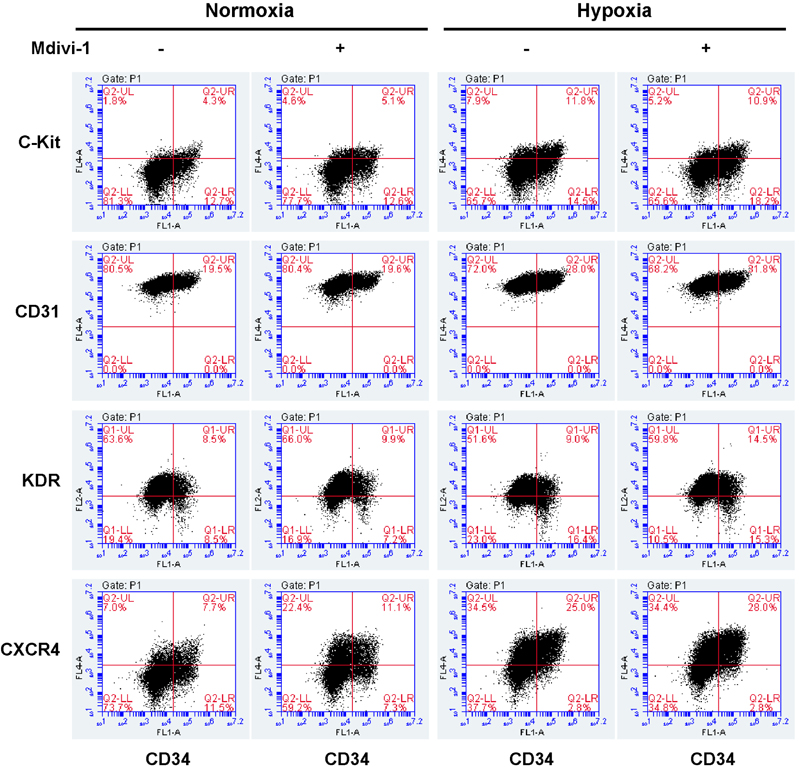
- Tumor undergo uncontrolled, excessive proliferation leads to hypoxic microenvironment. To fulfill their demand for nutrient, and oxygen, tumor angiogenesis is required. Endothelial progenitor cells (EPCs) have been known to the main source of angiogenesis because of their potential to differentiation into endothelial cells. Therefore, understanding the mechanism of EPC-mediated angiogenesis in hypoxia is critical for development of cancer therapy. Recently, mitochondrial dynamics has emerged as a critical mechanism for cellular function and differentiation under hypoxic conditions. However, the role of mitochondrial dynamics in hypoxia-induced angiogenesis remains to be elucidated. In this study, we demonstrated that hypoxia-induced mitochondrial fission accelerates EPCs bioactivities. We first investigated the effect of hypoxia on EPC-mediated angiogenesis. Cell migration, invasion, and tube formation was significantly increased under hypoxic conditions; expression of EPC surface markers was unchanged. And mitochondrial fission was induced by hypoxia time-dependent manner. We found that hypoxia-induced mitochondrial fission was triggered by dynamin-related protein Drp1, specifically, phosphorylated DRP1 at Ser637, a suppression marker for mitochondrial fission, was impaired in hypoxia time-dependent manner. To confirm the role of DRP1 in EPC-mediated angiogenesis, we analyzed cell bioactivities using Mdivi-1, a selective DRP1 inhibitor, and DRP1 siRNA. DRP1 silencing or Mdivi-1 treatment dramatically reduced cell migration, invasion, and tube formation in EPCs, but the expression of EPC surface markers was unchanged. In conclusion, we uncovered a novel role of mitochondrial fission in hypoxia-induced angiogenesis. Therefore, we suggest that specific modulation of DRP1-mediated mitochondrial dynamics may be a potential therapeutic strategy in EPC-mediated tumor angiogenesis.
MeSH Terms
Figure
Cited by 1 articles
-
Advanced tube formation assay using human endothelial colony forming cells for
in vitro evaluation of angiogenesis
Hyunsook Lee, Kyu-Tae Kang
Korean J Physiol Pharmacol. 2018;22(6):705-712. doi: 10.4196/kjpp.2018.22.6.705.
Reference
-
1. Ichihara E, Kiura K, Tanimoto M. Targeting angiogenesis in cancer therapy. Acta Med Okayama. 2011; 65:353–362.2. Zhao Y, Adjei AA. Targeting angiogenesis in cancer therapy: moving beyond vascular endothelial growth factor. Oncologist. 2015; 20:660–673.
Article3. Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, Witzenbichler B, Schatteman G, Isner JM. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997; 275:964–967.
Article4. Medina RJ, O'Neill CL, Sweeney M, Guduric-Fuchs J, Gardiner TA, Simpson DA, Stitt AW. Molecular analysis of endothelial progenitor cell (EPC) subtypes reveals two distinct cell populations with different identities. BMC Med Genomics. 2010; 3:18.
Article5. Hager G, Holnthoner W, Wolbank S, Husa AM, Godthardt K, Redl H, Gabriel C. Three specific antigens to isolate endothelial progenitor cells from human liposuction material. Cytotherapy. 2013; 15:1426–1435.
Article6. Marçola M, Rodrigues CE. Endothelial progenitor cells in tumor angiogenesis: another brick in the wall. Stem Cells Int. 2015; 2015:832649.
Article7. Ni HM, Williams JA, Ding WX. Mitochondrial dynamics and mitochondrial quality control. Redox Biol. 2015; 4:6–13.
Article8. Wai T, Langer T. Mitochondrial dynamics and metabolic regulation. Trends Endocrinol Metab. 2016; 27:105–117.
Article9. Otera H, Ishihara N, Mihara K. New insights into the function and regulation of mitochondrial fission. Biochim Biophys Acta. 2013; 1833:1256–1268.
Article10. Gao AW, Cantó C, Houtkooper RH. Mitochondrial response to nutrient availability and its role in metabolic disease. EMBO Mol Med. 2014; 6:580–589.
Article11. Mitra K, Rikhy R, Lilly M, Lippincott-Schwartz J. DRP1-dependent mitochondrial fission initiates follicle cell differentiation during Drosophila oogenesis. J Cell Biol. 2012; 197:487–497.
Article12. Luchsinger LL, de Almeida MJ, Corrigan DJ, Mumau M, Snoeck HW. Mitofusin 2 maintains haematopoietic stem cells with extensive lymphoid potential. Nature. 2016; 529:528–531.
Article13. Wolin MS. Novel role for the regulation of mitochondrial fission by hypoxia inducible factor-1α in the control of smooth muscle remodeling and progression of pulmonary hypertension. Circ Res. 2012; 110:1395–1397.
Article14. Wu W, Lin C, Wu K, Jiang L, Wang X, Li W, Zhuang H, Zhang X, Chen H, Li S, Yang Y, Lu Y, Wang J, Zhu R, Zhang L, Sui S, Tan N, Zhao B, Zhang J, Li L, Feng D. FUNDC1 regulates mitochondrial dynamics at the ER-mitochondrial contact site under hypoxic conditions. EMBO J. 2016; 35:1368–1384.15. Yu T, Sheu SS, Robotham JL, Yoon Y. Mitochondrial fission mediates high glucose-induced cell death through elevated production of reactive oxygen species. Cardiovasc Res. 2008; 79:341–351.
Article16. Sheridan C, Martin SJ. Mitochondrial fission/fusion dynamics and apoptosis. Mitochondrion. 2010; 10:640–648.
Article17. Han XJ, Yang ZJ, Jiang LP, Wei YF, Liao MF, Qian Y, Li Y, Huang X, Wang JB, Xin HB, Wan YY. Mitochondrial dynamics regulates hypoxia-induced migration and antineoplastic activity of cisplatin in breast cancer cells. Int J Oncol. 2015; 46:691–700.
Article18. Kwon SM, Lee JH, Lee SH, Jung SY, Kim DY, Kang SH, Yoo SY, Hong JK, Park JH, Kim JH, Kim SW, Kim YJ, Lee SJ, Kim HG, Asahara T. Cross talk with hematopoietic cells regulates the endothelial progenitor cell differentiation of CD34 positive cells. PLoS One. 2014; 9:e106310.
Article19. Sanderson TH, Raghunayakula S, Kumar R. Neuronal hypoxia disrupts mitochondrial fusion. Neuroscience. 2015; 301:71–78.
Article20. Neary MT, Ng KE, Ludtmann MH, Hall AR, Piotrowska I, Ong SB, Hausenloy DJ, Mohun TJ, Abramov AY, Breckenridge RA. Hypoxia signaling controls postnatal changes in cardiac mitochondrial morphology and function. J Mol Cell Cardiol. 2014; 74:340–352.
Article21. Bertholet AM, Delerue T, Millet AM, Moulis MF, David C, Daloyau M, Arnauné-Pelloquin L, Davezac N, Mils V, Miquel MC, Rojo M, Belenguer P. Mitochondrial fusion/fission dynamics in neurodegeneration and neuronal plasticity. Neurobiol Dis. 2016; 90:3–19.
Article22. Shen T, Wang N, Yu X, Shi J, Li Q, Zhang C, Fu L, Wang S, Xing Y, Zheng X, Yu L, Zhu D. The critical role of dynamin-related protein 1 in hypoxia-induced pulmonary vascular angiogenesis. J Cell Biochem. 2015; 116:1993–2007.
Article23. Zhao J, Zhang J, Yu M, Xie Y, Huang Y, Wolff DW, Abel PW, Tu Y. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene. 2013; 32:4814–4824.
Article24. Lin JR, Shen WL, Yan C, Gao PJ. Downregulation of dynamin-related protein 1 contributes to impaired autophagic flux and angiogenic function in senescent endothelial cells. Arterioscler Thromb Vasc Biol. 2015; 35:1413–1422.
Article25. Kashatus JA, Nascimento A, Myers LJ, Sher A, Byrne FL, Hoehn KL, Counter CM, Kashatus DF. Erk2 phosphorylation of Drp1 promotes mitochondrial fission and MAPK-driven tumor growth. Mol Cell. 2015; 57:537–551.
Article26. Kim H, Scimia MC, Wilkinson D, Trelles RD, Wood MR, Bowtell D, Dillin A, Mercola M, Ronai ZA. Fine-tuning of Drp1/Fis1 availability by AKAP121/Siah2 regulates mitochondrial adaptation to hypoxia. Mol Cell. 2011; 44:532–544.
Article27. Marsboom G, Toth PT, Ryan JJ, Hong Z, Wu X, Fang YH, Thenappan T, Piao L, Zhang HJ, Pogoriler J, Chen Y, Morrow E, Weir EK, Rehman J, Archer SL. Dynamin-related protein 1-mediated mitochondrial mitotic fission permits hyperproliferation of vascular smooth muscle cells and offers a novel therapeutic target in pulmonary hypertension. Circ Res. 2012; 110:1484–1497.
Article28. Campello S, Lacalle RA, Bettella M, Mañes S, Scorrano L, Viola A. Orchestration of lymphocyte chemotaxis by mitochondrial dynamics. J Exp Med. 2006; 203:2879–2886.
Article29. Wang J, Hansen K, Edwards R, Van Houten B, Qian W. Mitochondrial division inhibitor 1 (mdivi-1) enhances death receptor-mediated apoptosis in human ovarian cancer cells. Biochem Biophys Res Commun. 2015; 456:7–12.
Article30. Cassidy-Stone A, Chipuk JE, Ingerman E, Song C, Yoo C, Kuwana T, Kurth MJ, Shaw JT, Hinshaw JE, Green DR, Nunnari J. Chemical inhibition of the mitochondrial division dynamin reveals its role in Bax/Bak-dependent mitochondrial outer membrane permeabilization. Dev Cell. 2008; 14:193–204.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- High Glucose Causes Human Cardiac Progenitor Cell Dysfunction by Promoting Mitochondrial Fission: Role of a GLUT1 Blocker
- Expression of alpha3beta1 Integrin in ECV304 Endothelial Cells and Angiogenesis
- Hsp20 Promotes Endothelial Progenitor Cell Angiogenesis via Activation of PI3K/Akt Signaling Pathway under Hypoxia
- Physiological and Pathological Significance of Dynamin-Related Protein 1 (Drp1)-Dependent Mitochondrial Fission in the Nervous System
- Restoring mitochondrial dynamics in neuronal health through photobiomodulation